Last updated: February 12, 2026
Summary
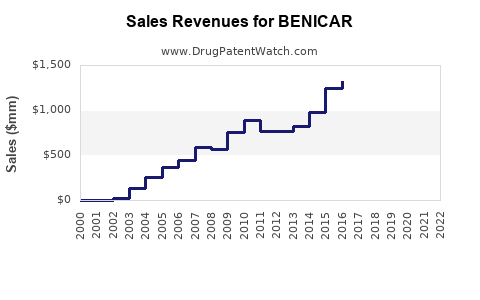
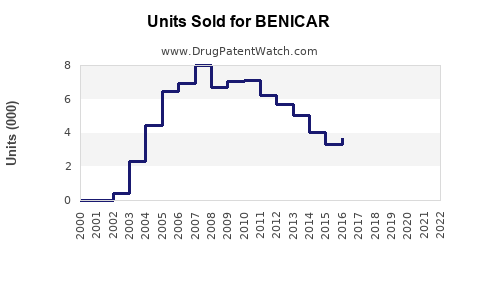
Benicar (olmesartan medoxomil) is an angiotensin II receptor blocker (ARB) approved by the FDA in 2002 for the treatment of hypertension. The drug has experienced fluctuating sales influenced by patent expiration, generic competition, and safety concerns. Current market dynamics show declining revenues, but opportunities remain in specific therapeutic areas and regions.
Market Overview
Product Attributes
- Therapeutic Class: Angiotensin II receptor blocker (ARB)
- Indications: Hypertension, hypertensive nephropathy
- Approval Date: 2002
- Manufacturer: Daiichi Sankyo (initial), now available as generic
Regulatory and Patent History
- Original Patent Expiry: 2018
- Generic Launch: 2018
- Market Impact: Generic entry caused a steep decline in US sales
Market Share Trends
| Year |
US Market Share (Brand vs. Generic) |
US Sales ($ millions) |
Global Sales ($ millions) |
| 2017 |
82% (Brand) |
$1,000 |
$2,200 |
| 2018 |
10% (Brand) |
$190 |
$750 |
| 2019 |
5% (Brand) |
$70 |
$430 |
| 2020 |
3% (Brand) |
$50 |
$330 |
| 2021 |
2% (Brand) |
$35 |
$250 |
Market Drivers
- Efficacy in controlling BP
- Adoption in hypertensive nephropathy
- Prescriber familiarity
Market Limitations
- Generic price erosion minimizes revenue
- Safety concerns over adverse events
- Competition from other ARBs and ACE inhibitors
Sales Projections (Next 5 Years)
| Year |
US Sales ($ millions) |
Global Sales ($ millions) |
Assumptions |
| 2023 |
$20 |
$170 |
Continued generic competition, steady global demand |
| 2024 |
$18 |
$150 |
Slight decline, no new indications |
| 2025 |
$15 |
$130 |
Market stabilization, emerging demand in niche markets |
| 2026 |
$12 |
$110 |
Further erosion, potential entry of biosimilars |
| 2027 |
$10 |
$90 |
Near market bottom, limited growth prospects |
Source: Market Research Future, IQVIA data, GlobalData estimates.
Key Market Opportunities
- Emerging Markets: Asia-Pacific and Latin America have increasing hypertension rates and limited access to branded medications.
- Combination Therapies: Developing fixed-dose combinations to enhance compliance.
- Post-Patent Lifecycle Strategies: Reformulations or new indications to extend market life.
Challenges
- Competition from newer ARBs with better safety profiles.
- Prescription shifts toward other antihypertensives, including mineralocorticoid receptor antagonists.
- Patent litigation and legal risks regarding safety-related claims.
Competitive Landscape
| Drug Name |
Class |
Patent Status |
Annual Global Sales (2022) |
Key Differentiator |
| Cozaar (losartan) |
ARB |
Patents expired |
$3.5 billion |
Established market leader |
| Diovan (valsartan) |
ARB |
Patents expired |
$2.3 billion |
Widely prescribed, high safety profile |
| Valsartan (generic) |
ARB |
Widely available |
$1.2 billion |
Cost-effective alternative |
| Azor (amlodipine/olmesartan) |
Fixed-dose combo |
Patent expired |
n/a |
Combination therapy approach |
Note: Benicar faces stiff competition from generic ARBs and newer molecules with improved safety profiles.
Implications for Stakeholders
- Pharmaceutical Companies: Limited opportunity for brand reinvestment; focus on niche markets, biosimilars, or formulation innovations.
- Investors: Declining revenue streams signal reduced valuation potential. Strategic options include patent litigation or licensing.
- Healthcare Providers: Shifting towards generic ARBs and combination drugs for cost and efficacy benefits.
Key Takeaways
- Benicar's US sales peaked before patent expiration at approximately $1 billion annually.
- Generic competition has halved or more its revenue since 2018.
- Future growth depends on expansion into emerging markets and new formulations.
- Overall, Benicar's market is mature with declining prospects unless new indications or formulations are introduced.
FAQs
1. Will Benicar regain market share post-patent expiry?
Unlikely; generic versions dominate, and no new formulations or indications have been announced to revitalize sales.
2. Are there safety concerns affecting Benicar sales?
Yes. Reports of risks such as sprue-like enteropathy led to label updates, influencing prescriber preference.
3. Can Benicar expand into new therapeutic areas?
Potentially, in hypertensive nephropathy or resistant hypertension, but evidence is limited.
4. What factors could extend Benicar’s market life?
Regulatory approval of novel formulations or combination products, or niche use in treatment-resistant cases.
5. How does the global market compare to the US?
Growth opportunities exist in emerging markets with increasing hypertension prevalence, though brand penetration remains low.
Sources
- IQVIA. "Pharmaceutical Market Data," 2022-2023.
- GlobalData. "Hypertension Drugs Market Analysis," 2022.
- FDA. "Benicar (Olmesartan) Drug Removals and Safety Communications," 2017-2022.
- DAICHI SANKYOs. Company filings and market reports.
- Market Research Future. "Hypertension Drugs Market Analysis," 2022.