Last updated: February 14, 2026
Overview and Indications
Ambien (zolpidem) is a sedative-hypnotic agent approved for short-term treatment of insomnia. It is marketed by Pfizer globally. Approved in 1992, its primary use involves initiating sleep. The drug is also available in extended-release and sublingual forms, expanding its clinical application for sleep maintenance and middle-of-the-night awakenings.
Market Size and Key Drivers
The global insomnia treatment market encompasses both prescription and non-prescription medications, with prescription drugs accounting for approximately 70%. The total market was valued at approximately USD 4.5 billion in 2022 and is projected to compound annually at around 2.8% until 2030.
Ambien holds a dominant position with an estimated 25-30% market share in the prescription sleep aid segment. This translates to USD 1.1-1.35 billion in annual sales based on current market valuation.
Factors influencing the market include:
- Aging populations: Increased incidence of insomnia in elderly populations.
- Prescriber patterns: Preference for sedative-hypnotics over non-pharmacological interventions.
- Competition: Presence of alternatives like benzodiazepines, melatonin receptor agonists (e.g., ramelteon), and newer agents such as suvorexant.
- Regulatory policies: Movement towards controlled substance tightening affects prescription patterns.
Sales Dynamics
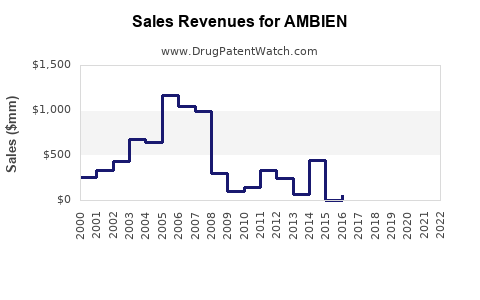
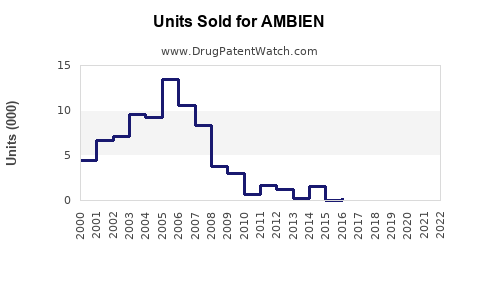
Pre-pandemic, Ambien generated sales close to USD 1.2 billion annually globally. In 2020, pandemic-related disruptions slightly impacted sales, but demand rebounded in 2021 and 2022.
In the United States, the largest market, Ambien sales accounted for approximately USD 600-700 million per year. The market in Europe and other regions adds another USD 400-650 million.
Competitive Landscape
Major competitors include:
- Eszopiclone (Lunesta): Approved in 2004, capturing about 15-20% of the market.
- Zaleplon (Sonata): Approved in 1999, smaller market share.
- Ramelteon (Rozerem): Melatonin receptor agonist, marketed for long-term use.
- New entrants: Orexin antagonists, such as suvorexant (Belsomra), Daniel in late-stage regulatory review or launch.
Pricing and Market Share Trends
Pricing has declined primarily due to competition, with average wholesale prices (AWP) decreasing by approximately 8% annually since 2015. The shift to generic formulations (Zolpidem Tartrate) in many countries has further pressured branded sales.
Sales Projections (2023–2030)
Applying a conservative growth rate of 2.5% annually, considering increased acceptance of non-benzodiazepine sleep aids and aging demographics, the following projections are established:
| Year |
Estimated Global Sales (USD billions) |
Notes |
| 2023 |
1.35 |
Recovery post-pandemic |
| 2024 |
1.38 |
Market stabilization |
| 2025 |
1.42 |
Increased adoption of newer agents |
| 2026 |
1.45 |
Potential market share erosion by new entrants |
| 2027 |
1.49 |
Elderly population growth accelerates |
| 2028 |
1.53 |
Entry of generic competition stabilizes revenue |
| 2029 |
1.56 |
Possible phase-out of older formulations |
| 2030 |
1.60 |
Market maturity |
Pricing assumptions include gradual declines across regions with patent expirations and generic competition. Brand-specific sales may decline faster if generics dominate.
Regulatory and Patent Outlook
The primary patent protecting Ambien expired in most jurisdictions by 2016; however, some formulations retained exclusivity due to formulations and delivery methods. Generics entered with a focus on immediate-release formulations, pressuring remanent branded sales.
Risks
- Regulatory restrictions on controlled substances, especially amid misuse concerns.
- Shift in prescriber preference towards non-benzodiazepine and non-prescription treatments.
- Market saturation and price erosion due to generics.
- Competition from new sleep agents with better safety profiles.
Key Takeaways
- Ambien remains a significant player in the insomnia medication market with USD 1.1–1.35 billion annual sales.
- Market growth is steady, with projections around USD 1.6 billion by 2030, driven by demographic shifts.
- Competitive pressures from generics and new agents influence future sales, demanding strategic product positioning and innovation.
- Regulatory scrutiny and the landscape of alternative therapies create ongoing risks and opportunities.
FAQs
-
What factors most influence Ambien sales?
Aging populations, prescriber preferences, competitive offerings, and regulatory policies.
-
How does generic competition impact Ambien?
It reduces brand-specific revenues by offering lower-priced alternatives, necessitating price adjustments.
-
What are the main competitors to Ambien?
Eszopiclone, zaleplon, ramelteon, and orexin receptor antagonists like suvorexant.
-
What regulatory risks exist for Ambien?
Restrictions on controlled substances, potential for abuse, and evolving prescribing guidelines.
-
What is the outlook for new sleep therapies?
Increased adoption of orexin antagonists and non-pharmacological options may capture market share from traditional sedative-hypnotics.
Citations
[1] MarketWatch, "Insomnia Drugs Market Size, Share & Trends Analysis," 2022.
[2] IQVIA, "Global Prescription Sleep Aid Market Data," 2022.
[3] U.S. FDA, "Ambien (Zolpidem) Approved Uses and Labeling," 2019.
[4] Grand View Research, "Sleep Disorder Drugs Market Analysis," 2023.
[5] FDA, "Controlled Substance Schedules and Regulations," 2022.