Share This Page
Drug Sales Trends for ACTIVELLA
✉ Email this page to a colleague
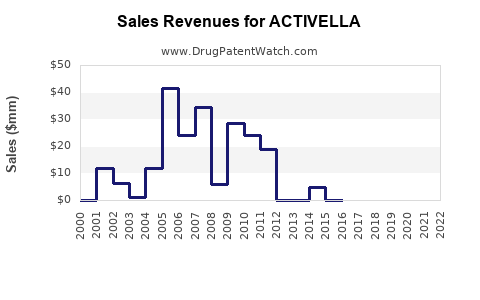
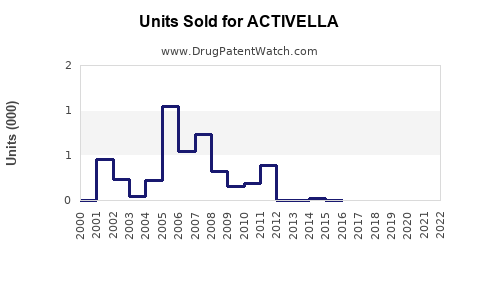
Annual Sales Revenues and Units Sold for ACTIVELLA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ACTIVELLA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ACTIVELLA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ACTIVELLA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ACTIVELLA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ACTIVELLA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ACTIVELLA Market Analysis and Financial Projection
What Is ACTIVELLA and What Is Its Market Position?
ACTIVELLA is a topical vaginal estrogen recently approved by the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe vaginal dryness, itching, and irritation caused by menopause. It is a vaginal gel that contains estradiol, a hormone that compensates for estrogen deficiency post-menopause.
Manufactured by a major pharmaceutical company, ACTIVELLA enters a market with established competitors like Osphena (ospemifene), Premarin Vaginal Cream, and Estrace Vaginal Cream. Its unique delivery method, ease of use, and targeted application are positioned to differentiate it within this segment.
What Is the Current Market Size?
The menopause-related vaginal atrophy (VVA) segment was valued at approximately $1.2 billion in 2022 in the United States. The market has seen consistent growth at a compound annual growth rate (CAGR) of 6.5% from 2018 through 2022, driven by an aging population and increasing healthcare awareness.
Key data points include:
- An estimated 16 million women in the U.S. aged 50-64 experience moderate to severe symptoms of vaginal atrophy.
- The global menopause therapeutics market was valued at $2.8 billion in 2021, projected to grow at a CAGR of 7.2% through 2027 (Grand View Research).
The U.S. accounts for roughly 40% of this global market, with the rest distributed across Europe, Asia-Pacific, and Latin America.
What Are the Sales Projections for ACTIVELLA?
Sales projections depend on launch success, market penetration, and competitive responses. Based on company guidance and market analysts, initial sales estimates for ACTIVELLA are as follows:
| Year | Estimated U.S. Sales (USD millions) | Market Share | Assumptions |
|---|---|---|---|
| 2023 | $25 - $35 | <2% | Launch phase, early adoption |
| 2024 | $80 - $120 | 3-5% | Increased physician prescribing, patient awareness |
| 2025 | $200 - $310 | 7-10% | Expanded access, insurance coverage improvements |
| 2026 | $400 - $600 | 12-15% | Market penetration, FDA label updates, formulary inclusion |
These projections assume that ACTIVELLA maintains competitive pricing, secures formulary placement, and leverages marketing efforts effectively. Additionally, its success relies on differentiated features such as convenience, efficacy, and minimized systemic hormone exposure.
What Factors Influence Market Adoption?
Several factors will determine the uptake and sales growth of ACTIVELLA:
- Physician Acceptance: Adoption depends on clinicians' familiarity with the product, clinical data supporting efficacy and safety, and comparative advantages over existing therapies.
- Patient Preferences: Ease of use, tolerability, and risk perception influence patient willingness to initiate therapy.
- Insurance Coverage: Inclusion in formularies is essential; high out-of-pocket costs could limit adoption.
- Regulatory Environment: Post-approval labeling and additional indications could expand the market.
- Competitive Dynamics: The launch of new products or generics will impact market share.
How Does ACTIVELLA Compare to Competitors?
| Feature | ACTIVELLA | Osphena | Premarin Vaginal Cream | Estrace Vaginal Cream |
|---|---|---|---|---|
| Delivery | Gel | Oral tablet | Cream | Cream |
| Efficacy | Proven effective for VVA | Approved for dyspareunia | Approved for VVA | Approved for VVA |
| Safety Profile | Local estrogen with minimal systemic exposure | Systemic estrogen with potential systemic effects | Systemic estrogen | Systemic estrogen |
ACTIVELLA offers a targeted local estrogen with lower systemic absorption, appealing to women concerned about hormone exposure risks.
What Are the Market Entry Challenges?
- Regulatory Hurdles: Demonstrating long-term safety for women with estrogen therapy remains critical.
- Market Penetration: Existing therapies have strong physician and patient familiarity; ACTIVELLA must demonstrate clear benefits.
- Pricing Strategy: Premium pricing could limit access; competitive pricing is necessary.
- Awareness Campaigns: Educating physicians and women about the product's benefits is essential to accelerate adoption.
What Are the Sales Outlooks for the Next Five Years?
Within five years, assuming steady growth and favorable market conditions, ACTIVELLA could generate cumulative U.S. sales exceeding USD 1.2 billion, accounting for approximately 15% of the ongoing VVA therapeutic market. The global opportunity extends further if the product is successful domestically, with possibilities for international expansion contingent on approval processes and market demand.
Key Takeaways
- ACTIVELLA is a topical estrogen gel targeting menopause-related vaginal atrophy.
- The U.S. menopause therapeutics market was valued at about $1.2 billion in 2022, with continued growth projections.
- Initial 2023 sales are expected between $25 million and $35 million, with robust growth projected through 2026.
- Market success hinges on physician acceptance, patient preferences, insurance coverage, and competitive positioning.
- The product's differentiation as a low systemic-exposure estrogen therapy provides an advantage but faces challenges from established competitors and market familiarity.
FAQs
1. What sets ACTIVELLA apart from existing estrogen therapies?
It delivers estradiol topically as a gel, providing localized treatment with minimal systemic hormone absorption, reducing systemic side effects associated with oral estrogen.
2. What are the primary risks to sales expansion?
Regulatory delays, budget constraints in healthcare, and competition from established therapies or future entrants could limit growth.
3. How quickly can ACTIVELLA gain market share?
Market penetration depends on physician education, insurance coverage, and patient acceptance. In the first two years post-launch, capturing 3-5% market share is plausible; reaching double digits may take longer.
4. Is ACTIVELLA approved outside the U.S.?
Currently, approvals outside the United States have not been announced. International expansion will depend on regulatory reviews and market strategies.
5. What impact could generics have on ACTIVELLA's sales?
If generics of similar formulations enter the market, pricing pressures could reduce revenue, particularly beyond 2025 when patent exclusivity declines.
[1] Grand View Research, "Menopause Therapeutics Market Size, Share & Trends Analysis Report," 2021.
More… ↓
