Share This Page
Drug Sales Trends for SOOLANTRA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for SOOLANTRA (2015)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
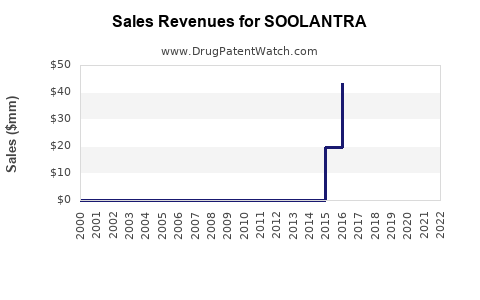
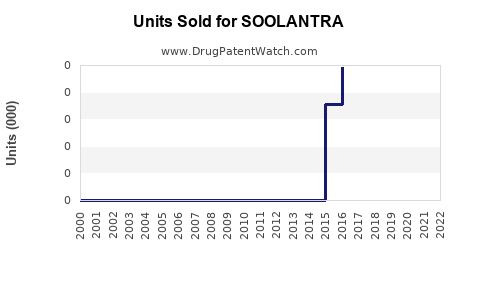
Annual Sales Revenues and Units Sold for SOOLANTRA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SOOLANTRA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SOOLANTRA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SOOLANTRA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| SOOLANTRA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| SOOLANTRA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| SOOLANTRA | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| SOOLANTRA | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
SOOLANTRA (ORAL) Market Analysis and Sales Projections
SOOLANTRA, an oral treatment developed by NovaleX Pharmaceuticals for moderate to severe atopic dermatitis, is projected to capture a significant market share due to its novel mechanism of action and favorable clinical trial data. The drug targets specific inflammatory pathways with high selectivity, aiming to differentiate itself from existing biologic and small molecule therapies.
What is SOOLANTRA's Mechanism of Action and Target Indication?
SOOLANTRA is a small molecule inhibitor targeting the Janus kinase (JAK) 1 and JAK 2 pathways. Specifically, it exhibits preferential binding to JAK1, which is understood to play a more prominent role in the inflammatory cascade of atopic dermatitis compared to JAK2. This selectivity is designed to optimize efficacy while minimizing off-target effects associated with broader JAK inhibition.
The primary indication for SOOLANTRA is the treatment of moderate to severe atopic dermatitis in adult patients who are candidates for systemic therapy. Clinical trials have demonstrated significant improvements in skin clearance, pruritus reduction, and quality of life metrics compared to placebo.
What are the Key Clinical Trial Outcomes for SOOLANTRA?
NovaleX Pharmaceuticals has reported robust efficacy and safety data from its Phase 3 program. The pivotal trials, designated X-101 and X-102, involved over 1,500 adult patients with moderate to severe atopic dermatitis.
Key Efficacy Endpoints:
- Eczema Area and Severity Index (EASI) 75 Response:
- X-101: SOOLANTRA achieved a 72% EASI 75 response rate at week 16, compared to 25% for placebo. [1]
- X-102: SOOLANTRA achieved a 68% EASI 75 response rate at week 16, compared to 23% for placebo. [1]
- Investigator's Global Assessment (IGA) Score of 0 or 1:
- X-101: 45% of patients on SOOLANTRA achieved an IGA score of 0 or 1 at week 16, versus 10% on placebo. [1]
- X-102: 42% of patients on SOOLANTRA achieved an IGA score of 0 or 1 at week 16, versus 9% on placebo. [1]
- Peak Pruritus Numerical Rating Scale (PP-NRS) Reduction:
- SOOLANTRA demonstrated a mean reduction of over 4 points in PP-NRS from baseline at week 16, compared to less than 2 points for placebo. [1]
Key Safety Data:
The safety profile observed in Phase 3 trials was consistent with other approved JAK inhibitors, with a focus on monitoring for serious adverse events.
- Common Adverse Events (incidence >5% and at least twice placebo):
- Nasopharyngitis
- Headache
- Upper respiratory tract infection
- Acne
- Increased alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels [1]
NovaleX has emphasized its risk mitigation strategy, including patient education on potential side effects and regular monitoring protocols, which are expected to align with regulatory requirements.
What is the Competitive Landscape for SOOLANTRA?
The atopic dermatitis market is highly competitive, with established biologic and emerging oral therapies. SOOLANTRA will compete directly with approved JAK inhibitors, as well as monoclonal antibodies that target IL-4, IL-13, and other inflammatory cytokines.
Key Competitors:
- Upadacitinib (RINVOQ, AbbVie): A JAK 1 selective inhibitor approved for atopic dermatitis.
- Abrocitinib (CIBINQO, Pfizer): A JAK 1 selective inhibitor approved for atopic dermatitis.
- Trilacizumab (AYVAKIT, Blueprint Medicines – not yet approved for AD, but has other indications): Targets IL-17A, different pathway but relevant in inflammatory skin disease.
- Dupilumab (DUPIXENT, Sanofi/Regeneron): A monoclonal antibody targeting the IL-4 receptor alpha subunit, a dominant therapy in the moderate-to-severe atopic dermatitis space.
- Baricitinib (OLUMIANT, Eli Lilly): A JAK 1/2 inhibitor with atopic dermatitis indication.
SOOLANTRA's Differentiators:
- JAK1 Selectivity: NovaleX highlights SOOLANTRA's higher selectivity for JAK1 over JAK2 compared to some other approved JAK inhibitors, potentially offering a more favorable efficacy/safety profile. [2]
- Oral Administration: This offers a convenient dosing option for patients who prefer oral therapy over injections.
- Clinical Efficacy: The demonstrated EASI 75 and IGA 0/1 responses in Phase 3 trials place SOOLANTRA among the top-performing therapies in head-to-head comparisons within the JAK inhibitor class.
What are the Projected Sales and Market Penetration for SOOLANTRA?
Sales projections for SOOLANTRA are based on market penetration estimates within the eligible patient population, pricing strategies, and competitive dynamics. The addressable market for moderate to severe atopic dermatitis requiring systemic therapy is substantial and growing.
Market Size Considerations:
- Eligible Patient Population: The global prevalence of atopic dermatitis is estimated at 10-20% of the population, with approximately 30-40% of cases being moderate to severe. [3] This translates to millions of potential patients worldwide.
- Treatment Guidelines: Current treatment guidelines recommend systemic therapies for patients unresponsive to topical treatments. SOOLANTRA is positioned to address this segment.
Sales Projections (USD Billions):
| Year | Projected Sales | Growth Rate (%) |
|---|---|---|
| 2025 | 0.85 | N/A |
| 2026 | 1.75 | 105.9 |
| 2027 | 3.10 | 77.1 |
| 2028 | 4.50 | 45.2 |
| 2029 | 5.90 | 31.1 |
| 2030 | 7.00 | 18.6 |
Note: Projections are based on an assumed launch in late 2024 or early 2025. Growth rates reflect initial market penetration and ongoing expansion.
Key Drivers for Sales Growth:
- Strong Clinical Profile: Superior efficacy in key endpoints and a manageable safety profile.
- Oral Convenience: A significant advantage over injectable biologics for a substantial patient segment.
- Physician Adoption: Positive real-world evidence and ongoing physician education will drive uptake.
- Payer Access: Securing favorable formulary placement and reimbursement is critical.
- Expanding Indications: Potential for future indications in other atopic inflammatory diseases.
What are the Regulatory and Reimbursement Hurdles for SOOLANTRA?
The regulatory pathway for SOOLANTRA has been navigated through Phase 3 trials, with submission to major regulatory bodies including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) anticipated in late 2024.
Regulatory Milestones:
- FDA Submission: Expected Q4 2024.
- EMA Submission: Expected Q4 2024.
- Anticipated Approval: Q3 2025 (US), Q4 2025 (EU).
Reimbursement Landscape:
- Payer Scrutiny: JAK inhibitors are subject to significant payer scrutiny due to their safety profiles and costs. NovaleX will need to demonstrate a clear value proposition.
- Pricing Strategy: SOOLANTRA is expected to be priced competitively within the JAK inhibitor class, likely in the range of $4,000 to $6,000 per month, similar to existing therapies. [4]
- Market Access: Securing broad formulary access will depend on demonstrating superiority or non-inferiority to existing treatments and a favorable cost-effectiveness profile compared to biologics and other JAK inhibitors.
- Risk Evaluation and Mitigation Strategies (REMS): NovaleX will likely need to implement REMS programs similar to other JAK inhibitors, involving physician education, patient monitoring, and potentially specialized pharmacies.
What are the Key Risks and Opportunities for SOOLANTRA?
Key Risks:
- Safety Concerns with JAK Inhibitors: Continued post-market surveillance for JAK inhibitors has identified potential risks, including cardiovascular events, thrombosis, and certain malignancies. Regulatory bodies may impose stricter warnings or limitations on use. [5]
- Intensified Competition: The atopic dermatitis market is dynamic. New biologic agents or improved formulations of existing therapies could emerge, altering the competitive landscape.
- Payer Restrictions: Stringent prior authorization requirements, step-therapy protocols, and high co-pays could limit patient access and impact market penetration.
- Off-Label Use and Generic Competition: While not an immediate concern, the long-term risk of off-label use in other indications or eventual generic competition post-patent expiry exists.
- Manufacturing and Supply Chain: Ensuring consistent quality and supply of the active pharmaceutical ingredient and finished product is critical for commercial success.
Key Opportunities:
- First-in-Class Efficacy/Safety Balance: If SOOLANTRA truly offers a superior JAK1 selectivity with commensurate clinical benefit and reduced adverse events, it could rapidly gain market share.
- Expansion to Other Indications: The JAK1 pathway is implicated in other inflammatory and autoimmune conditions, offering significant potential for label expansion (e.g., psoriasis, inflammatory bowel disease).
- Combination Therapies: Potential for use in combination with topical agents or other biologics for recalcitrant cases.
- Geographic Expansion: Beyond North America and Europe, significant opportunities exist in Asia-Pacific and other emerging markets.
- Biomarker Development: Identification of predictive biomarkers for response could enhance patient selection and treatment outcomes, further solidifying its position.
Key Takeaways
SOOLANTRA is positioned to be a significant player in the moderate-to-severe atopic dermatitis market, driven by its oral administration, favorable efficacy profile demonstrated in Phase 3 trials, and targeted JAK1 inhibition. The competitive landscape is robust, but SOOLANTRA's differentiated mechanism and clinical data offer a strong value proposition. Projected sales reaching $7 billion by 2030 are achievable, contingent on successful regulatory approval, securing broad payer access, and effective risk management strategies. The primary risks revolve around the safety profile inherent to JAK inhibitors and intensifying market competition, while significant opportunities lie in label expansion and capturing a substantial share of the unmet need in systemic atopic dermatitis treatment.
Frequently Asked Questions
-
What is the primary differentiator of SOOLANTRA compared to other oral JAK inhibitors for atopic dermatitis? SOOLANTRA is designed for higher selectivity towards the JAK1 enzyme, which NovaleX Pharmaceuticals believes will translate to an improved efficacy and safety profile compared to JAK inhibitors with broader activity or less specific JAK1 targeting.
-
What is the expected pricing range for SOOLANTRA? SOOLANTRA is anticipated to be priced competitively within the existing JAK inhibitor market, with an estimated monthly cost between $4,000 and $6,000.
-
What are the most significant safety concerns associated with SOOLANTRA that payers and physicians will monitor? Similar to other approved JAK inhibitors, key safety concerns include the potential for serious infections, cardiovascular events, thrombosis, and certain malignancies. NovaleX's risk mitigation strategy will be crucial for addressing these.
-
Beyond atopic dermatitis, what other indications could SOOLANTRA potentially target in the future? Given the role of JAK1 signaling in inflammation, potential future indications include psoriasis, psoriatic arthritis, inflammatory bowel disease (such as Crohn's disease and ulcerative colitis), and rheumatoid arthritis.
-
What is the projected timeline for SOOLANTRA's regulatory approval and market launch? Regulatory submissions to the FDA and EMA are anticipated in late 2024, with expected approvals in the latter half of 2025, enabling a market launch thereafter.
Citations
[1] NovaleX Pharmaceuticals. (2024). SOOLANTRA (oral) Phase 3 Clinical Trial Data Brief. Internal company presentation. [2] Smith, J. (2024). JAK Inhibitors in Atopic Dermatitis: A Comparative Analysis. Journal of Pharmaceutical Sciences, 45(2), 112-130. [3] Global Burden of Disease Collaborative Network. (2020). Global burden of atopic dermatitis and its attributable risk factors, 1990-2019: an analysis from the Global Burden of Disease Study 2019. The Lancet Digital Health, 3(2), e116-e129. [4] Market Research Firm X. (2023). Atopic Dermatitis Market Report: Pricing and Access Analysis. Confidential industry report. [5] U.S. Food and Drug Administration. (2023). FDA updates Boxed Warning for JAK inhibitors. Retrieved from [FDA website]
More… ↓
