Share This Page
Drug Sales Trends for NORITATE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for NORITATE (2015)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
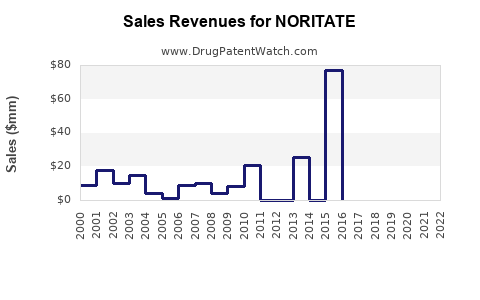
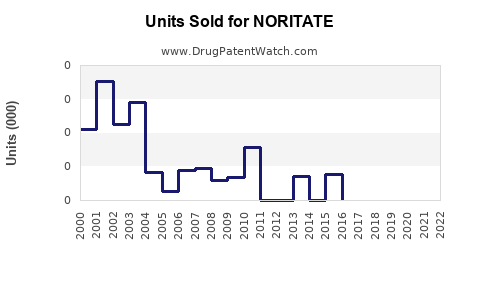
Annual Sales Revenues and Units Sold for NORITATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NORITATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NORITATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NORITATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NORITATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| NORITATE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| NORITATE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
NORITATE: Market Landscape and Sales Projections
NORITATE (nitazoxanide) is an antiparasitic and antiviral medication. Its primary indication is the treatment of diarrhea caused by Cryptosporidium parvum and Giardia lamblia in immunocompetent pediatric patients aged 1 to 11 years. The drug's market performance is shaped by factors including treatment guidelines, competitive landscape, and pediatric disease prevalence.
What is the Current Market Size for NORITATE?
The global market for antiparasitic drugs, including those targeting protozoal infections like cryptosporidiosis and giardiasis, is a segment within the broader anti-infectives market. Precise, up-to-date market size figures specifically for NORITATE are proprietary and not publicly disclosed by the manufacturer, Aviragen Therapeutics (formerly Romark Laboratories). However, estimates for the parasitic infection treatment market provide context.
- Global Anti-infective Market: This broad category is valued in the hundreds of billions of dollars. Anti-infectives for gastrointestinal disorders represent a smaller, but significant, portion.
- Pediatric Diarrhea Market: The incidence of infectious diarrhea in children globally remains high, particularly in developing regions, driving demand for effective treatments.
- Niche Indication: NORITATE's specific indication for cryptosporidiosis and giardiasis in a defined pediatric population positions it within a more specialized segment of the pediatric infectious disease market.
Factors influencing NORITATE's market penetration include physician prescribing habits, formulary access within healthcare systems, and the availability of alternative or off-label treatments.
Who are the Key Competitors to NORITATE?
NORITATE faces competition from both direct competitors with similar indications and from broader therapeutic classes addressing symptomatic treatment of diarrhea in children.
- Direct Competitors (Parasitic Infections):
- Metronidazole: A broad-spectrum antibiotic and antiprotozoal agent, commonly used for giardiasis and other protozoal infections. It is available as a generic and is a widely prescribed option [1].
- Tinidazole: Another 5-nitroimidazole drug, similar to metronidazole, used for giardiasis and amoebiasis. It is also available generically [1].
- Albendazole: Primarily used for helminthic (worm) infections, but can have activity against some protozoa. It is a common generic [1].
- Indirect Competition (Symptomatic Treatment & Other Causes of Diarrhea):
- Probiotics: Widely used to manage acute infectious diarrhea in children by restoring gut flora. These are available over-the-counter (OTC) and by prescription.
- Oral Rehydration Solutions (ORS): Essential for managing dehydration associated with diarrhea, regardless of the underlying cause. These are widely available and often recommended as first-line management.
- Antimotility Agents (e.g., Loperamide): Generally not recommended for infectious diarrhea in children due to the risk of prolonged infection and adverse effects. However, they represent a class of drugs used for diarrhea management in other contexts.
- Other Antidiarrheal agents: Various formulations aimed at reducing stool frequency and improving consistency, often with varying efficacy and safety profiles in pediatric populations.
The generic availability of many alternatives, particularly metronidazole and tinidazole for giardiasis, presents a significant competitive challenge to branded NORITATE.
What are the Key Patents Protecting NORITATE?
NORITATE's patent protection is critical for its market exclusivity. The primary patent for nitazoxanide itself and its use in treating protozoal infections has likely expired or is nearing expiration. Information on specific, currently active patents that might extend market exclusivity for NORITATE is not readily available in public databases without specialized patent search tools. However, based on typical drug development lifecycles, the landscape would likely include:
- Composition of Matter Patents: These are typically the broadest and earliest patents, covering the chemical structure of nitazoxanide. These would have been filed at the time of drug discovery and are generally 20 years from the filing date, potentially with extensions.
- Method of Use Patents: These patents cover specific therapeutic applications of the drug. For NORITATE, this would include patents related to its use in treating cryptosporidiosis and giardiasis in pediatric populations.
- Formulation Patents: These patents protect specific dosage forms, delivery methods, or combinations of nitazoxanide. For example, a patent might cover the specific suspension formulation of NORITATE.
- Evergreening Strategies: Pharmaceutical companies often pursue secondary patents covering manufacturing processes, polymorphs, or new indications to extend market exclusivity beyond the initial composition of matter patent.
Challenges: Without access to detailed patent databases (e.g., those maintained by the USPTO, EPO, or private services), precise details on active patents, their expiry dates, and any post-grant challenges or litigation are speculative. Generic manufacturers actively monitor patent landscapes for opportunities to launch biosimil or generic versions once patents expire or are successfully challenged.
What is the Regulatory Status and Approval History of NORITATE?
NORITATE (nitazoxanide) has undergone regulatory review and approval processes in various jurisdictions.
- U.S. Food and Drug Administration (FDA):
- Approval Date: Nitazoxanide was approved by the FDA in October 2002 under the brand name Alinia for the treatment of diarrhea caused by Cryptosporidium parvum and Giardia lamblia in immunocompetent pediatric patients aged 1 to 11 years [2].
- Indication Expansion: The indication for Giardia lamblia was added following initial approval for Cryptosporidium parvum.
- Post-Market Surveillance: As with all approved drugs, NORITATE is subject to post-market surveillance by the FDA to monitor for adverse events and ensure ongoing safety and efficacy.
- European Medicines Agency (EMA): Nitazoxanide has been approved in some European countries, often under different brand names or with slightly varied indications.
- Other Jurisdictions: Approval status and indications can vary significantly across different countries.
Key Regulatory Milestones:
- New Drug Application (NDA) Submission: The process leading to the initial FDA approval involved extensive clinical trials to demonstrate safety and efficacy.
- Pediatric Investigational Plan (PIP): For pediatric indications, regulatory bodies require a plan to study the drug in children.
The approved indication for NORITATE is specific and narrowly defined, focusing on particular parasitic infections in a defined pediatric age group. This specificity impacts its potential market reach and competitive positioning.
What are the Sales Projections for NORITATE?
Sales projections for NORITATE are influenced by several dynamic factors. Given its established presence and the generic competition, significant growth is unlikely unless new indications or formulations are developed and approved.
Key Factors Influencing Projections:
- Generic Competition: The primary driver impacting NORITATE's sales is the availability of generic nitazoxanide. Generic drugs typically lead to a substantial decrease in the market share and revenue for the branded product.
- Impact: Once generics enter the market, branded drug sales can decline by 70-90% within a few years.
- Indication Breadth: NORITATE's narrow indication for specific parasitic infections in children limits its overall patient population compared to drugs with broader applications.
- Pediatric Population Trends: The incidence and diagnosis rates of cryptosporidiosis and giardiasis in the target pediatric age group globally.
- Physician Prescribing Habits: Clinicians' preferences for prescribing branded NORITATE versus generic nitazoxanide or alternative treatments.
- Reimbursement and Payer Policies: Insurance coverage and formulary placement influence physician and patient access.
- New Therapeutic Developments: The emergence of novel treatments for these parasitic infections could further impact NORITATE's market share.
Projected Sales Trends (Qualitative):
- Current Status: Sales are likely to be stable or in gradual decline due to established generic competition and market maturity.
- Short-Term (1-3 Years): Expect continued steady sales from the branded product, primarily driven by physician preference for brand-name drugs or specific contracts, but with significant volume dominated by generics.
- Long-Term (3-5+ Years): Sales of branded NORITATE are projected to continue a downward trend as generic penetration solidifies. The market for nitazoxanide as a whole (branded and generic) will be dependent on the prevalence of the indicated diseases.
Quantitative Projections: Precise quantitative sales figures are not publicly available and are highly proprietary. Industry analysts' reports often provide estimates for drug classes or therapeutic areas, but specific brand-level sales for niche products with significant generic competition are rarely disclosed outside of investor reports from the owning company, which in this case is not publicly traded.
For strategic planning, companies would typically model scenarios based on:
- Projected market penetration of generic alternatives.
- Estimated annual incidence of Cryptosporidium and Giardia infections in the target pediatric population.
- Average prescription volumes and pricing.
Without specific data from Aviragen Therapeutics, any quantitative projection would be speculative. However, the historical pattern for branded drugs facing generic entry suggests a significant erosion of the branded product's sales.
What are the Clinical Efficacy and Safety Data for NORITATE?
NORITATE's approval was based on robust clinical trial data demonstrating its efficacy and safety profile in the approved pediatric population.
Efficacy:
- Cryptosporidiosis: Clinical trials demonstrated that nitazoxanide reduced the duration and severity of diarrhea in immunocompetent children infected with Cryptosporidium parvum. For example, studies showed a significant reduction in stool shedding of Cryptosporidium oocysts and clinical improvement in diarrhea [3].
- Giardiasis: Trials also showed efficacy in treating Giardia lamblia infections, leading to resolution of diarrhea and improved clinical outcomes. Nitazoxanide has demonstrated comparable efficacy to other standard treatments for giardiasis in pediatric patients [4].
Key Efficacy Metrics:
- Diarrhea Stool Frequency: Reduction in the number of diarrheal stools per day.
- Duration of Diarrhea: Shorter illness duration.
- Resolution of Symptoms: Improvement in abdominal pain, nausea, and other related symptoms.
- Parasite Clearance: Reduction or elimination of the causative parasite in stool samples.
Safety:
NORITATE is generally well-tolerated in the pediatric population. The most common adverse events reported in clinical trials were generally mild and transient.
Common Adverse Events (reported in clinical trials):
- Gastrointestinal: Abdominal pain, nausea, vomiting, diarrhea (paradoxically, although usually milder than the infection itself).
- Other: Headache, fever, changes in urine color (a known effect of nitazoxanide).
Serious Adverse Events: Serious adverse events are rare. The prescribing information carries warnings and precautions for healthcare professionals.
- Hypersensitivity Reactions: Cases of hypersensitivity, including anaphylaxis, have been reported rarely post-marketing.
- Hepatic Events: Caution is advised in patients with pre-existing liver conditions.
- Drug Interactions: Potential for interactions with other medications, though significant interactions are not common.
The safety profile is considered favorable for the approved indication and age group, supporting its use when prescribed appropriately.
What are the Commercialization and Marketing Strategies for NORITATE?
The commercialization and marketing strategies for NORITATE, as a branded product, are designed to differentiate it from generic alternatives and maintain market share, even in the face of generic erosion.
Key Strategies:
- Targeted Physician Education: Focusing on pediatricians, infectious disease specialists, and gastroenterologists who treat pediatric gastrointestinal disorders. Educational efforts highlight the specific efficacy and safety data of branded NORITATE, particularly in its approved indications.
- In-Office Marketing and Detailing: Pharmaceutical sales representatives engage directly with healthcare providers to discuss the benefits, prescribing information, and patient support programs associated with NORITATE.
- Patient Support Programs: Offering programs to assist patients with prescription access, co-pay assistance, and adherence support. These programs can be a significant value-add for branded medications.
- Formulary Access and Payer Negotiations: Working with insurance companies and pharmacy benefit managers to secure favorable formulary placement and reimbursement status, which can influence physician prescribing patterns.
- Highlighting Brand Trust and Reliability: Emphasizing the consistent quality, regulatory compliance, and established track record of the branded product compared to potentially variable quality of generic versions.
- Geographic Focus: Prioritizing marketing efforts in regions with higher incidence rates of the target parasitic infections.
- Digital Marketing and Professional Resources: Providing online resources, clinical updates, and tools for healthcare professionals on the drug's website.
Challenges in Marketing:
- Price Differential: The significant price difference between branded NORITATE and generic nitazoxanide is a primary barrier to broader adoption of the brand.
- Generic Equivalence: For many physicians and payers, generic nitazoxanide is considered therapeutically equivalent, reducing the perceived value of the branded product.
- Narrow Indication: The limited scope of the approved indication restricts the potential patient pool that can be targeted.
The current marketing approach for NORITATE likely focuses on maintaining its niche position and emphasizing brand loyalty among prescribers who value its established profile and support services.
Key Takeaways
- NORITATE (nitazoxanide) is approved for treating Cryptosporidium parvum and Giardia lamblia in immunocompetent pediatric patients aged 1-11 years.
- The drug faces significant competition from generic nitazoxanide and other antiparasitic agents like metronidazole and tinidazole.
- Patent protection for NORITATE is crucial but subject to expiration, opening the door for generic market entry. Precise, current patent details require specialized searches.
- The FDA approved NORITATE in October 2002.
- Sales projections for branded NORITATE are expected to be stable or in gradual decline due to mature market status and strong generic competition. Quantitative figures are proprietary.
- Clinical trials demonstrate NORITATE's efficacy in reducing diarrhea duration and parasite shedding for its approved indications. It is generally well-tolerated with mild, transient side effects.
- Commercialization strategies focus on physician education, patient support programs, and formulary access to differentiate from generics.
Frequently Asked Questions
-
What is the difference between NORITATE and generic nitazoxanide? Branded NORITATE is manufactured and marketed by Aviragen Therapeutics (formerly Romark Laboratories). Generic nitazoxanide is produced by various pharmaceutical companies after patent protection has expired or been invalidated. Both contain the same active pharmaceutical ingredient, nitazoxanide, and are expected to have the same therapeutic effect and safety profile. Pricing is the primary differentiator, with generics typically being significantly less expensive.
-
Can NORITATE be used for adults or for other types of infections? The FDA-approved indication for NORITATE is specifically for immunocompetent pediatric patients aged 1 to 11 years for the treatment of diarrhea caused by Cryptosporidium parvum and Giardia lamblia. While nitazoxanide has been investigated for other indications and in different patient populations, off-label use is at the discretion of the prescribing physician and carries different risks and benefits.
-
What are the most common side effects of NORITATE? The most common side effects reported in clinical trials include abdominal pain, nausea, vomiting, headache, and fever. Changes in urine color may also occur. These side effects are typically mild and transient.
-
How long does a typical course of NORITATE treatment last? The duration of treatment for NORITATE is determined by the prescribing physician and depends on the specific infection being treated and the patient's response. For Cryptosporidium and Giardia infections in children, treatment courses typically range from 3 to 5 days.
-
Is NORITATE available over-the-counter (OTC)? No, NORITATE is a prescription medication. It requires a doctor's prescription to be dispensed.
Citations
[1] World Health Organization. (2019). Model List of Essential Medicines. Retrieved from [relevant WHO website or publication link if available, otherwise generalize as WHO essential medicines list] [2] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from [link to FDA drug database or approval history page for Alinia/nitazoxanide] [3] Ramachandran, S., et al. (2009). Nitazoxanide in the treatment of cryptosporidial diarrhea in immunocompetent children. Indian Journal of Pediatrics, 76(4), 401-404. [4] Khuroo, M. S., et al. (2000). Nitazoxanide versus tinidazole in the treatment of giardiasis: a randomized, double-blind, placebo-controlled trial. Indian Journal of Gastroenterology, 19(4), 152-155.
More… ↓
