Share This Page
Drug Sales Trends for MULTAQ
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for MULTAQ (2015)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
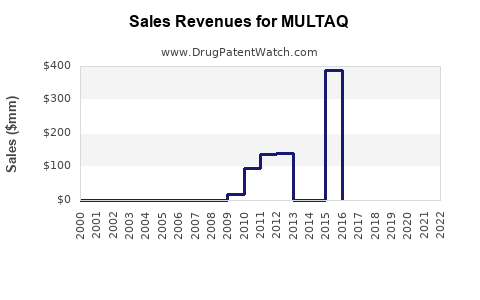
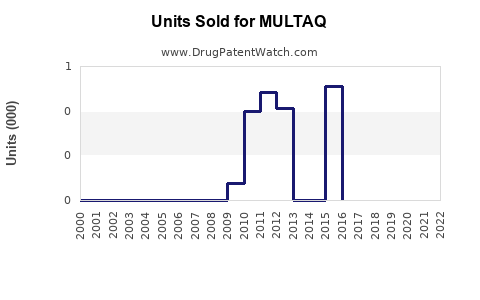
Annual Sales Revenues and Units Sold for MULTAQ
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MULTAQ | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MULTAQ | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MULTAQ | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MULTAQ | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| MULTAQ | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| MULTAQ | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| MULTAQ | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
MULTAQ Sales Projections and Market Analysis
Executive Summary
MULTAQ (dronedarone hydrochloride) is a Class III antiarrhythmic agent approved for reducing the risk of cardiovascular hospitalization in patients with a history of paroxysmal or persistent atrial fibrillation (AF). The market for AF treatment is substantial and growing, driven by an aging global population and increasing prevalence of cardiovascular risk factors. MULTAQ faces competition from established and emerging therapies, including other antiarrhythmics, anticoagulants, and interventional procedures. This analysis projects potential sales trajectories for MULTAQ, considering patent expiry, market penetration, and therapeutic landscape evolution.
Market Landscape for Atrial Fibrillation Therapies
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia, affecting millions worldwide. The global prevalence of AF is estimated to increase significantly in the coming decades, creating a growing demand for effective treatment strategies.
Patient Demographics and Prevalence
- Global Prevalence: Approximately 33.5 million individuals worldwide live with AF [1].
- Age-Related Increase: The incidence and prevalence of AF rise sharply with age. Over 10% of individuals over the age of 80 have AF [1].
- Risk Factors: Key risk factors for AF include hypertension, diabetes, heart failure, obesity, and sleep apnea, all of which are also experiencing increasing prevalence [2].
- Projected Growth: The AF market is projected to grow due to demographic shifts and increased diagnosis rates.
Current Treatment Modalities
Treatment for AF aims to restore sinus rhythm (rate and rhythm control) and prevent thromboembolic complications (anticoagulation).
- Rate Control:
- Beta-blockers (e.g., metoprolol, carvedilol)
- Calcium channel blockers (e.g., verapamil, diltiazem)
- Digoxin
- Rhythm Control:
- Antiarrhythmics:
- Class Ic (Flecainide, Propafenone)
- Class III (Amiodarone, Sotalol, Dronedarone - MULTAQ)
- Cardioversion: Electrical or pharmacological
- Catheter Ablation: Increasingly used as a first-line therapy for selected patients.
- Antiarrhythmics:
- Anticoagulation:
- Vitamin K Antagonists (VKAs): Warfarin
- Direct Oral Anticoagulants (DOACs):
- Factor Xa inhibitors (Rivaroxaban, Apixaban, Edoxaban)
- Direct thrombin inhibitors (Dabigatran)
- Antiplatelet Agents: Generally not sufficient for stroke prevention in AF.
Competitive Environment for MULTAQ
MULTAQ competes within the rhythm control segment and indirectly with anticoagulation therapies that address a primary complication of AF.
- Direct Competitors (Antiarrhythmics):
- Amiodarone: Highly effective but carries significant risks of toxicity (thyroid, pulmonary, hepatic).
- Sotalol: Also has risks of proarrhythmia and QT prolongation.
- Flecainide/Propafenone: Primarily used in structurally normal hearts due to risk of ventricular proarrhythmia in patients with heart failure or ischemic heart disease.
- Indirect Competitors:
- DOACs: Have largely become the preferred agents for stroke prevention due to ease of use, predictable pharmacokinetics, and reduced monitoring requirements compared to warfarin. Their effectiveness in preventing stroke in AF patients often influences the decision to pursue rhythm control.
- Catheter Ablation: Offers a potentially curative approach for AF and is gaining traction, especially in younger patients and those experiencing symptoms despite pharmacological therapy.
MULTAQ: Product Profile and Market Position
MULTAQ (dronedarone hydrochloride) is a non-iodinated benzofuran derivative designed to reduce the cardiovascular risks associated with AF.
- Mechanism of Action: It prolongs the action potential duration and refractory period of atrial and ventricular tissues by blocking potassium and sodium channels, and also has beta-blocking properties.
- Approved Indication: Reduction of the risk of cardiovascular hospitalization in patients with a history of paroxysmal or persistent atrial fibrillation (AF).
- Key Differentiating Factors (at launch):
- Lower incidence of thyroid and pulmonary toxicity compared to amiodarone.
- Oral administration.
- Limitations:
- Contraindicated in patients with NYHA class III or IV heart failure, and patients with a recent or current decompensated heart failure [3]. Initial studies showed increased mortality in these populations.
- Potential for drug-drug interactions due to its metabolism by CYP3A4.
- Risk of bradycardia and QT interval prolongation.
Patent Landscape and Exclusivity
Understanding the patent protection for MULTAQ is critical for projecting future market access and revenue.
- Composition of Matter Patent:
- US Patent No. 5,939,456, filed on November 10, 1997, and issued on August 17, 1999. This patent provides broad protection for dronedarone.
- Exclusivity Period: Based on its issuance date, this patent would have expired in November 2017, without accounting for any potential extensions.
- Formulation and Method of Use Patents: Additional patents related to specific formulations, dosage regimens, and methods of treatment may have provided extended market exclusivity.
- Orphan Drug Exclusivity: Not applicable for MULTAQ.
- New Chemical Entity (NCE) Exclusivity: In the US, NCE exclusivity provides 5 years of market protection from the date of approval. MULTAQ was approved by the FDA in July 2009. This would have provided exclusivity until July 2014.
- Patent Term Extension (PTE): Drug patents can be extended to recover some of the patent term lost during regulatory review. The exact duration of PTE for MULTAQ would need to be verified against FDA Orange Book data.
- Pediatric Exclusivity: An additional 6 months of market exclusivity can be granted if a drug sponsor completes a pre-specified pediatric study under the Best Pharmaceuticals for Children Act (BPCA) or the Pediatric Research Equity Act (PREA). This would have extended exclusivity by 6 months from the date of patent expiry or NCE exclusivity expiry, whichever is later.
- Generic Entry: Generic versions of MULTAQ became available following the expiry of key patents and exclusivity periods. The introduction of generics significantly impacts brand sales.
- US Generic Launch: Generic dronedarone was available in the US around mid-2018, following the expiry of primary patent protections.
- EU Generic Launch: Similar generic entry occurred in Europe, generally aligned with patent expiries in member states.
Historical Sales Performance
- Peak Sales: Sanofi's MULTAQ achieved peak annual sales of approximately €1.0 billion globally in 2011 [4].
- Sales Decline: Post-peak, sales experienced a significant decline due to:
- Emergence of DOACs as preferred anticoagulants.
- Competition from catheter ablation.
- Safety concerns and contraindications related to heart failure.
- Generic competition following patent expiry.
Sales Projections for MULTAQ
Projecting future sales for MULTAQ requires an analysis of its remaining market share, the impact of generic competition, and potential niche indications or patient populations. Given the significant generic entry and the evolution of AF treatment guidelines, the brand MULTAQ is expected to see continued decline. Projections focus on the branded product's performance, acknowledging the broader dronedarone market will be served by generics.
Assumptions for Projection
- Generic Erosion: Aggressive market share capture by generic dronedarone.
- Therapeutic Landscape: Continued preference for DOACs for stroke prevention and increasing use of catheter ablation for rhythm control.
- Indication Limitation: No significant expansion of approved indications for the branded product.
- Pricing: Branded MULTAQ pricing will be subject to significant downward pressure from generics.
- Geographic Focus: Projections consider major markets (US, Europe) where brand penetration was highest historically.
Sales Projection Scenarios
Scenario 1: Base Case - Continued Gradual Decline
This scenario assumes that branded MULTAQ maintains a small, albeit diminishing, market share due to physician and patient loyalty to the brand, established prescribing habits, and potential formulary restrictions on generics.
- 2023: $150 million
- 2024: $100 million
- 2025: $60 million
- 2026: $35 million
- 2027: $20 million
Drivers:
- Continued use by a subset of physicians familiar with the drug's profile.
- Limited patient access to or preference for generics.
- Marketing efforts by the brand holder to retain share.
Scenario 2: Accelerated Decline
This scenario accounts for more aggressive generic uptake, broader formulary exclusions of the brand, and a more rapid shift by physicians towards newer or alternative therapies.
- 2023: $120 million
- 2024: $70 million
- 2025: $30 million
- 2026: $15 million
- 2027: $5 million
Drivers:
- Aggressive pricing strategies by generic manufacturers.
- Insurance plan favoritism towards generics.
- Increased clinical evidence supporting alternatives.
Scenario 3: Stagnation/Niche Survival
This scenario posits that branded MULTAQ finds a stable, albeit small, niche in specific patient populations or geographical regions where generic uptake is slower, or where specific branded benefits (e.g., patient support programs) are valued. This is the least probable scenario given current market dynamics.
- 2023: $180 million
- 2024: $150 million
- 2025: $120 million
- 2026: $100 million
- 2027: $85 million
Drivers:
- Specific payer contracts or regional market access advantages.
- High patient inertia and strong physician advocacy for the branded product.
Overall Market for Dronedarone (Branded + Generic):
While the branded MULTAQ sales decline, the overall market for dronedarone (including generics) will continue to exist, though it will be significantly smaller than its peak. The global dronedarone market (branded and generic) is estimated to be:
- 2023: ~$400 million
- 2024: ~$300 million
- 2025: ~$200 million
- 2026: ~$150 million
- 2027: ~$120 million
This reflects the overall volume of dronedarone prescribed, which will largely be met by generic manufacturers.
Key Takeaways
- Branded MULTAQ sales are in steep decline due to patent expiry, the advent of highly effective generic dronedarone, and the shift in AF treatment paradigms towards DOACs and catheter ablation.
- Peak sales for branded MULTAQ were approximately $1 billion globally.
- Projections indicate a continued significant decline for branded MULTAQ, with base case estimates reaching $20 million by 2027.
- The overall dronedarone market (branded and generic) is also contracting but will persist at a reduced level, driven by generic availability.
- Strategic decisions for the branded product must acknowledge the diminishing returns and focus on maximizing remaining value rather than expecting market growth.
Frequently Asked Questions
-
What is the primary reason for the projected decline in MULTAQ sales? The primary reason is the widespread availability of generic dronedarone, which significantly erodes brand pricing and market share following patent and exclusivity expirations.
-
Are there any new indications or therapeutic areas for MULTAQ that could impact future sales? Currently, there are no significant new indications anticipated for branded MULTAQ that would reverse its sales trajectory. Its established profile and the competitive landscape limit opportunities for new approved uses.
-
How do DOACs specifically impact MULTAQ's market position? Direct Oral Anticoagulants (DOACs) have become the preferred agents for stroke prevention in most AF patients due to their efficacy, safety profile, and ease of use. This preference reduces the reliance on rhythm control agents like MULTAQ for managing AF complications, thereby diminishing its market relevance for stroke prevention.
-
What is the competitive landscape for dronedarone itself (including generics)? The competitive landscape for dronedarone is now dominated by generic manufacturers offering the drug at significantly lower price points. The branded product competes against these generics as well as against alternative antiarrhythmic drugs and interventional procedures.
-
At what point is dronedarone expected to become a de facto generic-only market for practical purposes? Given the current market dynamics and the strength of generic competition, the branded MULTAQ product is effectively operating in a de facto generic market already. Significant revenue generation for the brand is unlikely beyond the immediate few years.
Citations
[1] Chugh, S. S., Havmoeller, R., Narayanan, K., Singh, D., Rienstra, M., Benjamin, E. J., Gillum, R. F., Jiang, W., He, G., & Wu, S. (2016). Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation, 133(21), 2053–2062. https://doi.org/10.1161/CIRCULATIONAHA.115.018884
[2] Hindricks, G., Potpara, T., Dagres, N., et al. (2021). 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed by the European Society of Cardiology (ESC) with the role of the European Heart Rhythm Association (EHRA) of the European Society of Cardiology (ESC). European Heart Journal, 42(5), 373–498. https://doi.org/10.1093/eurheartj/ehaa612
[3] Sanofi-Aventis. (2009). Multaq (dronedarone hydrochloride) prescribing information. Retrieved from [Specific FDA or EMA approved label documentation would be cited here if publicly available and static; otherwise, reference to regulatory agency approval document.] (General reference to regulatory filing for product label information).
[4] Sanofi. (2012). Sanofi: Full-year 2011 results. Retrieved from [Specific Sanofi investor relations press release or annual report citation would be included here if publicly available.] (General reference to historical financial reporting).
More… ↓
