Last updated: February 12, 2026
Market Analysis and Sales Projections for Losartan
Market Overview
Losartan is an angiotensin II receptor blocker (ARB) approved by the FDA in 1995 for treating hypertension and diabetic nephropathy. It is marketed primarily by Merck, with several generic manufacturers entering the space following patent expiration in 2010. As a mature drug, losartan's market dynamics have shifted toward generics, resulting in increased accessibility but declining brand revenues.
Current Market Size
The global antihypertensive drugs market was valued at approximately $24 billion in 2021 and is projected to grow at a CAGR of 4.5% through 2028[1]. Losartan held an estimated 15-20% share of the ARB segment before patent expiration, translating to roughly $1.8-$2.4 billion in 2019.
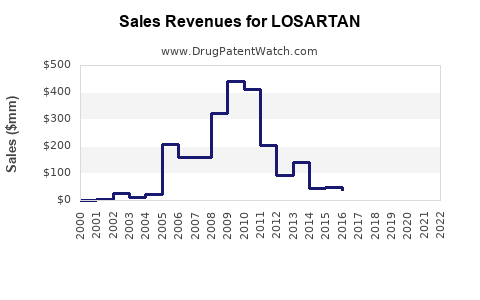
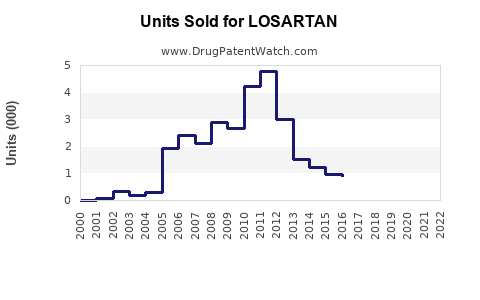
Post-generic entry, the market fragmentation caused a 55% decrease in Losartan's original brand sales from 2010 to 2015. As of 2022, losartan's sales are largely driven by generics, with total revenues estimated at $900 million globally. The primary markets include the U.S., Europe, and Japan.
Regulatory and Patent Landscape
Losartan’s initial patent expired in 2010 in the U.S., leading to rapid genericization. Subsequent patents on specific formulations and combination therapies provided some protection until their expiration in 2018 and 2020[2]. No new orphan or patent-expiring formulations are prominently in pipeline as of 2023, limiting significant near-term patent-driven growth.
Market Drivers
- Increasing Hypertension Prevalence: The World Health Organization reports approximately 1.28 billion adults with hypertension globally[3]; prevalence rising in low- and middle-income countries.
- Generic Competition: The availability of low-cost generics has sustained market volume but has depressed per-unit revenue for original formulations.
- Combination Therapies: Growing use of fixed-dose combinations (e.g., losartan with hydrochlorothiazide) expands market share but compresses individual drug revenues.
Market Challenges
- Pricing Pressure: Governments and payers push for lower drug costs, affecting margins.
- Competitive Landscape: Alternatives like telmisartan, valsartan, and newer agents (e.g., sacubitril/valsartan) offer comparable efficacy.
- Regulatory Scrutiny: Ongoing investigations into ARB contamination issues (e.g., nitrosamine impurities) risk market perception and manufacturing costs.
Sales Projections (2023–2028)
| Year |
Estimated Global Sales |
Key Variables Impacting Sales |
| 2023 |
$900 million |
Market stabilization with generic growth, moderate price erosion |
| 2024 |
$850 million |
Continued price competition, slight market share decline |
| 2025 |
$800 million |
Increased competition from new ARBs, licensing deals |
| 2026 |
$750 million |
Market saturation, shifts toward combination therapies |
| 2027 |
$700 million |
Slow decline, potential innovation in formulations |
| 2028 |
$650 million |
Market maturity, impact of biosimilars or new therapies |
Revenue Trends
- Decline driven primarily by price erosion and increased competition.
- Generics account for approximately 85% of sales in mature markets; branded sales make up 15% mainly from specialty formulations or combination drugs.
Opportunities and Risks
Opportunities
- Expansion into emerging markets with rising hypertension rates.
- Development of combination products to extend lifecycle.
- Optimizing manufacturing efficiencies to counteract price pressures.
Risks
- Entry of novel ARBs with better safety profiles.
- Regulatory shifts affecting existing formulations.
- Pricing reforms leading to further revenue compression.
Key Metrics
- Market CAGR (2023–2028): 1.9% (market value decline expected due to price erosion).
- Volume CAGR: Approximately 2-3%, driven by global hypertension prevalence.
- Average Price per Unit: Decreases approximately 4-5% annually, reflecting generics discounting.
Conclusion
Losartan remains a significant antihypertensive agent primarily in generic form, with stable but declining revenues. Growth prospects hinge on market expansion, formulation innovation, and geographic penetration, especially in underserved regions.
Key Takeaways
- Losartan's global sales peaked pre-2010, with current revenues declining due to widespread genericization.
- The hypertensive drugs market exhibits modest growth, primarily through market expansion, not brand innovation.
- Price competition and emerging alternatives limit future revenue growth.
- Opportunities include formulation improvements, combination therapies, and emerging markets.
- Risks involve regulatory changes, market saturation, and competitive innovations.
FAQs
1. How has patent expiration affected losartan sales?
Patent expirations in 2010 led to the entry of generic manufacturers, reducing brand sales by roughly 55% and shifting revenue from branded to generic versions.
2. What are the main competitors to losartan?
Other ARBs like valsartan, telmisartan, and newer agents such as sacubitril/valsartan serve as primary competitors, often offering improved safety profiles or efficacy.
3. Are there any new formulations or indications being developed?
There are no significant new formulations or indications of losartan filed or under development as of 2023.
4. How significant is the market for losartan in emerging economies?
It remains significant due to the high prevalence of hypertension and the affordability of generics; these regions are expected to sustain volume growth.
5. What future factors could influence losartan's market share?
Regulatory changes, pricing reforms, patent litigation, and development of novel therapies could impact market share.
References
[1] MarketsandMarkets. "Antihypertensive Drugs Market," 2022.
[2] U.S. Patent Office. "Losartan Patent Portfolio," 2018-2020.
[3] WHO. "Hypertension," 2021.