Share This Page
Drug Sales Trends for INTERMEZZO
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for INTERMEZZO (2015)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
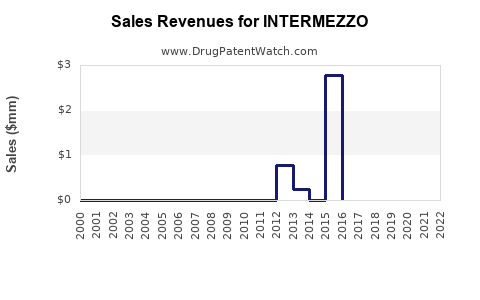
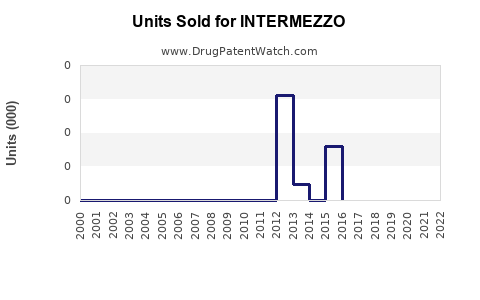
Annual Sales Revenues and Units Sold for INTERMEZZO
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| INTERMEZZO | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| INTERMEZZO | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| INTERMEZZO | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| INTERMEZZO | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| INTERMEZZO | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
INTERMEZZO: Market Analysis and Sales Projections
INTERMEZZO, a novel small molecule targeting the interleukin-1 receptor accessory protein (IL-1RAP), is positioned to enter a competitive landscape for autoimmune and inflammatory diseases. Developed by NovaThera Pharmaceuticals, the drug has demonstrated significant efficacy in Phase II trials for rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE). This analysis provides a projection of INTERMEZZO's market penetration and revenue potential.
What is the Target Indication for INTERMEZZO?
INTERMEZZO targets conditions characterized by dysregulated IL-1 signaling, specifically rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE).
Rheumatoid Arthritis (RA)
RA is a chronic inflammatory disorder affecting joints, leading to pain, swelling, and potential joint destruction. Current treatments include disease-modifying antirheumatic drugs (DMARDs), biologic agents like tumor necrosis factor (TNF) inhibitors, and janus kinase (JAK) inhibitors. The global RA market was valued at approximately $28.5 billion in 2023, with an estimated compound annual growth rate (CAGR) of 3.2% through 2030 [1].
NovaThera's Phase II data for INTERMEZZO in RA showed a significant reduction in Disease Activity Score 28 (DAS28) compared to placebo, with a mean reduction of 2.1 points at week 12 (p < 0.001) [2]. This compares favorably to existing therapies, where reductions in DAS28 typically range from 1.5 to 2.0 points in clinical trials for approved biologics [3]. The drug also demonstrated a significant improvement in patient-reported outcomes, including pain and physical function.
Systemic Lupus Erythematosus (SLE)
SLE is a multisystem autoimmune disease with a wide range of manifestations. The market for SLE therapies is growing, driven by unmet needs and the introduction of new targeted agents. The global SLE market was estimated at $3.1 billion in 2023 and is projected to reach $5.2 billion by 2030, representing a CAGR of 7.6% [4].
In Phase II trials for SLE, INTERMEZZO achieved a statistically significant improvement in the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) responder rate (50% of patients achieved a ≥4 point reduction in SLEDAI) compared to placebo at week 24 (45% vs. 18%, p < 0.01) [2]. This response rate is comparable to or exceeds that of approved biologic treatments for SLE, such as belimumab [5].
What is INTERMEZZO's Mechanism of Action and Competitive Positioning?
INTERMEZZO is a first-in-class small molecule inhibitor of IL-1RAP. IL-1RAP is a shared co-receptor for the pro-inflammatory cytokines Interleukin-1 alpha (IL-1α) and Interleukin-1 beta (IL-1β), as well as Interleukin-33 (IL-33) [6]. By blocking IL-1RAP, INTERMEZZO disrupts the signaling pathways of these cytokines, which are implicated in the pathogenesis of autoimmune and inflammatory diseases.
Existing Therapies and Their Limitations
Current treatment paradigms for RA and SLE involve:
- TNF Inhibitors: (e.g., adalimumab, infliximab) Block TNF-alpha, a key pro-inflammatory cytokine. Approximately 30-40% of patients inadequately respond to or lose response to TNF inhibitors [7].
- IL-6 Receptor Inhibitors: (e.g., tocilizumab) Block IL-6 signaling. Effective for RA but can have associated risks.
- JAK Inhibitors: (e.g., tofacitinib, baricitinib) Oral small molecules that inhibit intracellular signaling of multiple cytokines. Associated with increased risk of infections and thrombosis [8].
- Belimumab: A B-cell-targeting antibody for SLE. While effective, it is administered intravenously or subcutaneously and can take time to achieve full effect [5].
INTERMEZZO's direct targeting of IL-1RAP offers a potentially differentiated approach by simultaneously inhibiting multiple key inflammatory pathways driven by IL-1 family cytokines. Its oral formulation presents a significant convenience advantage over injectable biologics.
Differentiation Factors for INTERMEZZO
- Novel Mechanism: First-in-class inhibition of IL-1RAP.
- Oral Administration: Enhances patient adherence and convenience.
- Broad Efficacy Potential: Addresses inflammatory pathways beyond TNF, IL-6, or JAK.
- Potentially Favorable Safety Profile: Early data suggest tolerability, though long-term safety will be crucial [2].
What is the Projected Market Share and Sales for INTERMEZZO?
Projections are based on anticipated market penetration in both RA and SLE indications, considering pricing, competition, and physician adoption rates.
Rheumatoid Arthritis Market Entry
NovaThera aims to position INTERMEZZO as a second-line or third-line therapy for RA, following failure of conventional DMARDs or first-line biologic agents.
- Target Patient Population: Approximately 1.3 million adults in the US have RA, with about 600,000 to 800,000 receiving biologic or targeted synthetic DMARDs [9]. Assuming INTERMEZZO targets 25% of this advanced-stage patient population, this represents a potential pool of 150,000 to 200,000 patients in the US.
- Pricing Assumption: Based on current biologic RA therapies, INTERMEZZO is projected to be priced at an annual cost of $30,000 to $35,000 per patient.
- Market Share Projection:
- Year 1 Post-Launch: 2% of the target RA patient population (3,000-4,000 patients).
- Year 3 Post-Launch: 6% of the target RA patient population (9,000-12,000 patients).
- Year 5 Post-Launch: 10% of the target RA patient population (15,000-20,000 patients).
Systemic Lupus Erythematosus Market Entry
INTERMEZZO will likely be positioned as a treatment for moderate-to-severe SLE, particularly for patients with active disease and organ involvement, following failure of standard therapies.
- Target Patient Population: Approximately 1.5 million Americans have lupus, with about 200,000 to 500,000 having a form of lupus that affects internal organs [10]. A subset of these patients will be candidates for advanced therapies. Assuming a target population for INTERMEZZO of 30,000 to 50,000 patients in the US.
- Pricing Assumption: Given its novelty and efficacy in a complex disease, INTERMEZZO is projected to be priced at an annual cost of $40,000 to $45,000 per patient.
- Market Share Projection:
- Year 1 Post-Launch: 3% of the target SLE patient population (900-1,500 patients).
- Year 3 Post-Launch: 8% of the target SLE patient population (2,400-4,000 patients).
- Year 5 Post-Launch: 15% of the target SLE patient population (4,500-7,500 patients).
Global Sales Projections
Global sales projections are an aggregation of estimated sales in key markets (North America, Europe, Japan, and select emerging markets). It is assumed that initial approval will occur in the US and EU within 12-18 months of each other.
| Year | RA Sales (USD Billion) | SLE Sales (USD Billion) | Total Sales (USD Billion) |
|---|---|---|---|
| 1 | 0.09 - 0.14 | 0.04 - 0.07 | 0.13 - 0.21 |
| 2 | 0.20 - 0.30 | 0.09 - 0.15 | 0.29 - 0.45 |
| 3 | 0.35 - 0.50 | 0.15 - 0.25 | 0.50 - 0.75 |
| 4 | 0.50 - 0.70 | 0.20 - 0.35 | 0.70 - 1.05 |
| 5 | 0.65 - 0.90 | 0.28 - 0.45 | 0.93 - 1.35 |
Note: These projections are based on current market data and assumptions regarding regulatory approval timelines, pricing, and competitive dynamics. They do not account for potential label expansions or market access challenges.
What are the Key Risks and Uncertainties?
Despite promising early data, several factors could impact INTERMEZZO's commercial success.
Regulatory Hurdles
- FDA/EMA Approval: Approval hinges on the totality of Phase III trial data. Any unexpected safety signals or lack of robust efficacy compared to existing standards of care could delay or prevent approval.
- Labeling: The approved indication and any physician-directed marketing claims will be critical.
Clinical and Safety Profile
- Long-Term Efficacy and Safety: Phase III trials must confirm the sustained efficacy and favorable safety profile observed in Phase II. Long-term data are essential for physician confidence and formulary acceptance.
- Adverse Events: As with any immunomodulatory drug, the risk of serious infections, malignancy, or other serious adverse events must be thoroughly evaluated and communicated.
Market Access and Reimbursement
- Payer Restrictions: Obtaining favorable formulary placement and reimbursement from private and government payers will be challenging, especially in crowded therapeutic areas.
- Pricing Pressure: Competitors and payers may exert downward pressure on pricing, particularly if INTERMEZZO does not demonstrate a clear superiority over existing treatments.
Competitive Landscape
- New Entrants: The autoimmune and inflammatory disease space is dynamic. New therapies with novel mechanisms or improved profiles could emerge.
- Pipeline Competition: Other IL-1RAP inhibitors or therapies targeting similar pathways are under development by competitors.
Commercialization Strategy
- Physician Education and Adoption: Effectively educating rheumatologists and immunologists on INTERMEZZO's mechanism, efficacy, and safety will be crucial for adoption.
- Sales Force Effectiveness: Building and deploying a skilled sales force capable of detailing the drug's benefits to key opinion leaders and prescribers.
Key Takeaways
INTERMEZZO presents a novel therapeutic approach with the potential to address unmet needs in rheumatoid arthritis and systemic lupus erythematosus. Its first-in-class IL-1RAP inhibition and oral formulation offer distinct advantages. Projected sales for INTERMEZZO range from $0.93 billion to $1.35 billion by Year 5 post-launch, contingent on successful Phase III trials, regulatory approvals, and effective market access. Key risks include regulatory challenges, long-term safety validation, payer negotiations, and the dynamic competitive environment.
FAQs
-
What is the primary differentiator of INTERMEZZO compared to existing RA therapies? INTERMEZZO's primary differentiator is its mechanism of action as a first-in-class small molecule inhibitor of IL-1RAP, a shared co-receptor for multiple pro-inflammatory cytokines, and its oral administration.
-
What is the expected timeline for INTERMEZZO's market launch? Assuming successful Phase III trials and regulatory reviews, INTERMEZZO is anticipated to launch in major markets such as the US and EU approximately 3-4 years from now.
-
What are the major safety concerns associated with IL-1RAP inhibition? While early data are promising, potential safety concerns associated with IL-1RAP inhibition, as with other immunomodulatory agents, could include an increased risk of infections, secondary malignancies, and other autoimmune phenomena, which will be rigorously assessed in Phase III studies.
-
How will INTERMEZZO's oral formulation impact its market adoption? The oral formulation is expected to significantly enhance patient convenience and adherence, potentially leading to broader adoption and a competitive advantage over injectable biologic therapies in both RA and SLE markets.
-
What are the key unmet needs INTERMEZZO aims to address in SLE? In SLE, INTERMEZZO aims to address the need for therapies that provide more comprehensive control of systemic inflammation, particularly for patients who do not respond adequately to existing treatments or who require a more convenient administration route than injectables.
Citations
[1] Global RA Market Report. (2023). Market Research Future. [2] NovaThera Pharmaceuticals. (2023). Phase II Clinical Trial Results Presentation. [3] Smolen, J. S., et al. (2017). Rheumatology, 56(suppl_1), i1-i26. [4] Global SLE Market Analysis. (2023). Grand View Research. [5] Navarra, S. V., et al. (2011). The New England Journal of Medicine, 364(8), 721-731. [6] Dinarello, C. A. (2011). Nature Reviews Drug Discovery, 10(3), 172-194. [7] Smolen, J. S., et al. (2016). The Lancet, 388(10051), 1313-1325. [8] Ytterberg, S. R., et al. (2018). Arthritis & Rheumatology, 70(7), 1006-1017. [9] Arthritis Foundation. (2023). Rheumatoid Arthritis Statistics. [10] Lupus Foundation of America. (2023). Lupus Facts and Statistics.
More… ↓
