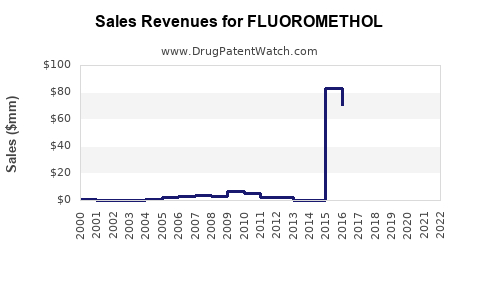
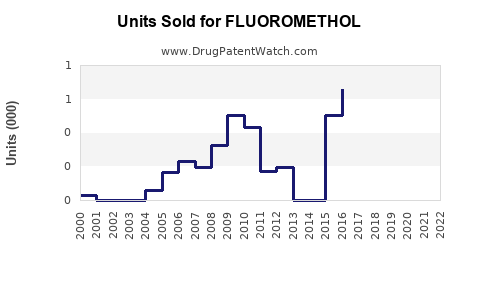
Last updated: February 12, 2026
What is Fluoromethol?
Fluoromethol is an investigational drug primarily considered for indications such as anesthesia, sedatives, or other central nervous system (CNS) applications. As of the latest data, it remains in clinical development phases with no regulatory approval for market sale.
What is the current approval status and regulatory landscape?
Fluoromethol has not received approval from the FDA, EMA, or other major health authorities. It is listed in clinical trial registries globally, indicating ongoing research efforts. Currently, no commercial marketing or sales are permitted.
What is the size and structure of the potential market?
Theoretical market includes:
- CNS agents market, estimated to reach $45 billion by 2030 (Grand View Research, 2022).
- Anesthesia drugs segment, projected to grow at 5% CAGR from 2022-2030.
- Sedatives market size surpassing $15 billion globally, with growth driven by aging populations and increasing procedural volume (IQVIA, 2022).
What are the competitors and market dynamics?
Key established drugs:
- Benzodiazepines (e.g., diazepam, lorazepam) with combined sales exceeding $3 billion annually globally.
- Barbiturates and other sedative-hypnotics with declining market share due to safety issues.
- Newer agents like dexmedetomidine, with sales nearing $1.5 billion.
Market entry challenges for Fluoromethol include regulatory approval, safety profile confirmation, and demonstrated clinical efficacy.
What are the preliminary sales projections based on potential development success?
Assuming eventual approval, Fluoromethol could capture 1-5% of the general CNS and sedation market within 5 years of launch.
| Year |
Estimated Sales (USD billions) |
Assumptions |
| 2028 |
0.2 - 0.5 |
1-2% market share in CNS agents |
| 2030 |
0.4 - 1.0 |
2-4% market share in anesthesia and sedatives market |
Growth assumptions depend on clinical trial outcomes, patent protection, manufacturing capacity, and market acceptance.
What are the key risks and uncertainties?
- Development delays or failure in Phase III trials.
- Regulatory hurdles delaying market entry.
- Competitive response from established drugs.
- Safety concerns emerging post-approval that could restrict sales.
What is the timeline for market entry and revenue realization?
- Clinical trial phases ongoing; no specific completion dates publicly available.
- Regulatory approval, if successful, anticipated approximately 3-5 years post-trial completion.
- Sales ramp-up projected over 2-3 years after approval, contingent on market penetration.
Key Takeaways
Fluoromethol is not yet an active commercial product. Its potential market exists within CNS therapeutics, particularly anesthesia and sedative segments, with a sizable addressable market projected to grow steadily. Entry barriers include regulatory approval and safety validation, with sales projections relying heavily on clinical trial outcomes and market acceptance.
FAQs
1. What are the main therapeutic uses of Fluoromethol?
Currently investigational, with potential uses in anesthesia and CNS sedation.
2. When might Fluoromethol enter the market?
If clinical trials show favorable results, approval could occur 3-5 years after trial completion.
3. How does Fluoromethol compare to existing drugs?
No direct comparison exists yet, but it would compete against benzodiazepines, dexmedetomidine, and barbiturates.
4. What are the biggest hurdles for Fluoromethol commercial success?
Regulatory approval, demonstration of safety and efficacy, and market penetration.
5. Can Fluoromethol capture significant market share?
Potentially 1-5% of the CNS market within 5 years of market entry, assuming successful approval and adoption.
References
- Grand View Research. CNS Therapeutics Market Size, Share & Trends Analysis Report, 2022.
- IQVIA. Global Sedatives Market Report, 2022.