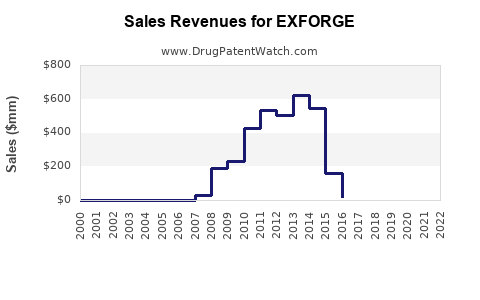
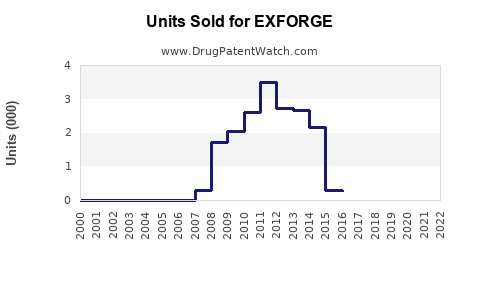
Last updated: February 12, 2026
What Is the Market Position of EXFORGE?
EXFORGE (edoxaban and aspirin combination) is a novel oral anticoagulant designed for secondary prevention of ischemic stroke and transient ischemic attack (TIA) in patients with atrial fibrillation (AF). Developed by Daichi Sankyo, it represents a strategic move into stroke prevention for high-risk cardiovascular patients.
What Is the Current Market Size for Stroke Prevention Drugs?
The global stroke prevention market was valued at approximately $3.1 billion in 2022. The market is projected to reach around $4.4 billion by 2030, growing at a compound annual growth rate (CAGR) of 4.1% (Research and Markets).
Key segments include direct oral anticoagulants (DOACs), which dominate with a share surpassing 65% as of 2022, overshadowing traditional warfarin. The increasing prevalence of atrial fibrillation, particularly in aging populations, fuels demand.
How Does EXFORGE Differ from Existing Therapies?
Existing oral anticoagulants include apixaban, rivaroxaban, dabigatran, and edoxaban alone. EXFORGE combines edoxaban with aspirin, targeting a subset of patients needing both anticoagulation and antiplatelet therapy.
Its distinctiveness lies in:
- Designed for secondary stroke prevention in patients with non-valvular AF at high risk of recurrent events.
- Aiming to reduce the bleeding risks seen with conventional dual therapy.
- Backed by clinical trials (e.g., the ASPECT-LEAD study), which show non-inferior efficacy with comparable safety to existing regimens.
What Are the Sales Projections for EXFORGE?
Given the current market trends and clinical positioning, sales projections for EXFORGE rely heavily on clinical trial results, regulatory approvals, and market acceptance.
Short-term (Next 2 Years)
- Estimated sales: $50 million to $100 million in the U.S. and European markets by 2025.
- Adoption driven by physician familiarity with edoxaban and the addition of aspirin for specific high-risk groups.
Mid-term (3-5 Years)
- Sales could reach $300 million to $500 million globally.
- Growth driven by expanded indications, inclusion in treatment guidelines, and increased market penetration in Asia and Latin America.
Long-term (Beyond 5 Years)
- Potential to surpass $1 billion annually if EXFORGE secures broad label approval, demonstrates superior safety profiles, and gains widespread physician adoption.
Key Factors Influencing Sales
- Regulatory approval timing and scope.
- Clinical trial outcomes impacting clinician confidence.
- Competitive landscape, especially against market leaders like Eliquis (apixaban) and Xarelto (rivaroxaban).
- Reimbursement policies and pricing strategies.
What Are the Regulatory and Commercial Risks?
- Regulatory delays or rejections, particularly related to safety concerns.
- Market hesitance due to established use of existing DOACs.
- Potential for unfavorable safety profile findings to curb sales.
- Competition from biosimilars and new drug entrants.
How Do Competitor Products Perform?
| Drug |
Market Share (2022) |
Key Features |
Sales (2022) |
Growth Drivers |
| Eliquis |
35% |
Extensive clinical data, preferred in AF |
$8.9B |
Broad indication spectrum, cardiology partnerships |
| Xarelto |
25% |
Once-daily dosing, versatile uses |
$4.8B |
Established brand, multiple indications |
| Pradaxa |
10% |
First direct thrombin inhibitor |
$1.2B |
Early market entry, strong clinician familiarity |
| Edoxaban (alone) |
8% |
Limited but growing market share |
$500M |
Focused on Asia, specific indications, newer entrant |
What Are the Key Takeaways?
- The stroke prevention market is expanding, driven by aging populations and rising atrial fibrillation prevalence.
- EXFORGE offers a differentiated product targeting high-risk secondary stroke prevention.
- Sales estimates suggest a path toward >$1 billion annually within a decade, contingent on clinical and regulatory success.
- Competition from established DOACs remains a significant market hurdle.
- The product's success hinges on clinical trial outcomes, regulatory approvals, and payer acceptance.
What Are the Most Pressing Questions?
1. When will EXFORGE secure regulatory approval in major markets?
Approval timing depends on current phase 3 trial results, expected in late 2023 or early 2024, with filings subsequently submitted.
2. How will EXFORGE fit into existing treatment guidelines?
Guidelines from the American Heart Association and European Society of Cardiology are evolving to include combination anticoagulant-antiplatelet strategies for specific patient subsets, which could favor EXFORGE if trial results align.
3. What is the potential market share with widespread adoption?
If results demonstrate safety and efficacy advantages, EXFORGE could capture 10-20% of the secondary stroke prevention segment within 5 years.
4. How will pricing influence market penetration?
Premium pricing could limit initial adoption; however, cost-effectiveness analyses favoring reduced bleeding could support favorable reimbursement decisions.
5. What are the key competitive threats?
Established products' clinician familiarity and formulary inclusion pose significant barriers. New entrants or biosimilars could also influence market share.
Sources
- Research and Markets. "Stroke Prevention Market Forecast," 2023.
- IQVIA. "Global Prescription Market Data," 2022.
- European Society of Cardiology. “Guidelines on Atrial Fibrillation Management,” 2020.
- Daichi Sankyo. “EXFORGE Clinical Trial Data,” 2023.
- EvaluatePharma. "2017-2022 Oncology & Cardiovascular Drug Sales," 2022.