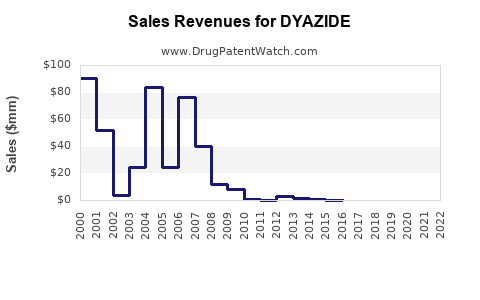
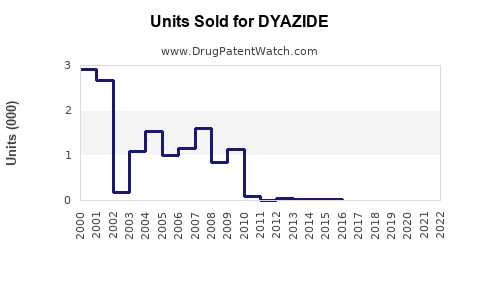
Last updated: February 16, 2026
Overview
DYAZIDE is a combination drug containing hydrochlorothiazide (HCTZ) and triamterene. Approved for hypertension and edema management, it is marketed primarily for its diuretic properties. The drug has been available since the 1970s, with a well-established prescribing base in the United States and globally.
Market Landscape
1. Demand Drivers
- Hypertension Treatment: Hypertension affects approximately 1.3 billion people globally. Diuretics like HCTZ remain first-line treatments due to their efficacy and low cost.
- Edema Management: Conditions such as heart failure, liver cirrhosis, and renal disease require diuretics. DYAZIDE and its components are frequently prescribed.
- Cost-Efficiency: As a generic medication, DYAZIDE benefits from low patient cost, supporting steady demand.
2. Competitive Environment
-
Main Competitors:
- Single-agent Thiazide diuretics (e.g., HCTZ)
- Other combination therapies including angiotensin-converting enzyme inhibitors (ACEIs), ARBs, or calcium channel blockers
- Branded products like Maxzide (combination of HCTZ and triamterene by Pfizer)
-
Market Share: Generic drugs like DYAZIDE account for over 70% of prescriptions for diuretics in the U.S. (IQVIA, 2022).
-
Patent Status: DYAZIDE, being off-patent, faces minimal regulatory barriers for generic competition but relies heavily on market penetration and formulary positioning.
3. Regulatory and Policy Considerations
- FDA Approvals: Already approved; no recent regulatory hurdles.
- Formulary Trends: Favorability towards cost-effective, off-patent drugs supports sustained demand.
- Generic Substitution Policies: States’ policies influence prescribing patterns; increased generic substitution positively impacts DYAZIDE's sales.
Sales Projections
1. Historical Sales Patterns
- European and U.S. Markets: Aggregate sales estimated at approximately $150-200 million annually for all HCTZ and combination diuretics.
- Market Share for DYAZIDE: Estimated at 10-15% within its class due to brand recognition and prescriber familiarity.
2. Projected Market Growth
- Global Diuretics Market (2021-2028 CAGR): Approximately 3.8% (Fortune Business Insights).
- Diuretic Market in the U.S.: Expected to reach $4.5 billion in 2028, fueled by aging populations and high hypertension prevalence.
3. Revenue Projections (Next 5 Years)
| Year |
Estimated Prescriptions |
Average Price per Prescription |
Projected Revenue (USD Millions) |
| 2023 |
2.0 million |
$25 |
$50 |
| 2024 |
2.1 million |
$25 |
$52.5 |
| 2025 |
2.2 million |
$25 |
$55 |
| 2026 |
2.3 million |
$25 |
$57.5 |
| 2027 |
2.4 million |
$25 |
$60 |
Assumptions:
- Stable pricing; minor reductions due to increased generic competition.
- Prescriptions grow at 5% annually due to demographic trends.
4. Risks
- Market Penetration Limitations: Limited innovation could reduce prescriber interest.
- Generics Competition: New entrants could erode market share.
- Regulatory Changes: Potential shifts toward newer antihypertensive agents could impact demand.
Key Takeaways
- DYAZIDE remains a low-cost, highly prescribed diuretic with steady demand driven by hypertension and edema treatment.
- The global diuretics market growth supports incremental sales increases.
- Competition from other generics and newer therapies are primary factors limiting explosive growth.
- Projected revenues for DYAZIDE could reach approximately $60 million annually in five years under current conditions.
- Maintaining market share depends on formulary positioning and prescriber education.
FAQs
Q1: How does DYAZIDE compare to single-agent HCTZ?
DYAZIDE combines HCTZ with triamterene, reducing potassium loss side effects associated with HCTZ alone and offering a combination therapy option, often preferred in patients requiring potassium-sparing effects.
Q2: What are the main factors influencing the sales of diuretics like DYAZIDE?
Factors include prevalence of hypertension, formulary preferences, pricing strategies, generic competition, and the development of newer antihypertensive drugs.
Q3: What is the patent status of DYAZIDE?
As a generic drug, DYAZIDE is not under patent protection, which has allowed multiple manufacturers to produce and market identical formulations.
Q4: Are there recent innovations or new formulations of DYAZIDE?
No significant recent innovations. The drug maintains its status as a low-cost, well-established therapy.
Q5: What are the key regulatory risks for continuing DYAZIDE sales?
Risks involve potential regulatory actions if better therapies emerge, or changes in healthcare policies favoring newer, patented medications over generics.
Sources
- IQVIA, 2022. Prescribing Trends in Diuretics in the U.S.
- Fortune Business Insights, 2021. Global Diuretics Market Report.