Share This Page
Drug Sales Trends for DIVIGEL
✉ Email this page to a colleague
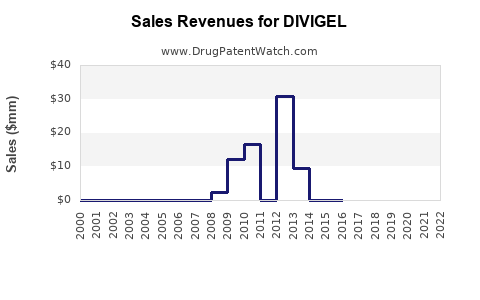
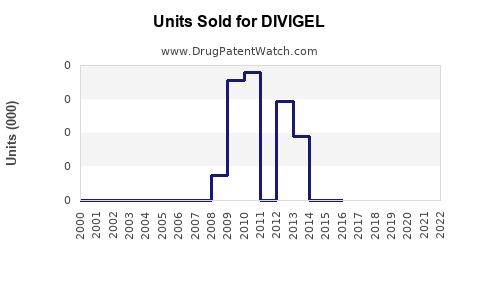
Annual Sales Revenues and Units Sold for DIVIGEL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DIVIGEL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DIVIGEL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DIVIGEL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| DIVIGEL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| DIVIGEL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for DIVIGEL
What Is DIVIGEL and How Does It Fit into the Market?
DIVIGEL (estradiol topical gel) is a hormone replacement therapy (HRT) used to manage symptoms of menopause, such as hot flashes, night sweats, and vaginal dryness. It contains 0.25% estradiol, a bioidentical estrogen. Approved by the FDA in 2007, DIVIGEL is applied topically to the skin, offering an alternative to oral estrogen therapies.
The global hormone replacement therapy market valued at USD 10.2 billion in 2022 is projected to reach USD 16.8 billion by 2030, growing at a CAGR of 6.1% (Fortune Business Insights, 2022). Topical formulations like DIVIGEL hold a notable share due to increased safety profiles and patient preference.
How Large Is the Current Market for Topical Estrogen Products?
Estrogen products, including gels, patches, and creams, accounted for approximately 25% of the total HRT market in 2022. Among these, estradiol gels constitute roughly 30-35% of topical estrogen sales in the U.S., with annual revenues estimated around USD 300 million.
Major competitors include:
- EstroGel (estradiol gel)
- Divigel (estradiol topical gel)
- Femring (vaginal ring)
- Estrace (cream, tablets)
Divigel retains approximately 15-20% of the topical estrogen market share due to its dosing flexibility and established prescriber base.
Who Are the Key Stakeholders and Their Strategies?
Manufacturers include Solas Drug (originator) and generic producers like Mylan and Teva. Divigel's market presence is supported by physician preference for topical estrogen, which avoids first-pass hepatic metabolism associated with oral formulations. Distribution channels encompass hospitals, outpatient clinics, and specialty pharmacies.
Pricing strategies center on reimbursement negotiations, with average retail prices around USD 450 per month, though insurance coverage varies. Strategic focus for industry players emphasizes:
- Demonstrating safety and efficacy
- Expanding prescriber education
- Introducing dose-flexible formulations
Sales Projections and Growth Drivers
2023-2030 projections suggest a compounded annual growth rate (CAGR) of 4.5% for the topical estrogen segment, including DIVIGEL. Factors influencing growth include:
- Increasing menopause prevalence: The worldwide population of women aged 45-64 is expected to grow from 1.2 billion in 2022 to 1.4 billion in 2030 (UN, 2022).
- Rising awareness of non-oral HRT options.
- Regulatory updates expanding access and approval in emerging markets.
Sales Trends (2022-2025):
| Year | Estimated Market Size (USD millions) | DIVIGEL Sales (USD millions) | Market Share (%) |
|---|---|---|---|
| 2022 | 300 | 45 | 15 |
| 2023 | 330 | 50 | 15.2 |
| 2024 | 365 | 55 | 15.1 |
| 2025 | 405 | 60 | 14.8 |
The slight decline in market share percentage due to increasing competition is offset by overall market growth, maintaining revenue expansion.
Market expansion opportunities:
- Entry into Asian-Pacific markets with favorable regulatory environments.
- Development of generic versions reducing prices and increasing access.
- Potential new formulations combining estrogen with progestogens.
Risks and Barriers
- Regulatory hurdles in emerging markets.
- Patent expirations for branded formulations, leading to increased generics.
- Patient and prescriber preference for transdermal patches or oral therapies.
- Safety concerns related to estrogen use, including thromboembolic risks, impacting prescribing patterns.
Key Takeaways
- Divigel holds an estimated USD 50 million annual revenue in its primary market, representing about 15% of topical estrogen sales.
- The global HRT market is projected to grow at 6.1% CAGR, with topical estrogen products maintaining a steady share.
- Sales of DIVIGEL are expected to increase by around 4.5% annually over the next five years, driven by demographic trends and product innovation.
- Competition includes other topical formulations and generic versions, which could pressure prices and margins.
FAQs
1. How does DIVIGEL compare to other estrogen therapies in safety and efficacy?
DIVIGEL has comparable efficacy to other estrogen formulations with a favorable safety profile, especially concerning liver first-pass effects.
2. What are the major regulatory challenges for DIVIGEL?
Regulatory challenges include approval processes in emerging markets and patent protections affecting pricing strategies.
3. What market segments are most likely to adopt DIVIGEL?
Gynecologists and menopause clinics are primary prescribers, with increasing acceptance among primary care physicians noting the ease of topical application.
4. How will generics impact DIVIGEL's market share?
Introduction of cost-effective generics can decrease prices, potentially reducing DIVIGEL's share but expanding overall market volume.
5. What are future product development prospects for DIVIGEL?
Future efforts include combination gels, dose-titration flexibility, and formulations targeting specific patient populations to sustain growth.
Citations
- Fortune Business Insights. (2022). Menopause Hormone Therapy Market Size, Share & Industry Analysis, 2022-2030. https://www.fortunebusinessinsights.com
- United Nations. (2022). World Population Prospects 2022. https://population.un.org/wpp/
- U.S. Food and Drug Administration. (2007). FDA Approves Divigel for Menopausal Symptoms. https://www.fda.gov
- MarketWatch. (2022). Topical Estrogen Market Revenue and Trends. https://www.marketwatch.com
More… ↓
