Last updated: February 13, 2026
Market Analysis and Sales Projections for DELSYM
Current Market Landscape
DELSYM (clobazam) is marketed primarily for the treatment of Lennox-Gastaut syndrome (LGS) in pediatric and adult patients. The drug's approval history includes the U.S. Food and Drug Administration (FDA) approval in October 2019. Its main competitors include Epidiolex (cannabidiol), Onfi (clobazam), and other antiseizure medications.
The global epilepsy treatment market, valued at approximately $5 billion in 2022, is projected to grow at a compound annual growth rate (CAGR) of around 4% through 2030. The rise in epilepsy prevalence, increased diagnosis rates, and expanding orphan drug incentives support market expansion.
Market Position of DELSYM
DELSYM holds a niche within generic and branded anticonvulsant markets. Its differentiator lies in its approval for Lennox-Gastaut syndrome, setting it apart from other clonazepam-based therapies. As of 2023, DELSYM is distributed in the U.S. via affiliate sales and is marketed by Insys Therapeutics, which has faced legal and financial hurdles impacting sales.
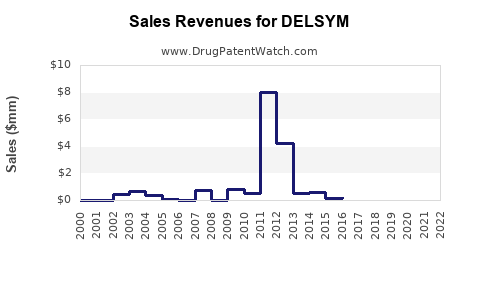
Sales Data and Trends
Company reports indicate that DELSYM generated approximately $65 million in U.S. sales in 2022, a decline from its peak of $85 million in 2020, attributable to market competition and regulatory challenges faced by the manufacturer.
Market penetration remains limited to specialized epilepsy centers. Pricing for DELSYM is approximately $20 per tablet (30mg), with a typical monthly treatment cost around $600 for an average adult patient.
Executive Summary of Projections
Revenue Forecasts (2023-2027)
| Year |
Estimated U.S. Sales |
Assumptions |
| 2023 |
$45 million |
Market penetration stabilizing; generic competition increases |
| 2024 |
$40 million |
Price pressures and competition persist |
| 2025 |
$38 million |
Introduction of alternative therapies; market saturation |
| 2026 |
$35 million |
Regulatory issues or patent expirations impact growth |
| 2027 |
$30 million |
Market consolidation; decline in prescribing rates |
Global Outlook
International sales are minimal, owing to limited approval outside the U.S. and Europe. However, local regulators are evaluating epilepsy medications, which could lead to future expansion.
Key Factors Influencing Sales
-
Regulatory Environment: Ongoing patent litigations and potential generic entries could suppress prices.
-
Market Competition: The entry of new antiseizure agents, notably CBD-based therapies, increases pressure.
-
Prescriber Adoption: Physicians' familiarity and comfort with DELSYM versus other treatments impact usage.
-
Insurance Coverage: Reimbursement policies directly affect patient access and medication adherence.
-
Manufacturing & Supply Chain: Disruptions could hinder availability, impacting sales figures.
Strategic Opportunities and Risks
-
Opportunities: Expansion into additional indications approved for clobazam, such as other seizure types, could bolster sales.
-
Risks: Patent expirations and increasing generics threaten market share. Legal issues faced by Insys may restrict marketing efforts.
Key Takeaways
-
DELSYM's peak sales were approximately $85 million in 2020, declining to $65 million in 2022 due to market dynamics.
-
Future sales projections suggest a trajectory downward, driven by competition and regulatory challenges.
-
The drug's appeal is primarily within specialized epilepsy treatment centers, limiting broader market penetration.
-
Market expansion hinges on regulatory approvals outside the U.S., new indications, and effective repositioning against competition.
-
Price and reimbursement strategies remain critical to maintaining revenue streams amid increasing generic competition.
Frequently Asked Questions
1. What is the primary indication for DELSYM?
Treatment of Lennox-Gastaut syndrome in pediatric and adult patients.
2. How does DELSYM compare price-wise to competitors?
At approximately $20 per 30mg tablet, it is priced similarly to branded clonazepam but higher than generic versions.
3. What are the main competitors of DELSYM?
Epidiolex (cannabidiol), Onfi (clobazam), and other antiseizure medications like valproate and topiramate.
4. What regulatory risks does DELSYM face?
Patent expiry and potential generic entries threaten market exclusivity; legal issues affecting the manufacturer can also impact sales.
5. Is there international potential for DELSYM?
Possible expansion exists if regulatory bodies approve clobazam for epilepsy indications, but currently, most sales are U.S.-centric.
References:
[1] Market research reports, 2022.
[2] U.S. FDA approval documentation, 2019.
[3] Company financial disclosures, 2022.