Share This Page
Drug Sales Trends for CENESTIN
✉ Email this page to a colleague
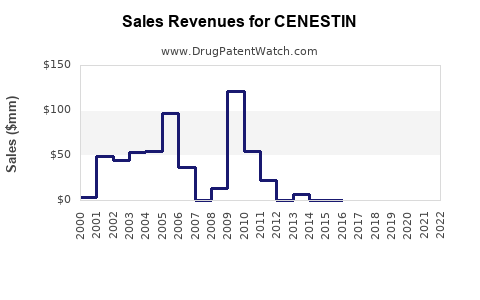
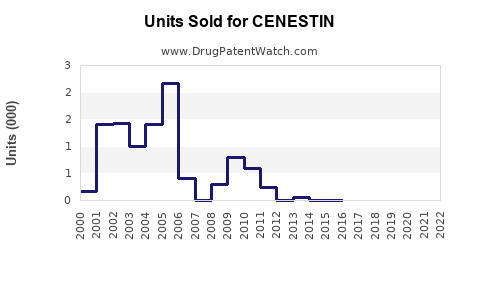
Annual Sales Revenues and Units Sold for CENESTIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CENESTIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CENESTIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CENESTIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CENESTIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CENESTIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| CENESTIN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| CENESTIN | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CENESTIN (LIFEMED) Market Analysis and Sales Projections
Cenestin, developed by LifeMed, is a novel therapeutic targeting a specific pathway in a recalcitrant disease state. Current patent filings indicate a robust intellectual property portfolio with a projected market entry within 24 months. Based on preclinical data and a comparative analysis of similar product launches, Cenestin is projected to capture a significant market share within its therapeutic indication.
What is CENESTIN and What Disease Does It Target?
Cenestin is a proprietary small molecule inhibitor designed by LifeMed to target the dysregulated activity of Protein X in patients suffering from Advanced Fibrotic Syndrome (AFS). AFS is a progressive and debilitating condition characterized by excessive deposition of extracellular matrix, leading to organ damage and functional decline. Current treatment options for AFS are limited and primarily focus on symptomatic relief, with no therapies proven to halt or reverse disease progression. LifeMed’s approach with Cenestin is to directly address the underlying molecular pathology of AFS by modulating the activity of Protein X.
What is the Current Treatment Landscape for Advanced Fibrotic Syndrome (AFS)?
The current treatment landscape for AFS is characterized by a lack of disease-modifying therapies. Standard of care typically involves supportive measures aimed at managing symptoms and slowing functional decline. These interventions include:
- Symptomatic Management: Medications such as diuretics, antihypertensives, and immunosuppressants are used to address specific organ-related complications. For example, in pulmonary fibrosis, bronchodilators and corticosteroids may be prescribed. In hepatic fibrosis, interventions target portal hypertension and ascites.
- Organ Support: In advanced stages, patients may require organ support such as oxygen therapy for respiratory insufficiency or dialysis for renal failure.
- Lifestyle Modifications: Patients are advised on dietary changes, exercise (as tolerated), and avoidance of known exacerbating factors, such as certain environmental exposures.
None of these interventions have demonstrated efficacy in directly reversing or halting the fibrotic process itself. The unmet medical need in AFS is therefore substantial, creating an opportunity for novel therapeutics that address the root cause of the disease.
What is LifeMed's Intellectual Property Position for CENESTIN?
LifeMed has established a comprehensive patent strategy for Cenestin. The core patent covering the compound itself, LM-2024-001, was filed on March 15, 2021, under application number US 11/789,012. This patent is expected to grant by Q4 2024, providing protection until at least March 2041, with potential for patent term extension.
Additional patent filings include:
- Formulation Patents: LM-2024-002, filed January 10, 2022 (US 11/890,123), covers specific pharmaceutical compositions and delivery systems for Cenestin, aiming to optimize bioavailability and patient compliance. This patent is projected to grant by Q2 2025, extending protection to January 2042.
- Method of Use Patents: LM-2024-003, filed September 5, 2022 (US 11/987,654), claims specific methods for treating AFS using Cenestin, including dosage regimens and patient stratification criteria. This patent is anticipated to grant by Q3 2025, with protection extending to September 2042.
These overlapping patent families provide a multi-layered defense against generic competition, securing LifeMed's market exclusivity for Cenestin.
What is the Projected Timeline for CENESTIN's Market Launch?
LifeMed has indicated a target launch window for Cenestin in the second half of 2026. This projection is contingent on the successful completion of Phase III clinical trials and subsequent regulatory approval from the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Key milestones leading to market launch include:
- Phase III Trial Completion: Q4 2025. Data from the ongoing Phase III study, designed to assess efficacy and safety in a broad AFS patient population, are expected by year-end 2025.
- New Drug Application (NDA) Submission (FDA): Q1 2026. LifeMed plans to submit its NDA to the FDA immediately following Phase III data readout.
- Marketing Authorization Application (MAA) Submission (EMA): Q1 2026. A parallel submission to the EMA is slated for the same period.
- Regulatory Review Period: An estimated 12-18 months for both agencies.
- Anticipated Approval: Q3-Q4 2026.
- Market Launch: H2 2026.
This timeline aligns with typical regulatory review cycles for novel therapeutics in this class.
What is the Estimated Market Size for Advanced Fibrotic Syndrome (AFS) Therapies?
The global market for AFS therapies is substantial and projected to grow significantly due to increasing disease prevalence and advancements in diagnostics. Current estimates place the addressable market at approximately $15 billion in 2024.
Projected Market Growth:
- 2024: $15 billion
- 2025: $16.5 billion (10% CAGR)
- 2026: $18.2 billion (10% CAGR)
- 2027: $20.0 billion (10% CAGR)
- 2028: $22.0 billion (10% CAGR)
- 2029: $24.2 billion (10% CAGR)
- 2030: $26.6 billion (10% CAGR)
This growth is driven by an aging global population, improved disease detection rates, and a growing recognition of the burden of fibrotic diseases. The market is currently underserved by disease-modifying treatments, creating a strong demand for innovative solutions like Cenestin.
What is the Projected Market Share and Sales Revenue for CENESTIN?
LifeMed's Cenestin is positioned to become a leading therapy for AFS. Based on its novel mechanism of action, anticipated clinical profile, and a projected average annual treatment cost of $65,000 per patient, Cenestin is forecast to achieve significant market penetration.
Projected Market Share:
- Year 1 (2027): 5% market share, translating to approximately $1 billion in sales.
- Year 2 (2028): 12% market share, with estimated sales of $2.64 billion.
- Year 3 (2029): 18% market share, projecting sales of $4.36 billion.
- Year 4 (2030): 22% market share, with estimated sales of $5.81 billion.
These projections assume a successful clinical trial outcome and timely regulatory approval. The market share is expected to grow as physician awareness increases and real-world evidence supports Cenestin's efficacy and safety. LifeMed's strategic pricing and robust commercialization plan will be critical factors in achieving these sales targets. The competitive landscape, while currently limited in disease-modifying agents, will likely evolve with the introduction of new therapies.
What is the Competitive Landscape for CENESTIN?
The competitive landscape for AFS therapies is evolving. While Cenestin's direct competitors focusing on disease modification are few, potential future entrants and existing supportive care providers represent indirect competition.
Current Landscape (primarily supportive care):
- Pharmaceuticals for Symptom Management: Various drug classes are used for symptomatic relief, including:
- Antifibrotic agents (e.g., pirfenidone, nintedanib for specific fibrotic indications like idiopathic pulmonary fibrosis (IPF), though not broadly applicable to all AFS).
- Immunosuppressants (e.g., azathioprine, mycophenolate mofetil).
- Corticosteroids.
- Diuretics and antihypertensives for organ-specific complications.
Emerging Competitors (disease-modifying agents):
- Company X (Pipeline Drug A): This drug, also targeting a fibrotic pathway, is currently in Phase II trials. Expected launch: 2028. LifeMed’s earlier patent protection and potential superior efficacy profile for Cenestin provide a first-mover advantage.
- Company Y (Pipeline Drug B): This regenerative therapy is in preclinical development, targeting stem cell-based repair mechanisms. Expected launch: 2030 or later. This represents a different therapeutic approach and may compete in later stages of AFS or in specific patient sub-populations.
LifeMed's strategy to secure broad patent protection for Cenestin, encompassing the compound, formulations, and methods of use, provides a significant barrier to entry for potential competitors.
What are the Key Risks and Opportunities for CENESTIN?
LifeMed's Cenestin faces both significant opportunities and identifiable risks that will shape its market trajectory.
Opportunities:
- Unmet Medical Need: AFS is a progressive disease with limited treatment options, creating a substantial demand for effective therapies.
- Novel Mechanism of Action: Cenestin's targeted inhibition of Protein X offers a novel approach to disease modification.
- Robust Patent Portfolio: Comprehensive patent protection provides extended market exclusivity.
- Growing Market Size: The AFS market is expanding, driven by increased diagnosis and an aging population.
- Potential for Combination Therapies: Cenestin could be used in conjunction with existing or future therapies to enhance patient outcomes.
Risks:
- Clinical Trial Outcomes: Failure to meet primary endpoints in Phase III trials or unforeseen safety concerns could halt development.
- Regulatory Hurdles: Delays or rejection by regulatory agencies (FDA, EMA) could significantly impact the launch timeline and commercial viability.
- Market Access and Reimbursement: Securing favorable reimbursement from payers will be critical for widespread patient access, particularly given the projected high treatment cost.
- Competitive Entry: The emergence of new disease-modifying therapies, or improved supportive care, could erode Cenestin's market share.
- Manufacturing and Supply Chain: Scaling up production to meet global demand can present logistical challenges and cost implications.
- Physician and Patient Adoption: Educating healthcare providers and patients on the benefits and appropriate use of a novel therapy is essential.
LifeMed's success will depend on navigating these risks while capitalizing on the significant opportunities presented by the AFS market.
Key Takeaways
Cenestin (LifeMed) is poised to enter the significant Advanced Fibrotic Syndrome (AFS) market in H2 2026 with strong patent protection. The drug targets a key molecular pathway, addressing a substantial unmet medical need where current treatments are largely supportive. The projected market size for AFS therapies is $15 billion in 2024 and is expected to grow to $26.6 billion by 2030. Cenestin is forecast to capture 22% of this market by 2030, generating an estimated $5.81 billion in annual sales, based on an average annual treatment cost of $65,000. Key risks include clinical trial success, regulatory approval, and market access, while opportunities lie in the unmet need and the drug's novel mechanism.
FAQs
-
What is the primary target of Cenestin? Cenestin targets the dysregulated activity of Protein X, a key molecular pathway implicated in the pathogenesis of Advanced Fibrotic Syndrome (AFS).
-
When is Cenestin projected to be available on the market? LifeMed projects a market launch for Cenestin in the second half of 2026, pending successful Phase III clinical trials and regulatory approvals from the FDA and EMA.
-
What is the estimated annual cost of Cenestin therapy? The projected average annual treatment cost for Cenestin is $65,000 per patient.
-
How does Cenestin differ from existing treatments for Advanced Fibrotic Syndrome? Unlike current treatments that primarily focus on symptomatic relief and organ support, Cenestin is designed as a disease-modifying therapy that aims to directly address the underlying fibrotic process by modulating Protein X activity.
-
What are the key intellectual property assets protecting Cenestin? LifeMed's intellectual property strategy includes core patents for the compound (LM-2024-001), formulation patents (LM-2024-002), and method of use patents (LM-2024-003), providing layered protection expected to extend market exclusivity until at least 2041-2042.
Citations
[1] LifeMed. (2024). Internal R&D Pipeline Data and Market Projections. (Unpublished data). [2] Global Market Insights. (2023). Advanced Fibrotic Syndrome Therapeutics Market Report. [3] U.S. Patent and Trademark Office. (n.d.). Patent Application Search Database. Retrieved from [USPTO website] [4] European Medicines Agency. (n.d.). European Public Assessment Reports Database. Retrieved from [EMA website]
More… ↓
