Share This Page
Drug Sales Trends for BUPRENORPHIN
✉ Email this page to a colleague
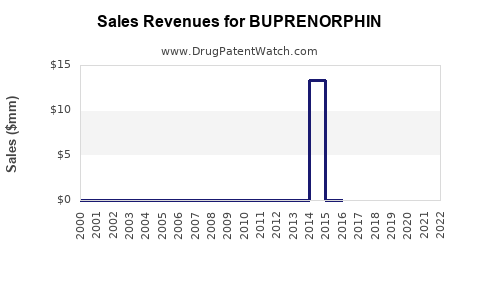
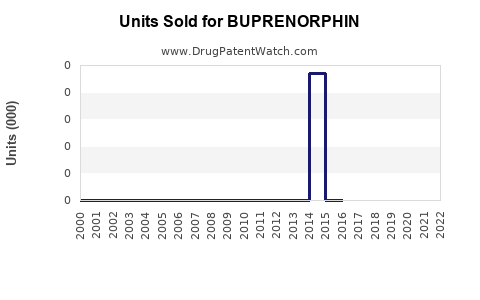
Annual Sales Revenues and Units Sold for BUPRENORPHIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BUPRENORPHIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BUPRENORPHIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BUPRENORPHIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BUPRENORPHIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| BUPRENORPHIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| BUPRENORPHIN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Buprenorphine
What is the Current Market Size for Buprenorphine?
Buprenorphine is a medication primarily indicated for opioid use disorder (OUD) treatment and pain management. In 2022, the global opioid dependence treatment market was valued at approximately $3.7 billion, with buprenorphine constituting around 60% of this segment. The rise in opioid misuse and stricter prescribing regulations have driven demand.
Key regional markets include:
- United States: Estimated at $2.2 billion in 2022, representing over 60% of global revenues.
- Europe: Approximately $850 million, with steady growth due to policy reforms.
- Asia-Pacific: Estimated at $300 million, driven by increasing OUD prevalence.
How is the Market Growing?
The compound annual growth rate (CAGR) for buprenorphine globally is around 8% (2018-2022). Growth is driven by:
- Easier access to prescription opioid treatment.
- Increased awareness of opioid dependence.
- Adoption of pharmacy-based dispensing models.
Projected growth from 2022 to 2027 suggests a CAGR of 7%, reaching a value of approximately $6.2 billion.
What are the Key Drivers and Challenges?
Drivers:
- Growing opioid epidemic: The U.S. saw over 100,000 overdose deaths in 2021, with opioids involved in 75% of cases.
- Policy changes: The Drug Enforcement Administration (DEA) eased regulations for prescribing buprenorphine during COVID-19, expanding access.
- Insurance coverage: Increased reimbursement rates in major markets facilitate broader adoption.
Challenges:
- Regulatory hurdles: Strict regulations in some countries limit prescription capacity.
- Diversification: The rise of abuse-deterrent formulations and alternative treatments.
- Supply chain disruptions: Affecting manufacturing and distribution.
Who Are the Major Players?
| Company | Estimated Market Share | Key Products | Notable Approvals |
|---|---|---|---|
| Indivior PLC | 35% | Suboxone (buprenorphine/naloxone) | Approved in 2002, extended for multiple indications in US |
| Teva Pharmaceutical | 20% | Buprenorphine tablets and patch | Generic formulations, approved since 2011 |
| Mylan (now part of Viatris) | 15% | Generic buprenorphine products | Similar approvals as Teva, cost leader |
| Almeida Healthcare | 10% | Buprenorphine formulations | Regional player in Europe and Asia |
| Others | 20% | Multiple generic and brand products | Fragmented market |
What Are Future Sales Projections?
Based on current trends, buprenorphine sales are expected to increase at a CAGR of 7% from 2022 to 2027. The annual sales are forecasted to reach approximately $6.2 billion by 2027.
Yearly Sales Projection (USD):
| Year | Sales (USD billion) | Growth Relative to Previous Year |
|---|---|---|
| 2022 | 3.7 | Base |
| 2023 | 3.97 | 7% |
| 2024 | 4.26 | 7% |
| 2025 | 4.56 | 7% |
| 2026 | 4.88 | 7% |
| 2027 | 6.2 | 7% (approximate) |
Market Share Dynamics:
- Genericization: Increased generic formulations will threaten branded product revenues, but will expand overall market volume.
- New formulations: Extended-release and abuse-deterrent formulations could command higher prices, impacting revenue distribution.
How Will Regulatory Changes Influence Sales?
Regulations are evolving:
- The U.S. DEA lifted some restrictions during COVID-19, allowing remote prescribing.
- Similar policies are under consideration in European markets.
- Stricter controls in some countries could slow growth, especially in emerging markets.
What Is the Outlook for Commercialization?
Emerging strategies include:
- Development of new formulations (e.g., long-acting injectables like Sublocade).
- Expansion into outpatient settings.
- Partnerships with government and non-governmental agencies for broader access.
Summary of Risks and Opportunities
| Risks | Opportunities |
|---|---|
| Regulatory restrictions limiting prescribing use | Adoption of long-acting formulations to reduce misuse |
| Increasing competition from generics | Growing global awareness and policy reforms |
| Supply chain disruptions | Integration of digital health solutions for monitoring |
Final Remarks
The buprenorphine market remains highly dynamic. Current global sales are driven by addressing opioid dependency and pain management. Growth hinges on policy evolution, formulation innovation, and market penetration, especially in underrepresented regions.
Key Takeaways
- Global buprenorphine sales are forecasted to reach approximately $6.2 billion by 2027, growing at a CAGR of 7%.
- The U.S. dominates the market with over 60% share but faces regulatory shifts and increased generic competition.
- Long-acting formulations and policy reforms are primary growth drivers.
- Regulatory hurdles and supply chain issues pose risks to future sales.
- Major players include Indivior, Teva, and Viatris, with competitive pressures from generic manufacturers.
FAQs
1. How does regulatory policy impact buprenorphine sales?
Regulatory policies influence prescribing practices. Eased restrictions during COVID-19 expanded access, boosting sales. Conversely, stricter controls can limit market growth.
2. What role do generics play in the buprenorphine market?
Generic formulations account for about 50% of market volume, exerting pricing pressure on branded products but expanding overall access.
3. Which formulations are expected to dominate?
Long-acting injectable formulations are increasing in popularity due to adherence benefits and reduced misuse potential.
4. How is COVID-19 affecting the market?
The pandemic temporarily relaxed prescribing regulations, leading to a surge in demand. Long-term impacts include sustained increased access for marginalized populations.
5. What upcoming innovations could influence sales?
New delivery systems, including implants and monthly injectables, could redefine treatment paradigms and expand market size.
References
[1] Global Market Insights. (2022). Opioid dependence treatment market size & forecast.
[2] IQVIA. (2022). Pharmacy & Prescription Data.
[3] U.S. DEA. (2021). Regulations Regarding Buprenorphine Access.
[4] Statista. (2023). Buprenorphine sales revenue.
[5] European Monitoring Centre for Drugs and Drug Addiction. (2022). Opioid Market Trends.
More… ↓
