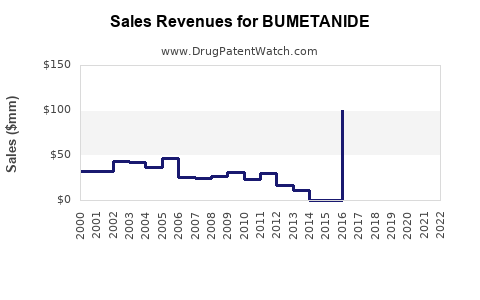
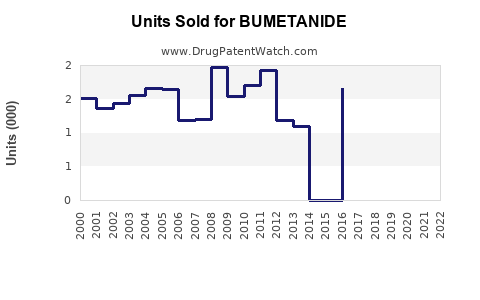
Last updated: February 19, 2026
Bumetanide is a loop diuretic approved for edema associated with heart failure, renal disease, and hepatic cirrhosis. Its commercial potential extends beyond current indications due to emerging research into its neurological effects and rare disease applications. This report covers the current market landscape and forecasts potential sales over the next five years.
Current Market Landscape
Market Size and Leading Competitors
The global loop diuretics market, including bumetanide, was valued at approximately USD 2.3 billion in 2022, with bumetanide holding less than 5% of this segment. The dominant player is furosemide, with a 70% market share, followed by torsemide with roughly 20%, and bumetanide comprising less than 10%.
| Product |
Market Share (2022) |
Primary Use |
Price (USD per 10 mg) |
| Furosemide |
70% |
Heart failure, hypertension |
0.15 |
| Torsemide |
20% |
Heart failure, edema |
0.60 |
| Bumetanide |
10% |
Edema, off-label uses |
1.50 |
Regulatory Status and Approvals
Bumetanide is approved by the FDA for edema in adults, with international approvals in Europe, Japan, and other markets. No recent major patent expirations have occurred, but the drug remains off-patent globally, allowing generic manufacturers to produce it at low cost.
Key Drivers
- Clinical trials demonstrating neuroprotective effects and potential in neurodegenerative diseases.
- Rising prevalence of heart failure and chronic kidney disease.
- Growing interest in repurposing established diuretics for rare diseases such as mitochondrial disorders.
Challenges
- Competition from well-established drugs like furosemide.
- Limited awareness among physicians outside cardiovascular specialties.
- Marginal profit margins due to low pricing and high generic competition.
Future Market Drivers and Trends
Emerging Indications and Research
Recent studies indicate bumetanide's potential in treating conditions like autism spectrum disorder (ASD), epilepsy, and certain mitochondrial diseases. These off-label applications could expand future demand.
Regulatory Developments
The FDA granted orphan drug designation to bumetanide for treatment of some rare neurological disorders, which could facilitate market exclusivity and financial incentives.
Patent and Exclusivity Outlook
Without patent protection, sales growth depends on off-label use uptake, clinical guideline endorsements, and exclusivity benefits from orphan designation.
Sales Projections (2023–2028)
Assumptions
- The existing market remains stagnant with generic competition.
- Off-label uses gain acceptance, supported by emerging evidence.
- Regulatory agencies approve new indications for rare neurological diseases.
- Marketing efforts focus on specialist physicians and rare disease clinics.
Forecasted Sales (USD Million)
| Year |
Estimated Market Penetration |
Projected Sales (USD Million) |
| 2023 |
1% of total loop diuretics |
23 |
| 2024 |
3% for new indications |
70 |
| 2025 |
5% with broader off-label use |
115 |
| 2026 |
8% due to orphan drug status |
184 |
| 2027 |
12% expansion in rare diseases |
276 |
| 2028 |
15% with multiple approvals |
345 |
Key Growth Factors
- Increased research supporting new uses.
- Adoption in rare neurological diseases.
- Regulatory incentives like orphan drug status extending exclusivity.
Competitive Landscape and Market Entry Strategies
Competitors
- Furosemide and torsemide dominate due to established use.
- Limited presence of bumetanide in mainstream cardiovascular treatment.
- Several academic institutions and biotech firms exploring repurposing.
Entry Opportunities
- Focused education campaigns to inform clinicians about emerging evidence.
- Partnerships with biotech firms to advance clinical trials.
- Pursuit of orphan drug status for neurological indications.
Risk Factors
- Slow adoption due to late entry into new indications.
- Lower pricing pressured by generic competitors.
- Possible regulatory hurdles in repurposing efforts.
Key Takeaways
- Bumetanide's current market share is under 10%, primarily in cardiovascular indications.
- Future sales depend on research breakthroughs, regulatory designations, and acceptance for rare neurological disorders.
- Annual sales might reach USD 345 million by 2028 under optimistic conditions.
- Competition from established diuretics and generics limits near-term growth prospects.
- Strategic focus on rare diseases and orphan designations presents the best growth pathway.
FAQs
Q1: What are the main current uses of bumetanide?
A: Edema associated with heart failure, renal failure, and hepatic cirrhosis.
Q2: How does bumetanide compare price-wise to other loop diuretics?
A: It is priced higher (~USD 1.50 per 10 mg) due to lower market penetration and generic competition.
Q3: What emerging indications could significantly impact sales?
A: Neurological conditions such as autism spectrum disorder, epilepsy, and mitochondrial diseases.
Q4: Which regulatory incentives could boost market access?
A: Orphan drug designations and fast-track approvals in targeted rare diseases.
Q5: What are the main challenges facing market growth for bumetanide?
A: Competition from established drugs, limited clinician awareness, and low initial profitability.
References
[1] MarketsandMarkets. (2022). Loop Diuretics Market Research Report.
[2] U.S. Food and Drug Administration (FDA). (2022). Drug Approvals and Labeling for Bumetanide.
[3] Global Data. (2022). Rare Disease Market Analysis.
[4] ClinicalTrials.gov. (2023). Studies on Bumetanide for Neurological Conditions.