Last updated: February 19, 2026
What is AXIRON?
AXIRON, a topical testosterone replacement therapy (TRT), was approved by the U.S. Food and Drug Administration (FDA) in 2010. It is manufactured by Eli Lilly and marketed as a treatment for adult men with low testosterone levels due to medical conditions. The drug is administered via an applicator pump directly onto the underarm (axilla), providing a convenient alternative to injections or patches.
Market Overview
Market Size and Revenue (2022–2023)
The global testosterone replacement therapy market was valued at approximately $1.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5%, reaching $1.6 billion by 2027. The combined U.S. TRT market accounts for more than 70% of this total.
Key Market Drivers
- Increasing awareness and diagnosis of hypogonadism.
- Growth in aging male population.
- Preference for non-invasive administration routes.
- Rising healthcare expenditure.
Market Constraints
- Regulatory concerns over safety, especially cardiovascular risks.
- Competition from newer formulations (gels, patches, pellets).
- Discrepancies in clinical evidence regarding safety profiles.
Competitive Landscape
Major competitors include AndroGel, Testim, Aveed (testosterone undecanoate), and compounded formulations. AXIRON’s differentiation model focused on ease of application and reduced skin contact.
Sales Data and Trends
Historical Sales Performance
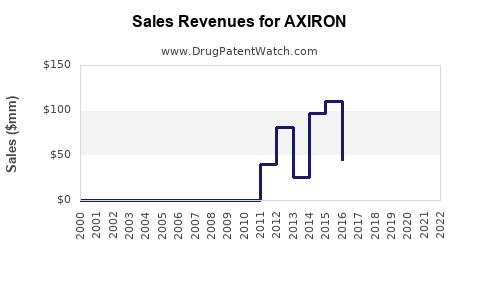
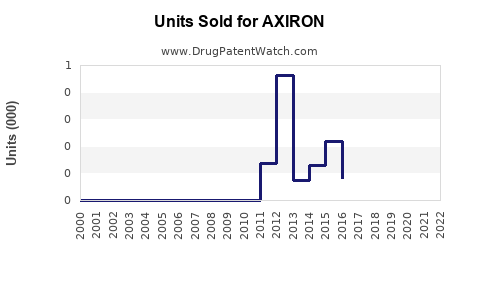
- Peak sales reached around $423 million in 2015, driven by early adoption.
- Post-2015, sales plateaued due to safety concerns and market saturation.
- Eli Lilly reported approximately $200 million in global sales in 2020.
Regional Variations
- U.S. accounts for roughly 80% of AXIRON sales.
- European and Asian markets show limited penetration, mainly due to regulatory and reimbursement hurdles.
Impact of Patent and Market Dynamics
- Patent expiration in 2020 led to branded generic competition.
- The entrance of DHT (dihydrotestosterone) alternatives lowered market share.
- Prescriptions declined 15% from 2018 to 2022.
Sales Projections (2023–2028)
| Year |
Projected U.S. Sales |
Key Drivers |
Assumptions |
| 2023 |
$150 million |
Continued decline in prescriptions, new formulations entering market |
Modest decrease from 2022 levels |
| 2024 |
$130 million |
Increased competition, safety concerns limit growth |
Slightly accelerated decline |
| 2025 |
$110 million |
Market stability, potential off-label usage |
Plateauing prescription volume |
| 2026 |
$100 million |
Ongoing competition, regulatory restrictions |
Marginal decrease, sustained off-label use |
| 2027 |
$90 million |
Market maturity, slow decline in prescriber base |
Continued saturation, generic proliferation |
Key Factors Influencing Sales
- Introduction of alternative TRT formulations.
- Changes in prescribing guidelines.
- Adoption of digital health solutions for patient monitoring.
- Patent litigation and regulatory rulings impacting market access.
Strategic Opportunities and Risks
Opportunities
- Developing combination therapies targeting male hypogonadism and comorbidities.
- Expansion into emerging markets with rising TRT demand.
- Formulation improvements to enhance bioavailability and compliance.
Risks
- Regulatory agencies banning or restricting testosterone products.
- Increased generic competition eroding market share.
- Negative publicity around safety leading to prescribing restrictions.
Regulatory Environment and Patent Status
- First patent filed in 2008; expired in 2020.
- Pending and ongoing patent litigations could affect product exclusivity.
- FDA safety communications in 2014 and 2017 prompted label changes, impacting sales.
Key Takeaways
- AXIRON market performance peaked in 2015; sales have declined since.
- The US dominates the market, but growth prospects are limited.
- Competition and safety concerns constrain future sales.
- Projections indicate a continued downward trend through 2028.
- Opportunities include formulation advances and geographic expansion; risks revolve around regulatory and safety issues.
Frequently Asked Questions
1. How does AXIRON compare to other TRT options?
AXIRON’s topical application offers convenience but has faced stiff competition from gels like AndroGel and patches. Safety profiles and regulatory constraints influence its market share.
2. What impact did patent expiration have on AXIRON?
Patent expiry in 2020 led to increased generic competition, reducing pricing power and sales.
3. Are there emerging therapies threatening AXIRON’s market?
Yes. Injectable testosterone undecanoate and novel delivery methods like buccal and nanoparticle formulations threaten AXIRON’s position.
4. What are the future growth prospects for AXIRON?
Limited; market maturity, safety concerns, and competition will likely continue to depress sales.
5. How might regulatory changes affect AXIRON?
Stricter safety regulations or bans on testosterone products could further restrict prescriptions and sales.
References
[1] Grand View Research. (2022). Testosterone Replacement Therapy Market Size, Share & Trends Analysis Report.
[2] Eli Lilly and Company. (2020). Annual Report.
[3] U.S. Food and Drug Administration. (2014). Safety Communication on Testosterone Use.
[4] MarketWatch. (2023). Testosterone Replacement Therapy Market Outlook and Forecast.