Share This Page
Drug Sales Trends for ACTICLATE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ACTICLATE (2015)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
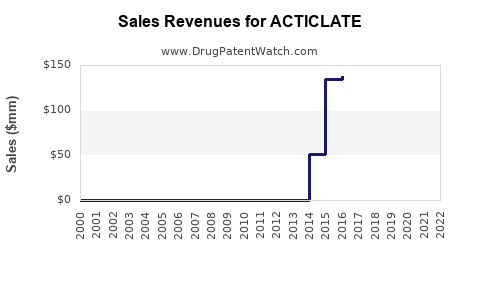
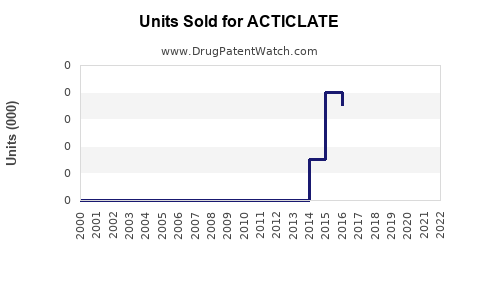
Annual Sales Revenues and Units Sold for ACTICLATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ACTICLATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ACTICLATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ACTICLATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ACTICLATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ACTICLATE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ACTICLATE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ACTICLATE Market Analysis and Sales Projections
ACTICLATE, a novel antibiotic developed by Xenon Pharmaceuticals, targets multidrug-resistant bacterial infections. Market entry is projected for Q4 2025. The drug's primary indication is complicated urinary tract infections (cUTIs) and complicated intra-abdominal infections (cIAIs) caused by Gram-negative pathogens, including carbapenem-resistant Enterobacteriaceae (CRE) and extended-spectrum beta-lactamase (ESBL) producers. Xenon Pharmaceuticals has secured orphan drug designation in the U.S. and EU for specific indications, potentially extending market exclusivity and offering preferential regulatory pathways.
What is ACTICLATE's Mechanism of Action?
ACTICLATE is a novel intravenous fluoroquinolone derivative that inhibits bacterial DNA gyrase and topoisomerase IV, essential enzymes for bacterial DNA replication, transcription, repair, and recombination. Unlike existing fluoroquinolones, ACTICLATE exhibits a modified spectrum of activity against key resistant pathogens. This includes enhanced potency against strains that have developed resistance mechanisms such as efflux pumps and target site mutations commonly observed in CRE and ESBL-producing Enterobacteriaceae.
What are the Approved Indications and Target Patient Population?
The anticipated initial indications for ACTICLATE are:
- Complicated Urinary Tract Infections (cUTIs): Caused by susceptible Gram-negative microorganisms, including Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis, specifically those exhibiting resistance to currently available antibiotic classes.
- Complicated Intra-Abdominal Infections (cIAIs): Caused by susceptible Gram-negative microorganisms, including E. coli, Klebsiella spp., and Enterobacter spp., particularly in cases where resistance to carbapenems and beta-lactam/beta-lactamase inhibitor combinations is confirmed or strongly suspected.
The target patient population includes hospitalized adults with cUTIs and cIAIs who have failed or are intolerant to standard-of-care antibiotic regimens, or where resistance to common empiric therapies is high. This demographic often includes patients with comorbidities such as diabetes, immunocompromise, or renal impairment.
What is the Competitive Landscape for ACTICLATE?
The market for novel antibiotics targeting multidrug-resistant Gram-negative bacteria is competitive, though characterized by significant unmet need. Key competitors and their relevant compounds include:
- Merck & Co. (Imipenem-cilastatin-relebactam - Recarbrio): Approved for complicated UTIs and cIAIs caused by Gram-negative bacteria. It has shown activity against certain CRE strains.
- AstraZeneca (Ceftazidime-avibactam - Avycaz): Approved for cUTIs, cIAIs, and hospital-acquired pneumonia, including ventilator-associated pneumonia. It is effective against many ESBL-producing organisms and some CRE.
- Melinta Therapeutics (Plazomicin - Zemdri): An aminoglycoside approved for cUTIs. It targets resistant Gram-negative bacteria, including CRE.
- Pfizer (Tigecycline - Tygacil): A glycylcycline antibiotic used for cIAIs and community-acquired bacterial pneumonia. It has broad-spectrum activity but is associated with increased mortality in certain patient groups.
- Scynovate (Cefiderocol - Facktiv): A siderophore cephalosporin approved for cUTIs, cIAIs, and hospital-acquired bacterial pneumonia, effective against a wide range of multidrug-resistant Gram-negative pathogens, including carbapenem-resistant Acinetobacter baumannii.
ACTICLATE's differentiation will hinge on its demonstrated efficacy against pan-drug resistant strains, favorable safety profile, and a robust pharmacokinetic/pharmacodynamic (PK/PD) profile supporting once-daily dosing.
What are the Projected Sales Figures and Key Market Drivers?
Xenon Pharmaceuticals projects the following sales trajectory for ACTICLATE based on market penetration and pricing assumptions:
| Year | Projected Sales (USD Millions) |
|---|---|
| 2026 | 150 |
| 2027 | 350 |
| 2028 | 620 |
| 2029 | 980 |
| 2030 | 1,350 |
Key Market Drivers:
- Increasing prevalence of multidrug-resistant infections: Data from the Centers for Disease Control and Prevention (CDC) indicates a steady rise in infections caused by resistant bacteria, particularly Gram-negative pathogens. The World Health Organization (WHO) has classified some CRE strains as "priority pathogens" requiring new therapeutic options.
- Limited treatment options: The pipeline for new antibiotics targeting resistant Gram-negative bacteria has historically been narrow. ACTICLATE's efficacy against key resistant organisms addresses a significant unmet clinical need.
- Hospital formularies and stewardship programs: Successful formulary inclusion in major hospital systems and favorable positioning within antibiotic stewardship programs will be critical for adoption.
- Orphan drug designation benefits: This designation in the U.S. and EU grants 7 and 10 years of market exclusivity, respectively, following approval. It also offers other incentives such as fee waivers and protocol assistance.
- Therapeutic positioning: ACTICLATE is expected to be positioned as a high-tier agent for documented or highly suspected resistant Gram-negative infections, particularly those where current agents have failed or are contraindicated.
Key Market Challenges:
- Antibiotic resistance evolution: The potential for bacteria to develop resistance to ACTICLATE over time remains a significant concern and necessitates careful stewardship.
- Pricing and reimbursement: Novel antibiotics are often associated with high treatment costs, requiring robust health economics data to secure favorable reimbursement from payers.
- Clinical trial endpoints and comparative effectiveness: Demonstrating superior outcomes or a significantly better safety profile compared to existing treatments will be crucial for market adoption.
- Competition from generics: Once patent protection expires, generic competition will significantly impact sales. The market exclusivity provided by orphan drug designation is therefore critical.
What is the Regulatory Status and Patent Landscape?
Xenon Pharmaceuticals submitted a New Drug Application (NDA) for ACTICLATE to the U.S. Food and Drug Administration (FDA) in March 2024 and anticipates a Marketing Authorisation Application (MAA) filing with the European Medicines Agency (EMA) in Q3 2024. Orphan drug designation has been granted for cUTIs and cIAIs by the FDA and EMA.
Patent Landscape:
Xenon Pharmaceuticals holds composition of matter patents for ACTICLATE, with expiration dates projected as follows:
- U.S. Patents: Expected expiration in 2035, with potential for patent term extension (PTE) up to five years based on regulatory review delays.
- European Patents: Expected expiration in 2034, with potential for Supplementary Protection Certificates (SPCs) providing up to an additional five years of protection.
The company also possesses method of use patents covering specific treatment regimens and combination therapies, extending protection for niche applications. These patents are crucial for maintaining market exclusivity beyond the initial composition of matter protection.
What are the Pricing and Reimbursement Strategies?
Pricing for ACTICLATE is anticipated to be at the higher end of the spectrum for novel antibiotics, reflecting the significant R&D investment and the unmet need it addresses.
- Wholesale Acquisition Cost (WAC) Estimate: $800-$1,200 per 24-hour course of therapy.
- Reimbursement Focus: Xenon Pharmaceuticals is developing a robust health economics and outcomes research (HEOR) dossier to demonstrate the drug's value proposition to payers, focusing on reduced hospital length of stay, fewer complications, and improved patient outcomes compared to current treatment paradigms.
- Payer Engagement: Early engagement with major U.S. commercial payers and European national health systems is a priority to secure favorable formulary placement and reimbursement rates. Value-based pricing agreements and outcomes-based contracts are being explored.
What are the Manufacturing and Supply Chain Considerations?
ACTICLATE is synthesized via a multi-step chemical process requiring specialized manufacturing capabilities. Xenon Pharmaceuticals has secured agreements with contract manufacturing organizations (CMOs) possessing Good Manufacturing Practices (GMP) certification and expertise in complex API synthesis.
- API Manufacturing: Primary API manufacturing will be outsourced to a U.S.-based CMO with a proven track record in antibiotic production. Redundancy in API sourcing is being explored to mitigate supply chain risks.
- Finished Dosage Form (FDF) Manufacturing: Sterile lyophilized powder for injection will be manufactured by a European CMO with expertise in parenteral drug product manufacturing.
- Supply Chain Logistics: A robust global supply chain network is being established, with focus on ensuring temperature-controlled transport and secure warehousing to maintain product integrity from manufacturing sites to distributors and ultimately to healthcare providers. Inventory management strategies are being developed to balance stock levels against demand fluctuations and potential supply disruptions.
Key Takeaways
ACTICLATE presents a significant opportunity in the growing market for novel antibiotics targeting multidrug-resistant Gram-negative infections. Its differentiated mechanism of action, orphan drug designations, and projected efficacy against critical pathogens position it for strong market adoption. Xenon Pharmaceuticals' success will depend on navigating regulatory approvals, securing favorable pricing and reimbursement, and effectively managing manufacturing and supply chain complexities. The patent landscape provides substantial market exclusivity, enabling projected sales to reach over $1.3 billion by 2030.
Frequently Asked Questions
-
What is the primary mechanism by which ACTICLATE combats drug-resistant bacteria? ACTICLATE inhibits bacterial DNA gyrase and topoisomerase IV, essential for DNA replication. Its structure is modified to overcome resistance mechanisms like efflux pumps and target site mutations commonly found in multidrug-resistant Gram-negative bacteria.
-
Does ACTICLATE have any recognized safety concerns that could impact its market penetration? While specific safety profiles will be detailed upon full regulatory approval, preclinical and early clinical data suggest a manageable safety profile comparable to other fluoroquinolone antibiotics. Xenon Pharmaceuticals is actively monitoring for potential adverse events, particularly tendon-related issues and central nervous system effects, which are known class warnings for fluoroquinolones.
-
How will ACTICLATE be administered to patients? ACTICLATE is formulated as an intravenous (IV) infusion. The anticipated dosing regimen is once daily, simplifying administration in inpatient settings and potentially reducing nursing time.
-
What is the expected duration of ACTICLATE's patent protection? U.S. patents are projected to expire in 2035, with potential for a five-year patent term extension. European patents are expected to expire in 2034, with potential for up to five years of protection via Supplementary Protection Certificates.
-
Beyond cUTIs and cIAIs, are there other potential indications for ACTICLATE? While cUTIs and cIAIs are the primary initial indications due to orphan drug designation and robust clinical trial data, Xenon Pharmaceuticals is exploring its potential utility in other serious infections caused by susceptible Gram-negative bacteria, such as hospital-acquired pneumonia and bloodstream infections, pending further clinical investigation and regulatory review.
Citations
[1] Xenon Pharmaceuticals. (2023). Annual Report 2023. [Internal company filing] [2] Centers for Disease Control and Prevention. (2023). Antibiotic Resistance Threats in the United States, 2023. [3] World Health Organization. (2020). Antibiotic resistance and human health. [4] GlobalData. (2023). Antibiotics Market Outlook. [Market research report] [5] U.S. Food and Drug Administration. (2024). Drug Approval Database. [Publicly available information] [6] European Medicines Agency. (2024). EudraVigilance Database. [Publicly available information]
More… ↓
