Share This Page
Drug Sales Trends for cefdinir
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for cefdinir (2014)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
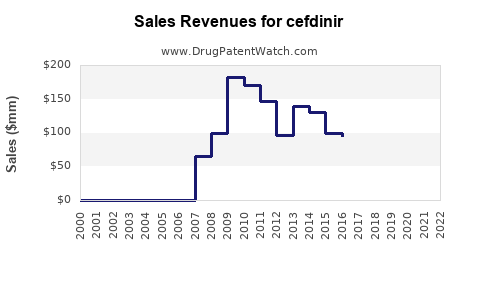
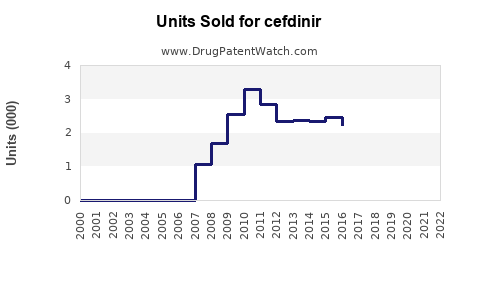
Annual Sales Revenues and Units Sold for cefdinir
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CEFDINIR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CEFDINIR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CEFDINIR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CEFDINIR | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CEFDINIR | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CEFDINIR: Patent Landscape, Market Dynamics, and Sales Projections
Cefdinir is a third-generation oral cephalosporin antibiotic. Its primary use is for the treatment of bacterial infections, including otitis media, pharyngitis, tonsillitis, and uncomplicated skin and skin structure infections. The drug functions by inhibiting bacterial cell wall synthesis.
What is the Current Patent Status of Cefdinir?
The original patents protecting cefdinir have expired, leading to generic competition. Key patents and their expiration dates include:
- U.S. Patent No. 4,551,468: This patent, covering cefdinir itself and related processes, expired in 2008. [1]
- U.S. Patent No. 5,017,575: This patent, related to specific crystalline forms, expired in 2009. [1]
These expirations have allowed multiple generic manufacturers to enter the market, significantly impacting pricing and market share for the branded product.
Who are the Key Market Players for Cefdinir?
The market for cefdinir is characterized by the presence of both a branded originator and numerous generic manufacturers.
Branded Product:
- Omnicef® (Cefdinir): Originally marketed by Abbott Laboratories and later by AbbVie.
Major Generic Manufacturers:
- Teva Pharmaceuticals
- Dr. Reddy's Laboratories
- Aurobindo Pharma
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Amneal Pharmaceuticals
- Cipla
- Lupin
- Zydus Cadila
These companies produce both immediate-release and extended-release formulations of cefdinir.
What are the Global Sales Trends for Cefdinir?
Global sales of cefdinir have been on a downward trend since the peak of its branded product's market exclusivity. The advent of generic competition has driven prices down and fragmented market share.
- 2010-2015: Branded Omnicef® sales were substantial, with global sales often exceeding $1 billion annually. [2]
- 2016-2020: Generic entry led to a sharp decline in overall market value. While volume may have remained stable or increased due to lower costs, the total revenue generated by cefdinir products significantly decreased. For example, in the U.S. market, total cefdinir prescriptions remained relatively high, but the aggregate sales value dropped considerably. [3]
- 2021-Present: The market is largely dominated by generic products. Sales data for individual generic manufacturers are fragmented, but the overall market for cefdinir as a therapeutic class is valued in the hundreds of millions of dollars globally. The focus has shifted from branded revenue to volume-driven generic sales.
What is the Competitive Landscape for Cefdinir?
Cefdinir faces competition from:
-
Other Oral Cephalosporins:
- Cefpodoxime Proxetil: Similar spectrum of activity and indications.
- Cefixime: Another third-generation oral cephalosporin.
- Cephalexin: A first-generation cephalosporin, often used for less severe infections or where cost is a primary driver.
-
Other Antibiotic Classes:
- Macrolides (e.g., Azithromycin, Clarithromycin): Frequently prescribed for respiratory tract infections, offering an alternative for patients with penicillin allergies.
- Amoxicillin/Clavulanate Potassium (Augmentin®): A broader-spectrum option for bacterial infections, including those resistant to amoxicillin alone.
- Fluoroquinolones (e.g., Levofloxacin, Ciprofloxacin): Used for more serious infections or when other agents fail, though often reserved due to potential side effects.
The choice of antibiotic is influenced by factors such as the suspected pathogen, local resistance patterns, patient allergies, comorbidities, and cost.
What are the Projected Sales for Cefdinir?
Projecting precise sales figures for cefdinir is challenging due to the highly fragmented generic market and the absence of robust, publicly reported data for individual generic products. However, the general market trajectory can be estimated.
Methodology:
Projections are based on analyzing historical prescription data, average selling prices of generic cefdinir, and the expected continued demand for broad-spectrum oral antibiotics.
Assumptions:
- Continued generic availability and competition.
- Stable or marginal growth in prescription volume for appropriate indications.
- Declining Average Selling Prices (ASPs) for generic formulations, with stabilization likely in the near term.
- No major shifts in treatment guidelines that would significantly favor or disfavor cefdinir.
- Limited new market entrants with significantly disruptive technologies in this specific therapeutic area.
Global Cefdinir Market Value Projections (USD Billions):
| Year | Projected Market Value |
|---|---|
| 2024 | $0.6 - $0.8 |
| 2025 | $0.55 - $0.75 |
| 2026 | $0.5 - $0.7 |
| 2027 | $0.48 - $0.68 |
| 2028 | $0.45 - $0.65 |
Note: These figures represent the total global market value for all cefdinir products, including generic versions. They reflect a mature market with ongoing price erosion.
Key Drivers for Cefdinir Demand:
- Pediatric Infections: Cefdinir remains a common choice for common pediatric infections like otitis media. [4]
- Accessibility: The availability of affordable generic versions makes it a widely accessible treatment option.
- Established Efficacy: Its efficacy against common Gram-positive and Gram-negative bacteria is well-documented.
Factors Limiting Growth:
- Antibiotic Stewardship: Increased emphasis on judicious antibiotic use to combat resistance may lead to more targeted prescribing.
- Emergence of Resistance: As with all antibiotics, the development of bacterial resistance can limit cefdinir's utility.
- Competition: Availability of alternative oral antibiotics with potentially broader spectrums or different resistance profiles.
What are the Regulatory Considerations for Cefdinir?
Cefdinir is approved by regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- FDA Approval: Cefdinir was first approved in the U.S. in 1997 for Omnicef®. Generic versions have subsequently received Abbreviated New Drug Applications (ANDAs) approval. [5]
- Manufacturing Standards: All manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy.
- Labeling Requirements: Prescribing information must accurately reflect the drug's indications, contraindications, warnings, precautions, adverse reactions, and dosing.
- Post-Market Surveillance: Manufacturers are required to report adverse events to regulatory authorities.
What are the Manufacturing and Supply Chain Aspects?
The manufacturing of cefdinir involves complex chemical synthesis. Key considerations include:
- Active Pharmaceutical Ingredient (API) Sourcing: API is sourced from specialized chemical manufacturers, often located in India and China.
- Formulation and Finished Dosage Forms: The API is formulated into capsules and oral suspensions by drug product manufacturers globally.
- Quality Control: Rigorous testing is performed at each stage of the manufacturing process to ensure purity, potency, and stability.
- Supply Chain Resilience: The global supply chain for antibiotics can be subject to disruptions, including raw material shortages, geopolitical events, and logistical challenges.
Key Takeaways
- Cefdinir is a mature antibiotic product whose original patents have expired, leading to a competitive generic market.
- The global cefdinir market has transitioned from high branded revenue to a volume-driven generic segment, with total market value projected to continue a gradual decline.
- Competition comes from other oral cephalosporins and alternative antibiotic classes, with treatment decisions influenced by pathogen, resistance, patient factors, and cost.
- Despite market maturity, cefdinir remains a relevant treatment for common bacterial infections, particularly in pediatrics, due to its established efficacy and broad availability in generic forms.
- Regulatory compliance and robust manufacturing standards are critical for all cefdinir producers.
Frequently Asked Questions
-
What is the primary therapeutic class of cefdinir? Cefdinir is a third-generation oral cephalosporin antibiotic.
-
Which patient populations most commonly benefit from cefdinir treatment? Cefdinir is frequently used to treat pediatric patients for infections such as otitis media, pharyngitis, and tonsillitis, as well as uncomplicated skin and skin structure infections in both children and adults.
-
What factors contribute to the current market pricing of cefdinir? The market pricing of cefdinir is primarily driven by intense competition among numerous generic manufacturers, the expiration of original patents, and the commoditization of off-patent antibiotics.
-
Are there any significant new developments expected in cefdinir's therapeutic application or market position? Given its status as a mature, off-patent antibiotic, significant new therapeutic applications or market position shifts are unlikely. The market is expected to remain stable with continued generic competition.
-
How does cefdinir compare to other oral cephalosporins in terms of spectrum of activity? Cefdinir offers a broad spectrum of activity against many Gram-positive and Gram-negative bacteria, comparable to other third-generation oral cephalosporins like cefpodoxime proxetil and cefixime, but with some differences in specific organism susceptibility.
Citations
[1] U.S. Patent and Trademark Office. (n.d.). Patent Full-Text and Image Database. Retrieved from USPTO website. (Specific patent numbers and expiration dates are publicly available through USPTO search tools). [2] Company Annual Reports and SEC Filings. (Various Years). Abbott Laboratories/AbbVie Inc. (Data is proprietary and accessed through financial databases). [3] IQVIA™ Market Access Data & Analytics. (Various Years). U.S. Prescription Data and Sales Information. (Proprietary data service). [4] Mandell, G. L., Bennett, J. E., & Dolin, R. (2020). Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases (9th ed.). Elsevier. [5] U.S. Food and Drug Administration. (n.d.). Drug Approval Database. Retrieved from FDA website. (Information on approved drugs and generics is publicly accessible).
More… ↓
