Share This Page
Drug Sales Trends for TAMIFLU
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TAMIFLU (2014)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
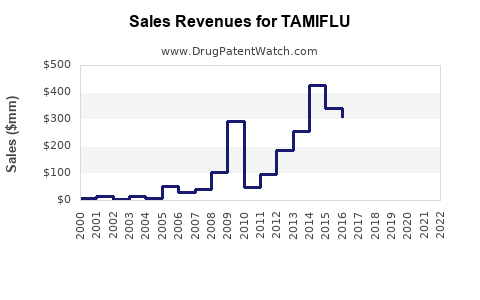
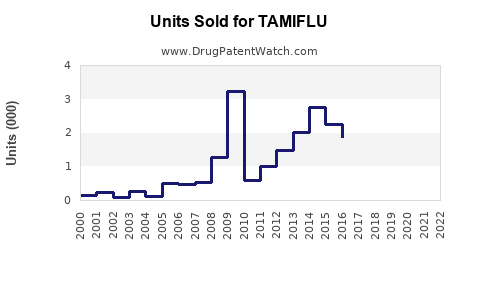
Annual Sales Revenues and Units Sold for TAMIFLU
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TAMIFLU | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TAMIFLU | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TAMIFLU | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TAMIFLU | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TAMIFLU | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Tamiflu: Patent Expiration and Market Landscape Analysis
Tamiflu (oseltamivir phosphate) is an antiviral medication approved for the treatment and prevention of influenza A and B. Its primary mechanism of action is the inhibition of neuraminidase, an enzyme essential for viral replication and release from infected cells. The drug's market has historically been influenced by seasonal flu outbreaks, pandemic preparedness strategies, and the expiration of key patents.
What is the Current Patent Status of Tamiflu?
The core patent for oseltamivir phosphate, held by Gilead Sciences and licensed to Roche, expired in major markets globally by 2017. This expiration opened the door for generic competition. For example, U.S. Patent 4,732,970, covering the compound itself, expired on August 31, 2002, with pediatric exclusivity extending its protection until February 2003. Further patents related to specific formulations or methods of use have also expired or are nearing expiration [1, 2]. The lapsation of these primary patents signifies the end of market exclusivity for Roche's branded Tamiflu, allowing other pharmaceutical companies to develop and market generic versions of oseltamivir phosphate.
What is the Global Market Size for Influenza Antivirals?
The global market for influenza antivirals is substantial and fluctuates based on several factors, including the severity of flu seasons, public health recommendations, and the availability of vaccines and treatments. In 2022, the global influenza antiviral market was valued at approximately USD 2.8 billion. Projections indicate a Compound Annual Growth Rate (CAGR) of around 5.5% from 2023 to 2030, potentially reaching USD 4.3 billion by 2030 [3]. This growth is driven by increased awareness of influenza's public health impact, government stockpiling initiatives for pandemic preparedness, and the ongoing development of new antiviral therapies.
Key Market Drivers and Restraints:
- Drivers:
- Seasonal Influenza Incidence: Higher rates of seasonal flu directly increase demand for antiviral treatments.
- Pandemic Preparedness: Governments maintain stockpiles of antivirals like oseltamivir to counter potential influenza pandemics, creating a consistent demand.
- Growing Geriatric Population: Older adults are more susceptible to influenza complications, leading to higher treatment rates.
- Increasing Healthcare Expenditure: Global investments in healthcare infrastructure and access to medicines contribute to market expansion.
- Restraints:
- Vaccine Efficacy and Uptake: Effective seasonal influenza vaccines can reduce the overall incidence of the flu, thereby lowering the demand for antiviral treatments.
- Emergence of Drug-Resistant Strains: The development of influenza strains resistant to existing antivirals poses a significant threat and can limit the efficacy of current treatments.
- Generic Competition: The expiration of patents for major antivirals like Tamiflu leads to price erosion due to the introduction of lower-cost generic alternatives, impacting revenue for branded products.
- Side Effects and Adverse Events: Concerns regarding the side effects of antiviral medications can influence prescribing patterns and patient adherence.
Who Are the Major Players in the Influenza Antiviral Market?
The influenza antiviral market comprises both originator companies and generic manufacturers. Since patent expirations, the competitive landscape has broadened considerably.
Key Companies and Their Contributions:
- Roche: The originator of Tamiflu, Roche continues to market the branded product. Its market share has been significantly impacted by generic competition.
- Generic Manufacturers: Numerous companies now produce generic oseltamivir phosphate. Notable players include:
- Dr. Reddy's Laboratories: A significant producer of generic oseltamivir in various markets.
- Teva Pharmaceutical Industries: A major global generic drug manufacturer with oseltamivir in its portfolio.
- Mylan N.V. (now Viatris): Another large generic pharmaceutical company offering oseltamivir.
- Sun Pharmaceutical Industries: A significant Indian multinational pharmaceutical company with generic antiviral offerings.
- Zydus Cadila: An Indian pharmaceutical company that manufactures and markets generic oseltamivir.
- Other Antivirals: Companies developing or marketing alternative influenza antivirals also compete, including those with neuraminidase inhibitors like peramivir and zanamivir, as well as cap-dependent endonuclease inhibitors like baloxavir marboxil [4].
What Are the Sales Projections for Oseltamivir Phosphate?
Following patent expirations, the sales trajectory for branded Tamiflu has declined, while the overall market for oseltamivir phosphate, encompassing both branded and generic versions, is expected to grow due to increased accessibility and broader market penetration.
Historical Sales Trends:
- Pre-Patent Expiration: Branded Tamiflu achieved peak sales driven by its market exclusivity and significant pandemic preparedness orders. For instance, in 2009, during the H1N1 pandemic, Tamiflu sales surged significantly.
- Post-Patent Expiration: Sales of branded Tamiflu have experienced a downward trend as generic equivalents entered the market. However, the aggregate sales of oseltamivir phosphate (including generics) have maintained a steady presence in the market.
Projected Market Performance (Oseltamivir Phosphate - Branded and Generic Combined):
- The global market for oseltamivir phosphate is projected to maintain a stable growth rate, driven by its established efficacy, ongoing seasonal demand, and continued use in government stockpiles.
- Estimates suggest the oseltamivir phosphate market, inclusive of all manufacturers, will continue to occupy a significant portion of the influenza antiviral market, potentially contributing USD 1.5 billion to USD 2 billion annually over the next five to seven years, depending on flu season severity and competitor market penetration [3, 5].
- The market value is increasingly distributed among generic manufacturers, with price competition being a dominant factor.
What is the Competitive Landscape for Influenza Antivirals?
The competitive landscape is characterized by the presence of multiple antiviral agents targeting different stages of the influenza virus lifecycle.
Key Competitors and Therapies:
- Neuraminidase Inhibitors:
- Oseltamivir Phosphate (Tamiflu and generics): Remains a first-line treatment and prophylaxis option.
- Zanamivir (Relenza): An inhaled neuraminidase inhibitor, less frequently used due to administration route and potential side effects.
- Peramivir (Rapivab): An intravenous neuraminidase inhibitor used for severe cases.
- Cap-Dependent Endonuclease Inhibitors:
- Baloxavir Marboxil (Xofluza): A newer class of antivirals that targets a different viral enzyme, offering a single-dose treatment option and demonstrating efficacy against resistant strains [4, 6]. Xofluza has captured a growing share of the market since its introduction.
- Adamantanes:
- Amantadine and Rimantadine: Largely rendered ineffective due to widespread resistance in circulating influenza strains and are no longer recommended for routine treatment or prophylaxis [7].
The introduction of baloxavir marboxil represents a significant competitive development, offering a differentiated mechanism of action and a convenient dosing regimen. This has led to increased market segmentation and requires ongoing assessment of treatment guidelines and physician preferences.
What are the Regulatory and Policy Considerations?
Regulatory approvals and public health policies play a crucial role in the market dynamics of influenza antivirals.
Key Policy and Regulatory Aspects:
- FDA and EMA Approvals: Oseltamivir phosphate has received approvals from regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for specific indications related to influenza A and B [1].
- Seasonal Flu Vaccine Recommendations: Annual recommendations for influenza vaccination by public health organizations (e.g., CDC, WHO) influence the overall incidence of influenza, thereby impacting the demand for antiviral treatments.
- Strategic National Stockpile (SNS) in the U.S.: The U.S. government maintains a significant stockpile of oseltamivir phosphate for pandemic preparedness. Purchases for these stockpiles contribute to consistent demand, though they are subject to review and renewal cycles [8].
- Antiviral Resistance Monitoring: Global surveillance programs monitor influenza virus susceptibility to antiviral drugs. The emergence and spread of resistance can impact the recommended use of specific antivirals and drive the development of new therapeutic agents [7].
- Generic Drug Regulations: Regulatory pathways for generic drug approval ensure bioequivalence and therapeutic interchangeability with the innovator product, facilitating market entry and price competition.
What Are the Future Market Trends and Opportunities?
The influenza antiviral market is dynamic, with ongoing research and development aiming to address unmet needs and enhance treatment efficacy.
Key Trends and Opportunities:
- Development of Novel Antivirals: Research continues into antivirals with new mechanisms of action to overcome drug resistance and improve safety profiles. This includes exploring broad-spectrum antivirals effective against multiple respiratory viruses.
- Combination Therapies: Investigating the efficacy of combining existing antivirals with different mechanisms of action or combining antivirals with other therapeutic agents to enhance treatment outcomes and prevent resistance.
- Improved Diagnostic Tools: Advances in rapid influenza diagnostic tests can facilitate earlier and more accurate diagnosis, enabling prompt initiation of antiviral therapy.
- Personalized Medicine Approaches: Tailoring antiviral treatment based on individual patient factors, such as strain susceptibility, comorbidities, and genetic predispositions.
- Addressing Global Health Disparities: Ensuring access to affordable and effective antiviral treatments in low- and middle-income countries remains a critical area for intervention and market expansion.
- Pandemic Preparedness Evolution: Governments and international health organizations are continuously evaluating and updating pandemic preparedness strategies, which may lead to shifts in procurement patterns and the types of antivirals prioritized for stockpiling.
Key Takeaways
- Tamiflu's foundational patents have expired, leading to widespread generic availability of oseltamivir phosphate and significant price erosion for the branded product.
- The global influenza antiviral market is projected to grow, driven by seasonal flu, pandemic preparedness, and an aging population.
- The market is increasingly competitive, with generic manufacturers of oseltamivir phosphate and newer antivirals like baloxavir marboxil vying for market share.
- Government stockpiling for pandemic preparedness represents a significant and stable demand driver for oseltamivir phosphate.
- Emerging antiviral resistance and the development of novel therapies are key factors shaping the future of the influenza antiviral market.
Frequently Asked Questions
1. How has the expiration of Tamiflu patents affected its price?
The expiration of key patents for oseltamivir phosphate has led to the entry of numerous generic manufacturers, resulting in substantial price reductions for oseltamivir phosphate compared to the original branded Tamiflu.
2. What is the current recommended treatment duration for oseltamivir phosphate?
For treatment of seasonal influenza in adults and children, the recommended duration of oseltamivir phosphate is typically five days of twice-daily dosing. For post-exposure prophylaxis, the recommended duration is usually 10 days of once-daily dosing [1].
3. Are there any significant concerns regarding drug resistance to oseltamivir phosphate?
Yes, the emergence and spread of influenza strains resistant to neuraminidase inhibitors, including oseltamivir phosphate, is a recognized concern. Viral surveillance programs continuously monitor for resistance patterns, which can influence treatment recommendations [7].
4. How does baloxavir marboxil (Xofluza) compare to oseltamivir phosphate in treating influenza?
Baloxavir marboxil is a first-in-class cap-dependent endonuclease inhibitor, offering a single-dose treatment option, whereas oseltamivir phosphate is a neuraminidase inhibitor typically administered over five days. Xofluza has demonstrated efficacy against oseltamivir-resistant strains [4, 6].
5. What is the role of oseltamivir phosphate in pandemic preparedness?
Oseltamivir phosphate has been a cornerstone of pandemic preparedness strategies worldwide, with governments maintaining substantial stockpiles to rapidly respond to influenza pandemics due to its established efficacy and broad applicability against various influenza strains [8].
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Information: Tamiflu. Retrieved from https://www.fda.gov/ (Note: Specific approval dates and patent expiry details can be found through FDA's drug database searches).
[2] Gilead Sciences, Inc. (n.d.). Annual Reports and SEC Filings. Retrieved from https://investors.gilead.com/ (Note: Historical patent information is often detailed in SEC filings).
[3] Grand View Research. (2023). Influenza Antivirals Market Size, Share & Trends Analysis Report By Drug Class (Neuraminidase Inhibitors, Polymerase Inhibitors), By Type (Branded, Generic), By Application, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030.
[4] Hayden, F. G. (2018). Antiviral therapy for influenza. Seminars in Respiratory and Critical Care Medicine, 39(4), 445–451.
[5] MarketsandMarkets. (2023). Influenza Therapeutics Market - Global Forecast to 2028.
[6] Jefferson, T., Smith, S. M., Demicheli, V., Rivetti, A., Di Pietrantonj, C., & Wee, H. L. (2020). Antivirals for the treatment of influenza in adults and children (including the elderly) – systematic review and meta-analysis. Cochrane Database of Systematic Reviews(8).
[7] World Health Organization. (2023). Antiviral resistance in influenza viruses. Retrieved from https://www.who.int/
[8] U.S. Department of Health and Human Services. Assistant Secretary for Preparedness and Response. (n.d.). Strategic National Stockpile. Retrieved from https://www.phe.gov/Preparedness/stockpile/Pages/default.aspx
More… ↓
