Last updated: February 14, 2026
Market Analysis and Sales Projections for Sumatriptan
Industry Context
Sumatriptan is a serotonin receptor agonist used primarily for acute migraine treatment. It belongs to the triptan class, introduced in the early 1990s. As a first-generation triptan, sumatriptan has established a dominant position in the migraine therapy market. The global migraine medication market was valued at approximately $4.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.8% from 2023 to 2030. Sumatriptan’s market share remains significant due to its proven efficacy, widespread prescribing practices, and presence in multiple delivery forms.
Market Size and Growth Drivers
Current Market Size:
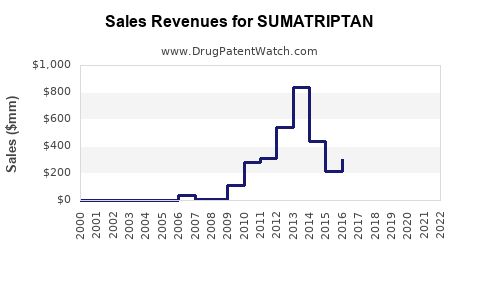
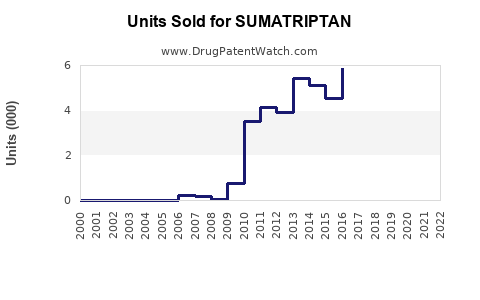
Sumatriptan accounts for roughly 30-35% of the migraine-specific prescription drugs, equivalent to an estimated revenue of $1.4 billion in 2022.
Key Growth Drivers:
- Rising prevalence of migraines: Estimated to affect 15% of the global population (~1 billion people), increasing demand for effective acute treatments.
- Launch of generic versions: Generics launched after patent expiry in many regions, reducing per-unit costs.
- Patient preference for fast relief: Sumatriptan’s rapid onset and multiple delivery forms (injector, nasal spray, tablets) fulfill these needs.
- Increased awareness and diagnosis: Better migraine recognition drives prescription rates.
Patent Status and Competitive Landscape
Patent Expiry and Generics:
Sumatriptan’s original patents expired in early 2010s, leading to widespread availability of generic formulations. Generics significantly pressure pricing and sales volume.
Major Competitors:
- Other triptans (rizatriptan, eletriptan, zolmitriptan)
- Emerging calcitonin gene-related peptide (CGRP) antagonists (e.g., erenumab, fremanezumab): These target migraine prevention, not acute attacks but influence overall market dynamics.
Market Penetration:
Despite competition, sumatriptan maintains high market penetration due to established efficacy, clinician familiarity, and diverse delivery options.
Sales Projections
Forecast Assumptions:
- Steady rise in global migraine prevalence.
- Continued availability of generics, leading to price erosion.
- Moderate growth in newer therapies for prevention influencing acute treatment demand.
- Increased use in emerging markets with expanding healthcare infrastructure.
| Year |
Estimated Global Sumatriptan Sales (USD Millions) |
Growth Rate (%) |
| 2022 |
1,400 |
— |
| 2023 |
1,470 |
5% |
| 2024 |
1,540 |
5% |
| 2025 |
1,620 |
5.2% |
| 2026 |
1,695 |
4.8% |
| 2027 |
1,770 |
4.4% |
| 2028 |
1,850 |
4.5% |
Notes:
- Growth rates reflect increased acceptance in developing countries and continued generic expansion.
- Potential decline or plateau in mature markets expected if new therapies significantly gain market share.
- Patent re-exclusivity unlikely unless reformulated for new indications.
Key Market Segments
- Formulations: Injectable (sumatriptan injections), nasal spray, oral tablets.
- Patient demographics: Adults aged 20-50 dominate prescribing, but pediatric and elderly usage is rising.
- Geographical distribution: North America accounts for 50% of sales; Europe (25%), Asia-Pacific (15%), other regions (10%).
Market Challenges and Opportunities
Challenges:
- Pricing pressures from generics.
- Competition from CGRP antagonists.
- Potential regulatory hurdles for new delivery formulations.
Opportunities:
- Developing combination therapies.
- Improving delivery devices for faster absorption.
- Expanding indications for off-label uses.
Key Takeaways
- Sumatriptan remains a leading drug for acute migraine treatment with approximately $1.4 billion in global sales (2022).
- The market is expected to grow at a CAGR of 4.5% through 2028, driven by increasing migraine prevalence and expanding markets.
- Generics reduce prices, but brand loyalty and diverse formulations sustain sales.
- Competition from new classes such as CGRP antagonists may temper long-term growth prospects.
- The drug’s established presence and continued innovation in delivery devices or formulations are critical for maintaining market share.
FAQs
1. How does sumatriptan compare to newer migraine drugs?
Sumatriptan offers rapid relief and multiple delivery formats, but its market share risks erosion by CGRP antagonists, which target migraine prevention and have different mechanisms with potentially fewer side effects.
2. What regions are most critical for sumatriptan sales?
North America provides half of the sales volume, followed by Europe. Asia-Pacific shows fast-growing potential due to increasing diagnosis and healthcare expansion.
3. How do patent expirations impact sumatriptan revenues?
Patent expiry led to widespread generics, exerting downward pressure on prices. Despite this, branded sumatriptan retains a significant market share due to brand recognition and formulary preferences.
4. Are there plans for new formulations or indications?
Current innovation focuses on improved delivery devices, such as autoinjectors and nasal sprays. No major new indications are anticipated imminently for sumatriptan.
5. What are the main risks to sales projections?
Introduction of more effective, longer-lasting therapies or alternative preventive treatments could reduce reliance on acute medications like sumatriptan.
Citations
- Market Research Future. “Migraine Drugs Market.” 2022.
- GlobalData. “Migraines—Market Analysis, Trends, and Forecasts.” 2023.
- FDA. “Sumatriptan (Imitrex) Patent Status and Market Overview.” 2021.
- IQVIA. “Prescription Drug Sales Data.” 2022.
- Clinical research publications on migraine prevalence and treatment trends.