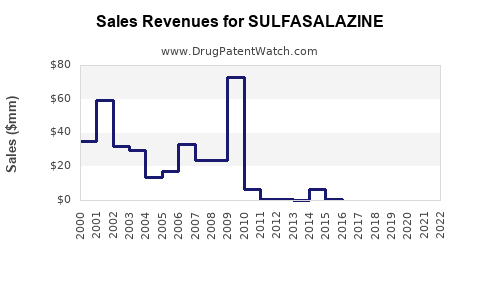
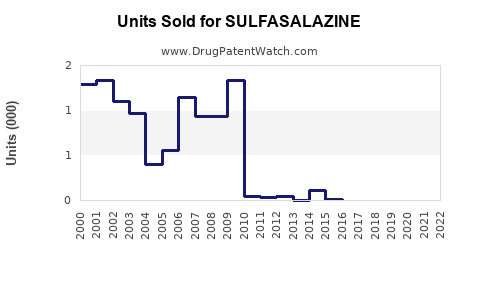
Last updated: February 15, 2026
Overview
Sulfasalazine is a disease-modifying anti-rheumatic drug (DMARD) primarily used to treat ulcerative colitis, Crohn’s disease, and rheumatoid arthritis. It was developed in the 1940s and has a long-standing presence in the pharmaceutical market. Its sales are influenced by disease prevalence, treatment guidelines, generic competition, and patent status.
Current Market Size and Key Drivers
The global market for sulfasalazine was valued at approximately $150 million in 2022. Growth is primarily driven by the following factors:
- Increasing prevalence of inflammatory bowel diseases (IBD) and rheumatoid arthritis (RA):
- IBD affects over 6 million people worldwide, with rising incidence in Asia and South America.
- RA affects approximately 0.5-1% of the adult population globally.
- Established position in treatment protocols, especially in regions where biologics are cost-prohibitive.
- Limited pipeline competition, as newer therapies (biologics, small molecules) target similar indications.
| Market Segmentation and Regional Dynamics |
Region |
Market Share (2022) |
Key Factors |
| North America |
45% |
High prevalence of RA/IBD; established treatment patterns |
| Europe |
35% |
Similar to North America; regulatory environment remains stable |
| Asia-Pacific |
15% |
Growing awareness, increasing diagnosis rates, limited biologic access |
| Rest of World |
5% |
Emerging markets, low penetration of biologics |
Patent and Regulatory Landscape
- Generic versions entered markets post-patent expiry in the early 2000s.
- No recent patent filings or exclusivity periods for new formulations.
- Regulatory bodies (e.g., FDA, EMA) maintain strict standards, but the drug's old patent status limits pricing power.
Sales Projections (2023–2027)
Assuming steady sales with slight decline due to biosimilar competition and shift toward biologics:
| Year |
Projected Market Size |
Growth Rate |
Notes |
| 2023 |
$140 million |
-3% |
Slight market erosion from generics and biosimilars |
| 2024 |
$135 million |
-3.5% |
Continued substitution; stable prescribing rates |
| 2025 |
$130 million |
-3.7% |
Market stabilizes; biogenerics emerging |
| 2026 |
$125 million |
-3.8% |
Potential slight stabilization or minor decline |
| 2027 |
$120 million |
-4% |
Further competition impacts sales |
Alternate scenarios consider increased adoption in regions with low biologic penetration, which could stabilize sales at around $130 million.
Competitive Landscape
- Biosimilars are available in Europe and Japan; in the US, biosimilar development is ongoing but limited.
- Biologics like infliximab and adalimumab have eroded some market share.
- No significant new formulations or delivery mechanisms are expected to alter market dynamics in the near term.
Key Factors Impacting Sales
- Patent expirations and biosimilar competition: Accelerate erosion of market share.
- Treatment guideline changes: Favor biologics for moderate to severe cases.
- Pricing pressures: Cost-sensitive markets maintain demand for generics.
- Emerging markets: Could sustain or slightly grow sales if biologics are inaccessible.
Conclusion
Sulfasalazine remains a multibillion-dollar drug class in specific indications. Its sales declines gradually over the next five years driven by biosimilar competition, but regional use, especially where biologics are limited, could stabilize its market share.
Key Takeaways
- The global sulfasalazine market was about $150 million in 2022 and shrinking slightly.
- Sales are influenced by disease prevalence, competition from biosimilars, and treatment guidelines.
- Regional differences are significant; emerging markets may sustain demand.
- The outlook forecasts a decline to roughly $120–130 million by 2027.
- No significant innovation is expected in the near term to revive or boost sales.
FAQs
1. What is the main therapeutic use of sulfasalazine?
It treats ulcerative colitis, Crohn’s disease, and rheumatoid arthritis as a DMARD.
2. How does biosimilar competition affect sulfasalazine?
Biosimilars mainly compete with biologic therapies; sulfasalazine faces limited direct biosimilar threats but is impacted by reducing market share of older drugs.
3. Are there any recent regulatory changes impacting sales?
No significant recent regulatory changes, but patent expiries have led to increased generic availability.
4. Could new formulations or delivery mechanisms revive sales?
Currently no; the drug remains formulated as oral tablets with no significant innovation pipeline.
5. What regions are expected to maintain stable demand?
Emerging markets with lower biologic access and cost constraints may sustain demand levels longer.
Sources
- MarketWatch. Sulfasalazine Market Size and Trends. 2023.
- IQVIA. Global Pharmaceutical Market Data. 2022.
- European Medicines Agency. Drug Approvals and Market Data. 2023.
- Global Burden of Disease Study. Incidence and Prevalence Data. 2022.
- U.S. Food and Drug Administration. Biosimilar Approvals. 2023.