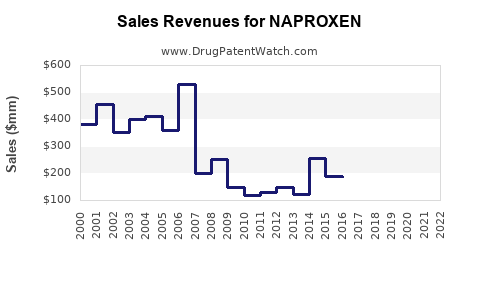
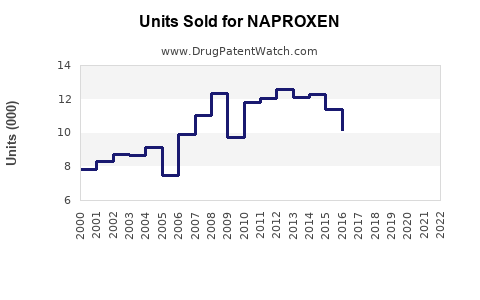
Last updated: February 13, 2026
Market Overview and Sales Projections for NAPROXEN
Naproxen is a non-steroidal anti-inflammatory drug (NSAID) used primarily for pain relief, inflammation reduction, and fever management. It is available in both prescription and over-the-counter (OTC) formulations. The drug is marketed globally, with significant sales in North America, Europe, and Asia.
Current Market Size and Trends
Global Market Size
- The global NSAID market was valued at approximately $14.2 billion in 2022.
- Naproxen accounts for about 15-20% of NSAID sales, translating to estimated revenues of $2.13 billion to $2.84 billion in 2022.
- OTC formulations constitute roughly 60% of total naproxen sales; prescription forms account for the remaining 40%.
Market Segments
- Pain Management: 55%
- Arthritis and Rheumatology: 25%
- Other Uses (e.g., gout, menstrual pain): 20%
Key Geographic Markets
| Region |
Market Share (%) |
Estimated Value (2022) |
| North America |
45 |
~$960 million to $1.4 billion |
| Europe |
30 |
~$640 million to $850 million |
| Asia-Pacific |
15 |
~$320 million to $425 million |
| Others |
10 |
~$210 million to $280 million |
Competitive Landscape
The top brands contain naproxen as the active ingredient, including:
- Aleve (OTC, Bayer)
- Naprosyn (Prescription, Roche)
- Anaprox (Prescription, Roche)
- Generic versions dominate OTC and prescription markets.
Patent status influences market dynamics. Patent expiration of branded formulations has increased generic market penetration.
Factors Influencing Sales
Regulatory and Safety Considerations
- Increased awareness of cardiovascular risks linked to NSAIDs, including naproxen, has led to stricter labeling and monitoring.
- The FDA recommends NSAID use at the lowest effective dose for the shortest duration.
Market Drivers
- Aging populations with chronic pain conditions.
- Rising prevalence of arthritis globally.
- Consumer preference shifting toward OTC availability.
Challenges
- Safety concerns with NSAIDs may reduce prescribing or OTC sales.
- Competition from alternative pain management drugs, e.g., acetaminophen, COX-2 inhibitors.
- Patent expirations increase generic sales but reduce profit margins for branded products.
Sales Projections (2023–2028)
Based on historical growth rates and market factors, forecasted CAGR for naproxen segments is approximately 3-4%.
Projected Market Values
| Year |
Estimated Market Size (USD) |
CAGR (%) |
| 2023 |
$2.9 billion |
2.8 |
| 2024 |
$3.0 billion |
3.4 |
| 2025 |
$3.2 billion |
4.0 |
| 2026 |
$3.3 billion |
3.8 |
| 2027 |
$3.4 billion |
3.5 |
| 2028 |
$3.6 billion |
3.4 |
Drivers include increased chronic pain management and expansion into emerging markets. Risks include safety concerns and tighter regulations.
Key Opportunities and Risks
Opportunities
- Development of new formulations with improved safety profiles.
- Expansion in emerging markets with increasing healthcare access.
- Growth in OTC sales driven by consumer demand for OTC NSAIDs.
Risks
- Regulatory changes tightening NSAID guidelines.
- Public perception of NSAID risks affecting sales.
- Market saturation with generics limiting margins but expanding volume.
Key Takeaways
- Naproxen remains a significant component of the NSAID market with an estimated value nearing $3 billion in 2023.
- The market grows at a CAGR of around 3-4%, driven by aging populations and pain management needs.
- OTC formulations dominate sales, making consumer preference and regulatory safety considerations critical.
- Patent expirations and generic competition have increased volume but reduced profit margins for branded products.
- Future growth depends on safety innovations and expanding geographic reach, especially in emerging markets.
FAQs
1. What are the main applications of naproxen?
Naproxen is primarily used for pain relief, including arthritis, gout, menstrual pain, and musculoskeletal injuries.
2. How does naproxen's safety profile affect its market?
Concerns about cardiovascular and gastrointestinal risks limit long-term use. Regulatory warnings and safety labeling influence prescribing and OTC purchase patterns.
3. What is the impact of patent expiration on naproxen sales?
Patent expiration allows generic competitors, increasing overall market volume but reducing revenues for branded formulations.
4. Which regions are experiencing the fastest growth in naproxen sales?
Emerging markets in Asia-Pacific and Latin America show increased demand due to expanding healthcare infrastructure and aging populations.
5. How might future regulatory changes influence naproxen?
Tighter safety regulations can restrict use, mandate labeling changes, or impose usage limits, potentially reducing sales volume.
References
- MarketWatch. "NSAID Market Size, Share & Trends Analysis Report," 2023.
- Grand View Research. "NSAID Market Analysis," 2022.
- FDA. "Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)," 2022.
- IQVIA. "Global Sales Data for NSAIDs," 2022.