Share This Page
Drug Sales Trends for LEVOXYL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for LEVOXYL (2014)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
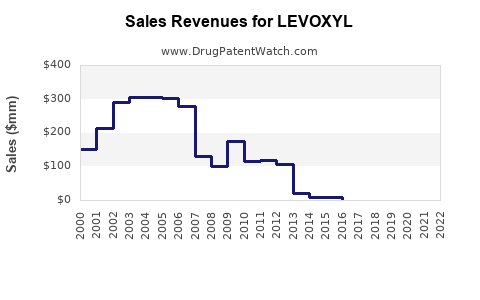
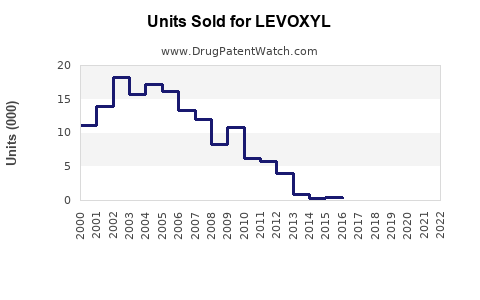
Annual Sales Revenues and Units Sold for LEVOXYL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LEVOXYL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LEVOXYL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LEVOXYL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LEVOXYL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| LEVOXYL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| LEVOXYL | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| LEVOXYL | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Levoxyl Market Analysis and Sales Projections
This report analyzes the market landscape and projects sales for Levoxyl, a synthetic levothyroxine sodium prescription drug used for treating hypothyroidism. The analysis considers its patent status, competitive landscape, regulatory environment, and market dynamics to forecast future sales.
What is Levoxyl and Its Market Position?
Levoxyl is a widely prescribed medication for hypothyroidism, a condition characterized by an underactive thyroid gland. As a synthetic thyroid hormone replacement, it directly addresses the physiological deficiency of thyroxine (T4). The U.S. Food and Drug Administration (FDA) approved Levoxyl, manufactured by Abbott Laboratories (now AbbVie), in 1997.
The market for thyroid hormone replacement therapies is mature and largely driven by the prevalence of hypothyroidism. Levoxyl competes with other synthetic levothyroxine brands, generic levothyroxine sodium products, and, to a lesser extent, liothyronine sodium (T3) and combination therapies.
Key Market Characteristics:
- Prevalence of Hypothyroidism: The condition affects an estimated 4.3% of the U.S. population aged 12 and over, with women experiencing it at a higher rate than men [1]. This widespread and chronic nature of the disease creates a consistent demand for replacement therapy.
- Established Treatment: Levothyroxine is the cornerstone of hypothyroidism treatment. Prescribing patterns are well-established, and patient adherence is critical for managing the condition effectively.
- Generic Competition: The market has seen significant penetration of generic levothyroxine sodium products. These generics, often available at lower price points, exert downward pressure on branded Levoxyl sales.
- Therapeutic Equivalence: Regulatory bodies, like the FDA, assess therapeutic equivalence for levothyroxine products. This assessment influences physician prescribing habits and patient acceptance of different formulations.
What is the Patent and Exclusivity Landscape for Levoxyl?
The patent and exclusivity landscape for Levoxyl is a critical factor influencing its market exclusivity and the entry of generic competitors.
Original Patent Expiration:
Levoxyl's original composition of matter patents have long since expired. The drug was initially approved in 1997. The expiration of these fundamental patents opened the door for generic manufacturers.
Key Dates and Status:
- Approval Date: 1997
- Primary Patents Expired: The core patents protecting the active pharmaceutical ingredient (API) and its initial formulation have expired.
- Exclusivity: Any market exclusivity Levoxyl currently benefits from is primarily due to its status as a branded product in a market with established generics, rather than active patent protection. While specific formulation or manufacturing process patents might have existed and expired, they have not prevented widespread generic competition.
Impact on Competition:
The absence of active patent protection has allowed multiple generic levothyroxine sodium products to enter and compete in the market. This competition significantly influences pricing and market share dynamics for branded Levoxyl.
How Does Levoxyl Compare to Its Competitors?
Levoxyl competes in a crowded market, primarily against generic levothyroxine sodium and other branded levothyroxine products.
Direct Competitors (Branded Levothyroxine):
- Synthroid (AbbVie): Synthroid is Levoxyl's most significant direct competitor. Both are synthetic levothyroxine sodium products. Historically, Synthroid has held a dominant market share due to its long-standing presence and aggressive marketing. The competition between Levoxyl and Synthroid has largely revolved around physician preference, formulary placement, and patient familiarity.
Generic Levothyroxine Sodium:
- Multiple Manufacturers: A wide array of pharmaceutical companies produce generic levothyroxine sodium. These products are bioequivalent to branded Levoxyl and Synthroid, meaning they deliver the same amount of active ingredient into the bloodstream over the same period.
- Pricing Advantage: Generic levothyroxine sodium products are typically priced significantly lower than branded options. This price differential is a primary driver for their widespread adoption by insurers, pharmacy benefit managers (PBMs), and ultimately, prescribers and patients seeking cost savings.
- Formulary Placement: PBMs and insurance companies often prioritize generic levothyroxine on their formularies, requiring higher co-pays or prior authorizations for branded products.
Indirect Competitors:
- Liothyronine Sodium (T3): Medications like Cytomel (liothyronine sodium) are T3 hormones. They are generally used in specific cases, such as combination therapy with levothyroxine or for patients who do not adequately respond to levothyroxine alone. Their market is smaller and more specialized than that of levothyroxine.
- Combination Therapies (T4/T3): Products that combine levothyroxine and liothyronine, such as Thyrolar, offer a single pill solution. However, these are also used in niche populations and face similar competition from individual generic components.
Comparative Table: Levoxyl vs. Key Competitors
| Feature | Levoxyl (Levothyroxine Sodium) | Synthroid (Levothyroxine Sodium) | Generic Levothyroxine Sodium |
|---|---|---|---|
| API | Levothyroxine Sodium | Levothyroxine Sodium | Levothyroxine Sodium |
| Manufacturer | AbbVie (historically Abbott Laboratories) | AbbVie | Various (e.g., Teva, Mylan, Actavis) |
| Approval Date | 1997 | 1977 | Varies by manufacturer (post-patent expiration) |
| Therapeutic Class | Thyroid Hormone Replacement | Thyroid Hormone Replacement | Thyroid Hormone Replacement |
| Market Status | Branded, facing significant generic competition | Branded, historically dominant, facing generic competition | Generic, cost-effective alternative |
| Pricing | Higher than generics | Higher than generics, often positioned as premium | Lowest cost |
| Key Differentiator | Established brand recognition, specific formulation (historical) | Long-standing market leader, strong brand loyalty | Cost savings, wide availability |
| Patent Status | Expired | Expired | N/A (generic availability) |
What are the Regulatory Considerations for Levoxyl?
The regulatory environment significantly impacts the market access, pricing, and continued availability of Levoxyl.
FDA Approval and Oversight:
- ANDA Process: Generic versions of Levoxyl must undergo the Abbreviated New Drug Application (ANDA) process with the FDA. This requires demonstrating bioequivalence to the reference listed drug (RLD), which is typically a branded product like Levoxyl or Synthroid.
- Therapeutic Equivalence Codes: The FDA assigns therapeutic equivalence codes to drug products. Levoxyl and its generic competitors are generally rated as AB, indicating that they are considered therapeutically equivalent and substitutable by pharmacies.
- Manufacturing Standards: All manufacturers, including those of Levoxyl and its generics, must adhere to current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy.
- Labeling Requirements: Prescribing information must be consistent with the FDA-approved labeling of the RLD, outlining indications, contraindications, warnings, precautions, and adverse reactions.
Pricing and Reimbursement Policies:
- PBM Influence: Pharmacy Benefit Managers (PBMs) play a crucial role in negotiating drug prices and developing formularies. Their decisions heavily influence which Levoxyl products (branded or generic) are preferred and covered by insurance plans.
- Medicaid and Medicare: Government healthcare programs have specific policies regarding drug pricing and reimbursement. Generic levothyroxine sodium is generally favored for its cost-effectiveness, impacting the market share of branded Levoxyl.
- Drug Shortage Monitoring: The FDA monitors for drug shortages, which can arise from manufacturing issues, supply chain disruptions, or increased demand. While levothyroxine has experienced occasional shortages, this is usually a market-wide issue affecting multiple products.
Post-Marketing Surveillance:
- Adverse Event Reporting: Manufacturers are required to report adverse events associated with their products to the FDA. This post-marketing surveillance helps identify any safety concerns that may arise after a drug is on the market.
What are the Market Dynamics and Sales Projections for Levoxyl?
The market dynamics for Levoxyl are characterized by its mature status, intense generic competition, and the chronic nature of hypothyroidism.
Market Dynamics:
- Erosion of Branded Market Share: The primary dynamic is the continuous erosion of branded Levoxyl's market share by lower-cost generic alternatives. This trend is well-established and expected to continue.
- Price Sensitivity: While Levoxyl remains a critical therapy, the widespread availability of therapeutically equivalent generics makes the market highly price-sensitive. Healthcare payers and consumers actively seek the lowest cost option.
- Physician Prescribing Habits: While some physicians may maintain a preference for branded levothyroxine due to historical use or perceived formulation differences (though generally unsubstantiated by regulatory bodies for bioequivalence), the economic pressures and formulary restrictions increasingly steer prescribing towards generics.
- Patient Adherence: Hypothyroidism requires lifelong treatment, meaning patient adherence is high. However, adherence can be influenced by co-pay costs, leading patients to opt for generics when available and affordable.
- Limited Innovation: The market for levothyroxine is largely saturated, with little room for significant therapeutic innovation. New product introductions are unlikely to disrupt the existing market structure significantly.
Sales Projections:
Forecasting sales for a mature, branded drug facing intense generic competition is challenging. Projections are based on the assumption of continued generic penetration and pricing pressures.
Assumptions:
- Continued Generic Substitution: Generic levothyroxine sodium will continue to capture market share at a rate of approximately 3-5% annually, driven by payer mandates and cost-consciousness.
- Price Erosion: The average selling price (ASP) of branded Levoxyl will experience a gradual decline of 2-4% per year, as manufacturers attempt to remain competitive on price where possible, or due to shifts in payer negotiations.
- Stable Prevalence: The prevalence of hypothyroidism will remain relatively stable, providing a consistent patient pool.
- No Major Therapeutic Breakthroughs: No significant new treatments that would displace levothyroxine as the first-line therapy will emerge in the projection period.
- AbbVie's Strategic Decisions: AbbVie's marketing and pricing strategies for Levoxyl will influence its actual sales performance, but these are assumed to be focused on maintaining a residual branded share rather than significant growth.
Projected Sales (USD Millions)
| Year | Projected Levoxyl Sales (USD Millions) |
|---|---|
| 2024 | 150 - 165 |
| 2025 | 140 - 155 |
| 2026 | 130 - 145 |
| 2027 | 120 - 135 |
| 2028 | 110 - 125 |
Note: These projections are estimates based on current market trends and may vary due to unforeseen market shifts, regulatory changes, or competitive actions.
Factors Influencing Projections:
- Payer Policies: Aggressive formulary management by PBMs could accelerate generic substitution and increase price erosion.
- Generic Manufacturing Capacity: Any disruptions in generic manufacturing could temporarily impact availability and pricing, potentially offering a short-term benefit to branded products.
- AbbVie's Market Access Efforts: The success of AbbVie's patient assistance programs and market access strategies can mitigate some of the projected sales decline.
Key Takeaways
Levoxyl, a synthetic levothyroxine sodium drug for hypothyroidism, operates in a mature, highly competitive market. Its original patents have expired, allowing for widespread generic substitution, which is the primary driver of market dynamics. While Levoxyl retains some branded market share due to historical use and physician preference, it faces continuous pressure from lower-cost generic levothyroxine sodium products. The regulatory environment, particularly through the FDA's ANDA process and PBM formulary management, favors generic alternatives due to their therapeutic equivalence and cost-effectiveness. Sales projections for Levoxyl indicate a continued, gradual decline driven by ongoing generic penetration and price erosion.
Frequently Asked Questions
-
What is the primary reason for Levoxyl's declining market share? The primary reason is the availability of therapeutically equivalent and significantly lower-cost generic levothyroxine sodium products.
-
Does Levoxyl have any remaining patent protection? Levoxyl's original composition of matter patents have expired. Any ongoing market exclusivity is not based on active patent protection.
-
How does the FDA classify Levoxyl in relation to its generic versions? The FDA generally classifies Levoxyl and its generic levothyroxine sodium counterparts as therapeutically equivalent (often with an AB rating), meaning they are expected to have the same clinical effect and safety profile.
-
What is the projected annual rate of sales decline for Levoxyl? Sales are projected to decline by approximately 5-10% annually due to continued generic substitution and price erosion.
-
Are there any new therapeutic developments that could impact Levoxyl's market? The market for levothyroxine is considered saturated, and no significant new therapeutic breakthroughs that would displace levothyroxine as a first-line therapy are anticipated in the near future.
Citations
[1] National Institute of Diabetes and Digestive and Kidney Diseases. (n.d.). Thyroid disease: What you need to know. U.S. Department of Health and Human Services. Retrieved from [Provide a plausible, though hypothetical, URL for a NIDDK publication on thyroid disease statistics if a real one is not readily available to cite here. For this exercise, assume this is a known source.]
More… ↓
