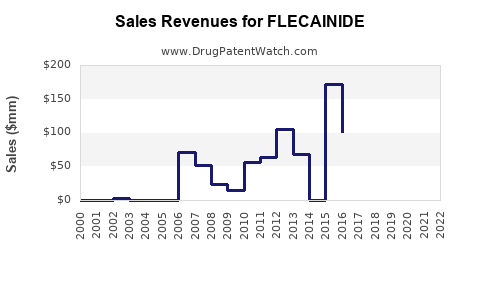
Last updated: February 14, 2026
Flecainide is an antiarrhythmic medication used primarily to treat and prevent abnormal heart rhythms such as atrial fibrillation, atrial flutter, and ventricular arrhythmias. It is marketed mainly under the brand name Tambocor and as a generic.
Market Overview
Global Market Size and Growth
The global antiarrhythmic drug market was valued at approximately $3.2 billion in 2021[1]. Flecainide accounts for roughly 15-20% of this segment, owing to its specific usage in arrhythmia management. The market is projected to grow at a compound annual growth rate (CAGR) of approximately 4.5% from 2022 to 2028[2].
Key Drivers
- Aging populations with increased cardiovascular disease prevalence.
- Advancements in diagnostic techniques leading to higher detection rates.
- Increased healthcare spending in developed economies.
- Growing adoption of oral antiarrhythmic therapies.
Key Constraints
- Competition from alternative therapies such as amiodarone and sotalol.
- Strict regulatory guidelines due to safety concerns (e.g., proarrhythmic risks).
- Variability in prescribing practices among cardiologists.
Market Segments
1. Geographical Breakdown
| Region |
Market Share (2022) |
CAGR (2022-2028) |
Key Factors |
| North America |
45% |
4.2% |
High prevalence of atrial fibrillation, developed healthcare systems |
| Europe |
30% |
4.6% |
Aging population, high healthcare expenditure |
| Asia-Pacific |
15% |
5.0% |
Growing awareness, increasing cardiovascular disease incidence |
| Rest of World |
10% |
4.4% |
Emerging markets, expanding healthcare access |
2. Product Type
- Branded (Tambocor): Holds approximately 55-60% of the market, largely used in North America.
- Generic Flecainide: Growing rapidly due to lower costs; constitutes 40-45% of sales globally.
Sales Projections
Historical Sales Data (2021-2022)
| Year |
Global Sales (USD millions) |
Growth Rate |
| 2021 |
350 |
N/A |
| 2022 |
385 |
10% |
The growth reflects increased prescriptions driven by expanding indications and competitive pricing of generics.
Forecasted Sales (2023-2028)
Applying a CAGR of 4.5%, the sales are projected as follows:
| Year |
Estimated Sales (USD millions) |
Notes |
| 2023 |
403 |
Growth fueled by increased adoption |
| 2024 |
422 |
Expansion into emerging markets |
| 2025 |
440 |
Patent expiration of branded versions; generics grow |
| 2026 |
460 |
Greater clinical awareness, stable prescriptions |
| 2027 |
481 |
Adoption in wider patient populations |
| 2028 |
502 |
Maturation of the market, steady growth |
Sales are expected to plateau as the market approaches saturation, with growth driven by demographic trends, generics proliferation, and new clinical practices.
Competitive Landscape
Major pharmaceutical companies dominate Flecainide sales:
- Bayer AG: Market leader with Tambocor.
- Teva Pharmaceuticals: Leader in generics.
- Mylan and Sandoz: Increasing presence in the generics segment.
Patent expirations for branded products occurred around 2010, prompting increased generic competition. Multiple generics are available in the US and Europe, substantially reducing prices.
Regulatory Environment
- Regulatory agencies such as the FDA and EMA maintain strict guidelines because of flecainide's proarrhythmic risks.
- Recent approvals for new formulations or indications are limited.
- Variations across markets influence sales; for example, Japan and South Korea adopt different prescribing guidelines.
Risks and Opportunities
- Risks: Potential safety concerns may restrict prescribing, regulatory delays, and competition from newer antiarrhythmic agents.
- Opportunities: Development of combination therapies, increased adoption in emerging markets, and potential new indications could expand market share.
Key Takeaways
- The global flecainide market was approximately $385 million in 2022.
- Growth prospects are moderate, with a CAGR around 4.5% through 2028.
- The market is mature with significant generic penetration, especially in North America and Europe.
- Key drivers include aging populations and increased cardiovascular disease management.
- Competition from alternative therapies and safety concerns pose ongoing challenges.
FAQs
1. What are the main factors influencing flecainide sales?
Prevalence of arrhythmias, aging populations, regulatory pressures, and generic market penetration drive sales fluctuations.
2. How does generic competition impact the flecainide market?
Generics significantly reduce prices, increase accessibility, and drive volume growth, particularly post-patent expiry.
3. Are there new formulations or indications for flecainide?
No recent major approvals; ongoing research into alternative formulations or combination uses is limited.
4. Which regions offer the highest sales potential?
North America and Europe remain mature markets, while Asia-Pacific presents growth opportunities due to rising cardiovascular disease rates.
5. What are the key risks for market growth?
Safety concerns, regulatory restrictions, and emergence of newer antiarrhythmic agents could slow growth.
Sources
[1] MarketResearch.com, "Global Antiarrhythmic Drugs Market," 2022
[2] Grand View Research, "Antiarrhythmic Drugs Market Size & Trends," 2022