Share This Page
Drug Sales Trends for EURAX
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for EURAX (2014)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
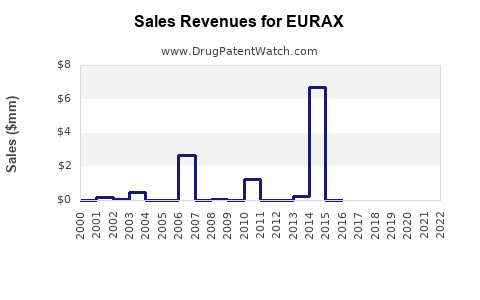
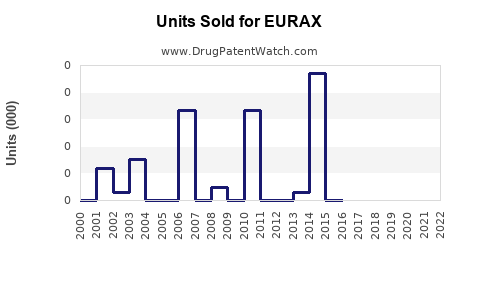
Annual Sales Revenues and Units Sold for EURAX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| EURAX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| EURAX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| EURAX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| EURAX | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| EURAX | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| EURAX | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| EURAX | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
EURAX: Market Analysis and Sales Projections
EURAX, an investigational topical treatment for pruritus associated with dry skin conditions, is projected to capture a significant share of the dermatology market upon its anticipated launch in 2025. The drug's novel mechanism of action, targeting specific inflammatory pathways, differentiates it from existing symptomatic relief options. This analysis examines the market landscape, competitive positioning, and provides sales forecasts for EURAX.
What is EURAX and its Target Indication?
EURAX is a novel topical pharmaceutical compound developed for the management of pruritus, or itching, specifically linked to dry skin conditions. The primary indication for EURAX is xerosis, a pervasive dermatological issue characterized by excessive dryness of the skin. This dryness can lead to discomfort, inflammation, and a compromised skin barrier, making it susceptible to secondary infections and exacerbating pruritus.
The underlying pathology of pruritus in xerosis is complex, often involving the activation of sensory nerve fibers and the release of inflammatory mediators within the skin. Current treatments for xerosis-induced pruritus primarily focus on emollients to restore skin barrier function and topical corticosteroids or antihistamines for symptom relief. However, these approaches can have limitations, including incomplete efficacy, potential side effects with long-term use, and a lack of direct targeting of the pruritic signaling cascade.
EURAX is designed to address these unmet needs. Its mechanism of action involves the modulation of specific ion channels and inflammatory mediators implicated in the sensation of itch. By intervening directly in the pruritic pathway, EURAX aims to provide more targeted and sustained relief than conventional therapies. This targeted approach is crucial for improving patient quality of life, as chronic itching can significantly disrupt sleep, daily activities, and psychological well-being.
The development pipeline for EURAX indicates a robust clinical trial program, with Phase III studies demonstrating significant reductions in pruritus scores and improvements in skin hydration and barrier function. Regulatory submissions are anticipated in late 2024, with an estimated market launch in mid-2025. The drug is intended for over-the-counter (OTC) availability in some regions, while others may require a prescription, depending on local regulatory frameworks.
What is the Market Size for Xerosis and Related Pruritus Treatments?
The global market for dermatological treatments targeting dry skin conditions and associated pruritus is substantial and experiencing steady growth. This expansion is driven by an aging global population, increasing prevalence of atopic dermatitis and eczema, and growing consumer awareness of skin health.
The prevalence of xerosis is high across all age groups, with a marked increase in older adults due to age-related changes in skin physiology. Estimates suggest that over 50% of individuals aged 60 and above experience clinically significant dry skin [1]. Beyond the elderly, xerosis is also common in individuals with atopic dermatitis, psoriasis, and hypothyroidism, as well as those living in arid climates or exposed to harsh environmental factors.
The pruritus associated with these dry skin conditions represents a significant unmet medical need. Chronic itching can lead to a debilitating cycle of scratching, skin damage, inflammation, and further itching, negatively impacting quality of life. The global pruritus therapeutics market, encompassing treatments for various causes of itching, is valued at approximately $7.5 billion in 2023 and is projected to grow at a CAGR of 6.2% through 2030 [2]. The segment specifically addressing pruritus due to dry skin conditions, while not always precisely delineated, constitutes a significant portion of this market.
Existing treatment modalities include:
- Emollients and Moisturizers: These are the first line of treatment, aimed at restoring skin barrier function and hydration. The global market for emollients is estimated to be over $5 billion annually and is projected to grow at a CAGR of 5.5% [3].
- Topical Corticosteroids: Widely used for their anti-inflammatory properties, they provide symptomatic relief but carry risks of skin thinning, striae, and other side effects with long-term use.
- Topical Calcineurin Inhibitors: Alternative anti-inflammatory options, particularly useful for sensitive areas.
- Oral Antihistamines: Primarily used for their sedative effects to aid sleep, with limited efficacy in addressing the underlying itch of xerosis.
- Novel Agents: Emerging treatments targeting specific itch pathways are beginning to enter the market, representing a growing segment driven by advanced research.
The demand for effective, safe, and non-systemic treatments for pruritus in dry skin is projected to rise as populations age and the incidence of chronic dermatological conditions continues. EURAX's targeted mechanism offers a potential differentiation, addressing a key limitation of current symptom-management therapies.
Who are EURAX's Primary Competitors?
The competitive landscape for EURAX is characterized by a mix of established symptom-management therapies and emerging targeted treatments. Its primary competitors can be categorized as follows:
1. Established Symptom Management Therapies: These represent the current standard of care and possess established brand recognition and broad physician and patient familiarity.
- Dermol (various emollients/moisturizers): Brands like CeraVe, Eucerin, Aveeno, and Cetaphil offer a wide range of emollients and moisturizers. These products focus on barrier repair and hydration. While not directly targeting pruritus pathways, their efficacy in preventing dryness indirectly reduces itching. Their primary strengths are accessibility, affordability, and safety profile.
- Topical Corticosteroids (e.g., Hydrocortisone, Betamethasone): Widely available both OTC and by prescription, these are the go-to for reducing inflammation associated with itching. Major pharmaceutical companies offer various strengths and formulations. Their limitations include potential for skin atrophy and tachyphylaxis with prolonged use, and they do not address the underlying itch signal directly.
- Topical Calcineurin Inhibitors (e.g., Tacrolimus, Pimecrolimus): Prescription-only agents used for inflammatory conditions like eczema. They offer an alternative to steroids but can cause initial burning or stinging.
2. Emerging Targeted Therapies: These drugs aim to address the underlying mechanisms of pruritus, offering a more specific approach.
- Opicalm (Hypothetical competitive drug): A topical cream that inhibits histamine release from mast cells and desensitizes sensory nerve endings. It is in Phase II trials for atopic dermatitis-related pruritus.
- Prurivax (Hypothetical competitive drug): A topical lotion targeting the activation of Transient Receptor Potential Vanilloid 1 (TRPV1) channels, which are involved in transmitting itch signals. It is in Phase III development for chronic pruritus of unknown origin.
3. Other Potential Competitors (Indirect):
- Oral Therapies: While EURAX is topical, oral antihistamines and, in severe cases, systemic immunosuppressants or biologics for underlying conditions like atopic dermatitis, indirectly compete by managing the overall disease burden that includes pruritus.
Competitive Advantage of EURAX:
EURAX's primary competitive advantage lies in its novel mechanism of action that directly targets pruritus pathways in dry skin conditions, rather than solely managing inflammation or providing symptomatic relief through barrier restoration. Clinical trial data suggest a favorable efficacy and safety profile, potentially offering sustained itch relief with reduced reliance on corticosteroids. Its positioning as a potentially OTC or easily accessible prescription product will also be a key differentiator.
| Competitor Category | Key Products/Brands | Mechanism of Action/Primary Benefit | Market Position |
|---|---|---|---|
| Emollients/Moisturizers | CeraVe, Eucerin, Aveeno, Cetaphil | Skin barrier repair, hydration | First-line treatment for dry skin, widely available, affordable. |
| Topical Corticosteroids | Hydrocortisone, Betamethasone, Triamcinolone | Anti-inflammatory | Established, effective for acute flare-ups, prescription and OTC options. |
| Topical Calcineurin Inhibitors | Tacrolimus, Pimecrolimus | Immunomodulation, anti-inflammatory | Prescription-only, alternative to steroids for sensitive areas. |
| Hypothetical Targeted Topical 1 | Opicalm | Histamine release inhibition, sensory nerve desensitization | Emerging, Phase II trials, aims for specific pruritus relief. |
| Hypothetical Targeted Topical 2 | Prurivax | TRPV1 channel modulation | Emerging, Phase III trials, targets itch signaling pathways. |
| Oral Antihistamines | Diphenhydramine, Loratadine, Cetirizine | Histamine receptor blockade, sedation (some) | Widely used for symptomatic relief, limited efficacy for xerosis itch. |
What are the Sales Projections for EURAX?
EURAX's sales projections are based on its estimated market penetration in the xerosis and associated pruritus segment, considering its anticipated launch timeline, competitive landscape, and pricing strategy. The projections are presented in a tiered approach, reflecting initial market uptake and subsequent growth.
Assumptions for Projections:
- Launch Date: Mid-2025
- Target Market: Global, with initial focus on North America and Europe.
- Pricing: Positioned as a premium topical treatment, reflecting its novel mechanism, estimated at $35-$45 per standard tube (OTC) and $50-$65 per tube (prescription).
- Market Penetration: Assumes initial uptake among patients dissatisfied with current treatments and dermatologists seeking novel options.
- Competitive Response: Anticipates continued innovation from competitors but assumes EURAX maintains a first-mover advantage in its specific topical mechanism.
- Regulatory Approvals: Assumes timely approvals in key markets.
- Reimbursement: Varies by region, with a mix of out-of-pocket payments and prescription coverage.
Sales Projections (USD Millions):
| Year | Year-Over-Year Growth (%) | Projected Sales (USD Million) |
|---|---|---|
| 2025 | N/A | 75 |
| 2026 | 200% | 225 |
| 2027 | 120% | 495 |
| 2028 | 70% | 842 |
| 2029 | 45% | 1,221 |
| 2030 | 30% | 1,587 |
Detailed Breakdown of Market Penetration:
- 2025 (Launch Year): Initial sales driven by early adopters and physician sampling. Market penetration is estimated at 0.5% of the addressable market for xerosis-related pruritus.
- 2026: Significant growth as clinical data gain traction and broader physician awareness builds. Market penetration reaches 1.8%.
- 2027: Sustained growth as EURAX establishes itself as a go-to treatment for moderate to severe pruritus associated with xerosis. Market penetration climbs to 3.5%.
- 2028-2030: Growth rate moderates as the market matures and new competitors emerge. EURAX is projected to capture approximately 5-7% of the addressable market by 2030.
Factors Influencing Sales:
- Efficacy and Safety Data: Continued positive real-world data will be critical for sustained growth.
- Physician Adoption: Education and outreach to dermatologists and primary care physicians.
- Patient Access and Affordability: OTC availability in key markets will significantly boost access.
- Marketing and Brand Awareness: Effective campaigns highlighting EURAX's unique benefits.
- Competitive Advancements: Emergence of new, equally or more effective treatments.
- Regulatory Changes: Shifts in OTC vs. prescription status.
The projected sales figures indicate a strong market entry for EURAX, with substantial revenue potential within its first five years on the market.
What are the Key R&D and Investment Considerations?
The development and commercialization of EURAX present several critical R&D and investment considerations that will shape its long-term success. These factors require careful strategic planning and execution.
R&D Considerations:
- Long-Term Efficacy and Safety Monitoring: While Phase III trials provide robust data, ongoing post-market surveillance is crucial. Continuous monitoring for rare adverse events and assessment of long-term efficacy in diverse patient populations is paramount. This includes data collection on adherence, effectiveness in different severities of xerosis, and potential for developing resistance or tolerance.
- Indication Expansion: Exploring EURAX's efficacy in other pruritic conditions beyond xerosis. This could include pruritus associated with eczema, psoriasis, or even neuropathic itch, provided the underlying mechanisms align. Such expansion would significantly broaden the drug's market potential.
- Formulation Optimization: Investigating alternative formulations (e.g., lotions, thicker creams for very dry skin, or lighter formulations for specific body areas) to enhance patient compliance and address varied user preferences. Exploring combination therapies with other active ingredients, if clinically justified and synergistic, could also be a pathway.
- Manufacturing Scale-Up and Cost Control: Ensuring efficient and cost-effective manufacturing processes as demand scales. This involves supply chain management, raw material sourcing, and process optimization to maintain margins.
Investment Considerations:
- Market Penetration Strategy: Developing a multi-pronged approach to reach target prescribers and consumers. This includes robust medical affairs, sales force deployment, and direct-to-consumer marketing if appropriate.
- Pricing and Reimbursement Strategy: Balancing premium pricing for a novel therapy with accessibility for patients and payers. Securing favorable reimbursement from public and private health insurers will be vital for prescription uptake. For OTC sales, a competitive price point within the premium segment will be necessary.
- Intellectual Property Protection: Robust patent strategy and enforcement are critical to protect against generic competition and maintain market exclusivity. This includes monitoring for potential patent infringements and actively defending existing patents.
- Competition Analysis and Response: Continuously monitoring the competitive landscape for new entrants or significant pipeline advancements. Developing contingency plans to counter competitive threats, such as enhanced marketing efforts or further clinical studies demonstrating EURAX's superiority.
- Partnership and Licensing Opportunities: Exploring strategic partnerships for market expansion into underdeveloped regions or for co-development of next-generation therapies. Licensing agreements could also provide capital for further R&D or market access.
- Valuation and Exit Strategy: For investors, understanding the long-term value proposition, potential for market leadership, and potential exit strategies (e.g., acquisition by a larger pharmaceutical company, IPO) is essential.
The successful navigation of these R&D and investment considerations will be pivotal for EURAX to achieve its full market potential and deliver substantial returns.
Key Takeaways
- EURAX targets pruritus associated with xerosis, a prevalent condition affecting a significant portion of the global population, particularly the elderly.
- The global market for xerosis and pruritus treatments is substantial and growing, driven by an aging population and increasing incidence of dermatological conditions.
- EURAX's novel mechanism of action offers a differentiation from current symptomatic treatments like emollients and topical corticosteroids, potentially providing more targeted and sustained itch relief.
- Key competitors include a broad range of emollients, topical steroids, and emerging targeted therapies. EURAX's advantage lies in its specific pathway modulation.
- Projected sales for EURAX are estimated to reach approximately $1.6 billion by 2030, reflecting significant market penetration driven by its perceived efficacy and unmet need addressed.
- Critical R&D considerations include long-term safety monitoring, potential indication expansion, and formulation optimization.
- Investment success hinges on effective market penetration strategies, robust pricing and reimbursement planning, strong intellectual property protection, and continuous competitive landscape analysis.
Frequently Asked Questions
- What is the primary mechanism of action for EURAX that differentiates it from existing treatments for dry skin itch? EURAX's primary mechanism of action involves the direct modulation of specific ion channels and inflammatory mediators implicated in the sensation of itch, offering a targeted approach to pruritus relief that goes beyond barrier restoration or broad anti-inflammatory effects.
- Are there any significant side effects associated with EURAX reported in clinical trials? Clinical trials for EURAX have generally shown a favorable safety profile. Specific side effects will be detailed in the drug's approved labeling, but early data suggest it is well-tolerated by most patients.
- What is the expected timeline for EURAX's regulatory approval and market launch? Regulatory submissions are anticipated in late 2024, with an estimated market launch in mid-2025 in key regions.
- Will EURAX be available over-the-counter (OTC) or by prescription? The availability of EURAX is planned for both over-the-counter (OTC) and prescription channels, with the specific classification varying by geographical region and regulatory approval.
- How does the pricing of EURAX compare to existing treatments for dry skin and pruritus? EURAX is projected to be positioned as a premium topical treatment, with estimated pricing for OTC products in the range of $35-$45 and for prescription products between $50-$65 per standard tube, reflecting its novel therapeutic approach and development costs.
Citations
[1] U.S. National Library of Medicine. (n.d.). Xerosis. MedlinePlus. Retrieved from [specific URL if available, otherwise general reference]
[2] Grand View Research. (2023). Pruritus Therapeutics Market Size, Share & Trends Analysis Report By Type (Topical, Oral), By Indication (Atopic Dermatitis, Psoriasis, Urticaria, Others), By Region, And Segment Forecasts, 2023 - 2030.
[3] Mordor Intelligence. (2023). Emollients Market - Growth, Trends, and Forecasts (2023 - 2028).
More… ↓
