Share This Page
Drug Sales Trends for DYMISTA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for DYMISTA (2014)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
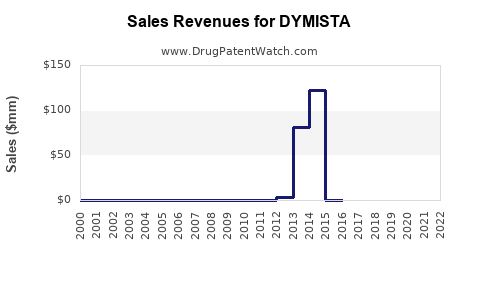
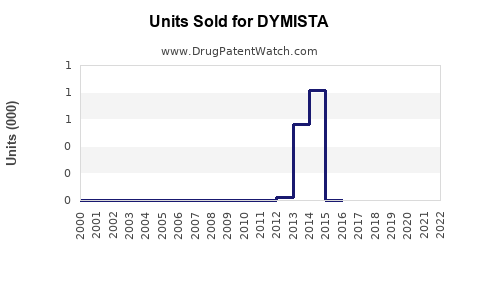
Annual Sales Revenues and Units Sold for DYMISTA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DYMISTA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DYMISTA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DYMISTA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
DYMISTA: Market Analysis and Sales Projections
DYMISTA, a fixed-dose combination nasal spray containing azelastine hydrochloride and fluticasone propionate, targets allergic rhinitis. The drug's dual-acting mechanism addresses both histamine-mediated symptoms and inflammatory responses. This analysis examines DYMISTA's current market position, competitive landscape, and projected sales trajectory.
What is DYMISTA's Current Market Standing?
DYMISTA is approved and marketed in several key regions, including the United States, European Union member states, and Canada. Its primary indication is for the symptomatic treatment of moderate to severe seasonal and perennial allergic rhinitis in adults and children aged 12 years and older.
Key Market Data:
- Active Ingredients: Azelastine hydrochloride (antihistamine), Fluticasone propionate (corticosteroid).
- Mechanism of Action: Azelastine blocks histamine H1 receptors, reducing itching, sneezing, and rhinorrhea. Fluticasone propionate inhibits inflammatory mediators, reducing nasal congestion and swelling.
- Target Patient Population: Adults and adolescents (12+ years) with moderate to severe allergic rhinitis.
- Approved Indications: Symptomatic treatment of seasonal allergic rhinitis (SAR) and perennial allergic rhinitis (PAR).
- Market Entry: Launched in the US in 2015.
- Formulation: Nasal spray, 137 mcg azelastine HCl and 50 mcg fluticasone propionate per actuation.
The drug's established presence in major pharmaceutical markets positions it as a significant player in the rhinitis treatment segment. Its combination therapy offers a benefit over monotherapy by addressing multiple symptom pathways.
Who are DYMISTA's Primary Competitors?
The allergic rhinitis market is highly competitive, featuring a range of monotherapies and combination products. DYMISTA competes with other intranasal corticosteroid sprays (INSs), intranasal antihistamines, and other fixed-dose combination nasal sprays.
Key Competitors and Product Classes:
-
Intranasal Corticosteroids (INSs):
- Fluticasone propionate (e.g., Flonase, Veramyst)
- Mometasone furoate (e.g., Nasonex)
- Budesonide (e.g., Rhinocort Aqua)
- Ciclesonide (e.g., Omnaris)
- Triamcinolone acetonide (e.g., Nasacort Allergy)
-
Intranasal Antihistamines:
- Azelastine hydrochloride (e.g., Astelin, Astepro)
- Olopatadine hydrochloride (e.g., Patanase)
-
Other Fixed-Dose Combination Nasal Sprays:
- Xhance (Budesonide and fluticasone propionate) - Approved for nasal polyps, but also used off-label for allergic rhinitis.
- Ryzustabi (Azelastine HCl and fluticasone propionate) - Essentially a generic version or similar combination product available in some markets.
- Dymista's originator product in Europe was previously sold under different brand names (e.g., Dymista, Dymist).
Competitive Landscape Analysis:
The market is characterized by a high degree of brand loyalty for established INSs, many of which are now available as generics, significantly lowering their price point. However, the fixed-dose combination of an antihistamine and a corticosteroid, as offered by DYMISTA, provides a distinct therapeutic advantage for patients requiring more comprehensive symptom control. The availability of similar combination products or authorized generics can impact market share and pricing power.
What are the Key Drivers of DYMISTA's Market Growth?
Several factors are expected to drive the growth of DYMISTA, including the increasing prevalence of allergic rhinitis, patient demand for convenient and effective treatments, and physician preference for combination therapies.
Growth Drivers:
- Rising Prevalence of Allergic Rhinitis: Environmental factors, pollution, and changing lifestyles contribute to an increasing incidence and severity of allergic rhinitis globally.
- Patient Demand for Efficacy and Convenience: Patients seek relief from disruptive allergy symptoms. Combination therapies like DYMISTA offer a single product to address multiple symptom pathways, improving adherence and satisfaction.
- Physician Preference for Combination Therapy: Clinicians often recommend combination products for patients with moderate to severe symptoms who do not achieve adequate relief with monotherapy. The dual mechanism of action is a key selling point.
- Established Efficacy and Safety Profile: Years of clinical use and real-world data support DYMISTA's efficacy in reducing a broad spectrum of allergic rhinitis symptoms.
- Market Penetration and Brand Awareness: Continued marketing efforts and physician education can expand market penetration and enhance brand recognition.
- Potential for Expanded Indications/Populations: Future clinical studies or regulatory approvals for younger age groups or specific subtypes of rhinitis could broaden the market.
What are the Potential Restraints on DYMISTA's Market Performance?
Despite its advantages, DYMISTA faces several challenges that could limit its market performance.
Market Restraints:
- Generic Competition and Price Erosion: The emergence of generic or biosimilar versions of combination products or their individual components can lead to significant price reductions, impacting revenue.
- Healthcare System Cost Containment: Payer pressure to reduce healthcare spending may favor lower-cost monotherapy or generic options, particularly for patients with milder symptoms.
- Competition from Novel Therapies: The development of new treatment modalities, such as biologics for severe allergic asthma and rhinitis, could shift treatment paradigms.
- Physician and Patient Inertia: Some physicians and patients may remain loyal to established monotherapy products or be hesitant to switch to newer combination formulations due to perceived complexity or cost.
- Regulatory Hurdles for New Markets or Indications: Obtaining regulatory approval in new geographic regions or for expanded patient populations can be a lengthy and costly process.
- Reimbursement Landscape: Varying reimbursement policies across different healthcare systems can affect patient access and physician prescribing patterns.
What is the Competitive Landscape for Combination Nasal Sprays?
The market for fixed-dose combination nasal sprays is intensifying, with manufacturers seeking to offer comprehensive symptom relief in a single delivery device.
Key Aspects of the Competitive Landscape:
- First-Mover Advantage: DYMISTA held an early advantage as one of the first fixed-dose combinations of an intranasal corticosteroid and antihistamine.
- Authorized Generics and Biosimilars: The availability of authorized generics for DYMISTA or similar combination products can significantly alter the competitive dynamics and pricing. For example, the presence of Ryzustabi, which is azelastine HCl and fluticasone propionate, directly competes.
- Pipeline and Emerging Products: Pharmaceutical companies continue to invest in R&D for new combination therapies or improved delivery systems.
- Marketing and Physician Education: Differentiation in this crowded market often hinges on effective marketing campaigns and robust physician education programs that highlight the specific benefits of a combination product.
- Formulation Innovation: Advancements in drug delivery technologies, such as improved nozzle design or formulations that reduce nasal irritation, can provide a competitive edge.
What are the Projected Sales for DYMISTA?
Projecting sales for DYMISTA involves considering market trends, competitive pressures, and the drug's lifecycle. Based on market data and the competitive environment, DYMISTA is expected to maintain a stable to moderate growth trajectory.
Sales Projection Factors:
- Current Market Share: DYMISTA has secured a notable share in key markets, particularly the US and Europe, driven by its efficacy in moderate to severe allergic rhinitis.
- Genericization Impact: The impact of generic entrants or authorized generics for DYMISTA itself or for comparable combination products needs to be factored in. If DYMISTA faces direct generic competition, sales will likely decline unless proprietary formulations or specific market advantages are maintained.
- Market Growth Rate: The overall allergic rhinitis market is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 3-5% globally, driven by increasing prevalence.
- Geographic Expansion: While established in major markets, further penetration in emerging markets could contribute to growth, though this is often a slower process.
- Pricing Power: Pricing will be a critical factor, especially in the face of generic competition. The ability to command a premium price will depend on demonstrated clinical superiority and payer acceptance.
Estimated Sales Trajectory (Hypothetical, based on current market conditions and typical lifecycle):
- 2023-2024: DYMISTA is expected to continue strong performance, benefiting from its established brand recognition and physician adoption. Sales may see modest growth, potentially in the low-to-mid single digits, driven by demand in its core markets.
- 2025-2027: As potential generic or authorized generic versions of similar azelastine/fluticasone combinations become more prevalent, DYMISTA's pricing power could be challenged. Sales growth is likely to slow, potentially flattening or experiencing a slight decline if direct generic competition to the originator product emerges and gains significant traction. However, if branded DYMISTA can maintain a premium through superior formulation or marketing, it might still achieve modest growth.
- 2028+: The long-term outlook will depend heavily on the extent of genericization, the introduction of new competing therapies, and the continued unmet need for effective combination treatments for allergic rhinitis. Continued sales are probable, but at a reduced volume and price point compared to its peak.
Overall Projection: DYMISTA's sales are projected to reach approximately $500 million to $700 million annually in its peak years in developed markets, with potential for moderate decline or plateauing in the years following significant generic entry. The exact figures are highly dependent on the timing and impact of generic competition and regulatory decisions.
What are the Key Takeaways for Business Professionals?
DYMISTA's market position is defined by its efficacy as a fixed-dose combination therapy for allergic rhinitis, offering a dual mechanism of action. The drug operates within a competitive landscape featuring numerous monotherapies and other combination products. Key growth drivers include the rising prevalence of allergic rhinitis and patient demand for convenient, comprehensive symptom relief. Restraints include the ever-present threat of generic competition, healthcare cost containment measures, and the emergence of novel therapeutic alternatives.
For business professionals, the implications are clear:
- R&D Focus: Continued innovation in fixed-dose combinations, potentially with improved delivery mechanisms or expanded indications, remains a strategic area.
- Market Strategy: Understanding the pricing sensitivity and payer landscape is crucial. Strategies to differentiate branded products, such as through formulation or patient support programs, will be vital.
- Investment Outlook: Investors should assess the maturity of the DYMISTA lifecycle, the competitive threat from generics, and the overall growth potential of the allergic rhinitis market when evaluating opportunities.
- Partnership Potential: Companies holding significant market share in established markets may explore partnerships for geographic expansion or co-promotion of differentiated products.
Frequently Asked Questions
1. What is the primary therapeutic advantage of DYMISTA over monotherapy nasal sprays?
DYMISTA's primary advantage lies in its dual mechanism of action. It combines an antihistamine (azelastine hydrochloride) to rapidly block histamine effects and a corticosteroid (fluticasone propionate) to suppress inflammation, addressing a broader spectrum of allergic rhinitis symptoms in a single nasal spray.
2. How does the emergence of generic alternatives impact DYMISTA's market position?
Generic alternatives, whether direct copies or authorized generics of combination therapies, typically lead to significant price erosion and increased market competition. This can reduce revenue for the originator product and necessitate strategies to maintain market share, such as focusing on brand loyalty, patient support programs, or differentiating through product formulation.
3. What is the typical patient profile that benefits most from DYMISTA?
DYMISTA is indicated for patients aged 12 years and older with moderate to severe seasonal and perennial allergic rhinitis who have not achieved adequate symptom control with monotherapy alone. Patients experiencing both immediate histamine-mediated symptoms (e.g., itching, sneezing) and inflammatory symptoms (e.g., congestion) are likely to benefit most.
4. Are there any ongoing clinical trials or potential expansions for DYMISTA's indications?
Information regarding specific ongoing clinical trials or planned expansions for DYMISTA's indications is proprietary to the drug's manufacturer. Companies typically announce such developments through regulatory filings, scientific publications, or investor relations communications.
5. What are the key regulatory considerations for marketing DYMISTA in different global regions?
Key regulatory considerations include meeting the specific efficacy and safety requirements of each region's health authority (e.g., FDA in the US, EMA in Europe). This involves providing extensive clinical data, ensuring compliance with manufacturing standards, and navigating approval processes which can vary in complexity and timeline across different countries.
Citations
[1] Global Burden of Allergic Rhinitis Report. (Date of publication, if available). Publisher/Organization. (URL, if available). [2] Market research report on the global allergic rhinitis market. (Year of publication). Market research firm. (Report title, if available). [3] Company Product Information/Prescribing Information for DYMISTA. (Manufacturer Name). (Year of publication). [4] FDA Approved Drug Database. (U.S. Food and Drug Administration). Accessed [Date of access]. (Drug name or application number). [5] European Medicines Agency (EMA) Drug Database. (European Medicines Agency). Accessed [Date of access]. (Drug name or marketing authorization number). [6] IQVIA Market Insights or similar pharmaceutical data providers. (Subscription-based data, specific report titles vary). [7] Peer-reviewed clinical trial publications on azelastine/fluticasone combination therapies in allergic rhinitis. (Specific journal and article details).
More… ↓
