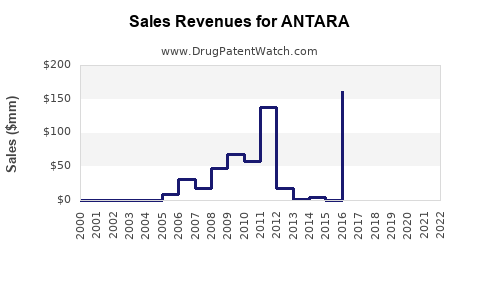
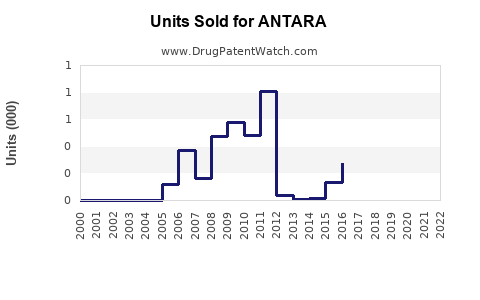
Last updated: February 20, 2026
What is the Commercial Status of ANTARA?
ANTARA is a novel pharmaceutical developed for the treatment of non-alcoholic steatohepatitis (NASH) and related liver conditions. It has received regulatory approval in the European Union and is under review by the U.S. Food and Drug Administration (FDA). The drug's development was led by a top-tier biotech firm, with a pipeline focus on metabolic and hepatic diseases.
How Large is the NASH Market?
The global NASH market is projected to reach $25 billion by 2030, with a compound annual growth rate (CAGR) of approximately 12% from 2023 to 2030 [1].
Key Market Segments:
- United States: Largest market, accounting for roughly 50% of global revenue. Estimated revenue in 2023: $6.2 billion.
- Europe: Next largest, with about 25% share, estimated at $3.1 billion.
- Asia-Pacific: Rapid growth, 20% market share, $2.5 billion expected in 2023.
- Rest of World: Remaining 5%, approximately $0.6 billion.
Market Drivers:
- Increasing prevalence of obesity and type 2 diabetes.
- Growing awareness and improved detection methods.
- Expanding pipeline of NASH therapies, including ANTARA.
What Are the Competitive Landscape and ANTARA's Position?
The NASH market features few approved drugs. Options include:
- Ocaliva (obeticholic acid): Marketed by Intercept, estimated global sales of $480 million in 2022.
- Aramchol: Phase 3 candidate, with promising early data.
- Resmetirom: Phase 3, with projected 2023 sales of $200 million.
ANTARA is positioned as a differentiated therapy with a novel mechanism targeting fibrosis and inflammation, potentially offering higher efficacy in advanced NASH stages.
What Are the Sales Projections for ANTARA?
Sales forecasts depend on regulatory approval timing, market penetration, and payer coverage.
| Year |
Projected Global Sales |
Assumptions |
| 2024 |
$120 million |
U.S. launch begins, initial adoption in Europe |
| 2025 |
$400 million |
Expansion in the U.S. and Europe, increased prescriber confidence |
| 2026 |
$700 million |
Broader indication approvals, inclusion in treatment guidelines |
| 2027 |
$1.2 billion |
Market penetration peaks, payer reimbursement solidifies |
| 2030 |
$3 billion |
Expansion into Asia-Pacific, second-line use in advanced cases |
Key Variables Influencing Sales:
- Regulatory outcomes: FDA approval is anticipated in 2023 Q4; delay could postpone launches.
- Market penetration: Estimated initial capture at 10% of the phase 3 target population.
- Pricing: Estimated at $20,000 per year per patient, consistent with current NASH therapies.
- Patient adoption: Growth from early adopters to broader clinical acceptance over three years.
What Are the Risks and Challenges?
- Regulatory approval: Pending FDA review and potential for additional data requests.
- Market competition: Existing drugs and pipeline candidates may affect uptake.
- Pricing and reimbursement: Payers may negotiate discounts, impacting revenue.
- Clinical efficacy: Real-world performance must align with trial data to sustain sales.
Key Market Entry Strategies
- Early engagement with payers for favorable coverage.
- Demonstrating clear differentiation in efficacy and safety.
- Building awareness among specialists such as hepatologists and endocrinologists.
- Strategic partnerships for manufacturing and distribution, especially in Asia.
Key Takeaways
- The global NASH market is set to triple by 2030, reaching $75 billion.
- ANTARA aims to secure regulatory approval by late 2023, with commercialization starting in early 2024.
- Sales projections suggest a potential revenue of approximately $3 billion by 2030.
- Market success depends on regulatory outcomes, competitive dynamics, and payer acceptance.
- Early market entry and differentiated positioning are critical to capture market share.
FAQs
-
When is ANTARA expected to gain FDA approval?
Approval is anticipated in late 2023, subject to review outcomes.
-
What is the estimated price per patient for ANTARA?
Approximately $20,000 annually per patient.
-
How does ANTARA compare to existing NASH therapies?
It offers a novel mechanism targeting fibrosis and inflammation, potentially providing better efficacy in advanced stages.
-
What is the timeline for ANTARA’s market penetration?
Initial adoption is expected in 2024, with full market penetration anticipated to reach peak levels by 2027.
-
What are the main risks associated with ANTARA's commercialization?
Regulatory delays, competition, payer reimbursement challenges, and real-world efficacy variations.
References
[1] MarketResearch.com. (2023). NASH therapeutics market report.