Share This Page
Drug Sales Trends for ketoprofen
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ketoprofen (2013)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
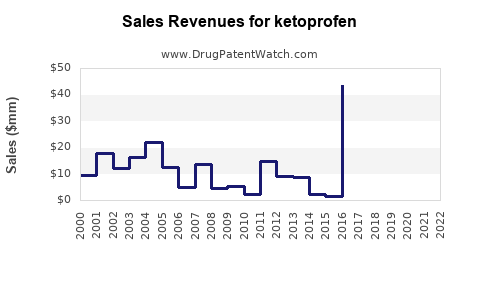
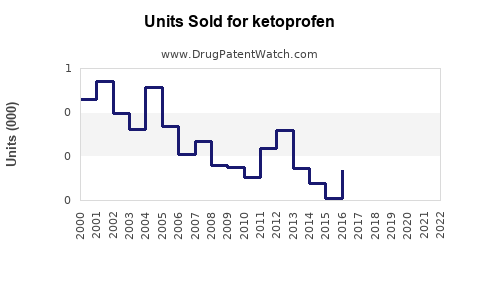
Annual Sales Revenues and Units Sold for ketoprofen
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| KETOPROFEN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| KETOPROFEN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| KETOPROFEN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| KETOPROFEN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| KETOPROFEN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| KETOPROFEN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| KETOPROFEN | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Ketoprofen Market Analysis and Sales Projections
Ketoprofen, a non-steroidal anti-inflammatory drug (NSAID), presents a mature market with stable demand driven by its efficacy in pain and inflammation management. Global sales are projected to grow at a compound annual growth rate (CAGR) of 2.5% from 2024 to 2030, reaching an estimated \$750 million. Growth will be primarily fueled by an aging global population experiencing increased rates of musculoskeletal disorders and continued adoption in emerging markets. Patent expirations for key formulations have led to a highly genericized landscape, intensifying price competition and limiting significant revenue growth from novel product introductions.
What is the Current Global Market Size for Ketoprofen?
The global market for ketoprofen was valued at approximately \$650 million in 2023. This figure encompasses sales of ketoprofen in various formulations, including oral tablets, capsules, topical gels, and injectable solutions, across both prescription and over-the-counter (OTC) segments. The market is characterized by a significant presence of generic manufacturers, contributing to its moderate valuation.
Which Indications Primarily Drive Ketoprofen Sales?
Ketoprofen's primary applications are in the management of pain and inflammation associated with a range of conditions. Key indications contributing to its sales volume include:
- Osteoarthritis: A significant driver, particularly in older demographics.
- Rheumatoid Arthritis: Chronic inflammatory condition requiring long-term pain management.
- Ankylosing Spondylitis: Inflammatory disease affecting the spine.
- Acute Pain: Relief from post-operative pain, dental pain, and musculoskeletal injuries.
- Dysmenorrhea: Management of menstrual cramps.
What is the Competitive Landscape for Ketoprofen?
The ketoprofen market is highly competitive and largely genericized. Key characteristics of the competitive landscape include:
- Dominance of Generic Manufacturers: The majority of ketoprofen sales are from generic products. Major generic drug manufacturers with significant market share include Teva Pharmaceutical Industries, Mylan (now Viatris), Sanofi, and Sandoz.
- Price Sensitivity: Intense competition among generic players leads to significant price pressure, impacting overall market revenue.
- Limited Branded Market: Branded ketoprofen products, such as Orudis® and Oruvail®, have faced extensive generic erosion following patent expirations.
- Regional Variations: Market dynamics can vary by region due to differences in regulatory approvals, healthcare policies, and prescription patterns.
Major Ketoprofen Manufacturers by Estimated Market Share (2023)
| Manufacturer | Estimated Market Share (%) |
|---|---|
| Teva Pharmaceutical Industries | 15-20 |
| Viatris Inc. | 12-17 |
| Sanofi | 8-12 |
| Sandoz (Novartis) | 7-10 |
| Others | 30-45 |
Note: Market share estimates are based on publicly available sales data and industry reports. "Others" includes a multitude of smaller generic manufacturers.
What are the Key Growth Drivers for Ketoprofen?
Despite its maturity, several factors are expected to sustain modest growth in the ketoprofen market:
- Aging Global Population: The increasing prevalence of age-related conditions like osteoarthritis and chronic pain, particularly in developed economies, will maintain a consistent demand for effective pain relievers. The World Health Organization projects the number of people aged 60 and over to more than double by 2050 [1].
- Rising Healthcare Expenditure in Emerging Markets: As healthcare infrastructure improves and disposable incomes rise in countries across Asia, Latin America, and Africa, access to and demand for established pain management medications like ketoprofen is expected to increase.
- Availability of Cost-Effective Formulations: The widespread availability of affordable generic ketoprofen makes it a preferred choice for healthcare providers and patients managing chronic pain conditions where long-term treatment is necessary.
- Continued Use in Topical Formulations: Topical ketoprofen gels and creams offer localized pain relief with potentially fewer systemic side effects, maintaining their utility for musculoskeletal pain and sprains.
What are the Restraining Factors for Ketoprofen Market Growth?
Several factors are limiting more robust growth for ketoprofen:
- Intensified Generic Competition: The commoditized nature of the ketoprofen market, driven by a high number of generic competitors, exerts downward pressure on prices and profit margins.
- Availability of Newer NSAIDs and Alternative Therapies: The development of newer NSAIDs with improved safety profiles or novel mechanisms of action, as well as the increasing use of biologic therapies and non-pharmacological treatments for chronic pain, present competitive alternatives.
- Adverse Event Profile: Like other NSAIDs, ketoprofen carries risks of gastrointestinal bleeding, cardiovascular events, and renal complications, particularly with long-term use or in susceptible patient populations. This can lead to increased caution in prescribing and a preference for agents with perceived better safety profiles.
- Patent Expirations and Lack of New Drug Applications: Major patents for ketoprofen have expired, and there is limited investment in novel drug discovery or significant pipeline development for ketoprofen itself. The focus is on efficient manufacturing and distribution of existing generic versions.
What are the Projected Sales Figures for Ketoprofen?
Based on current market trends, market penetration, and projected CAGR, ketoprofen sales are estimated to evolve as follows:
- 2024: \$660 million
- 2025: \$675 million
- 2026: \$690 million
- 2027: \$705 million
- 2028: \$720 million
- 2029: \$735 million
- 2030: \$750 million
These projections assume a steady CAGR of 2.5% and are contingent on continued demand from key indications and market access in emerging regions.
What is the Patent Landscape for Ketoprofen?
The patent landscape for ketoprofen is largely characterized by expired primary patents. The original patents for ketoprofen itself have long since expired, allowing for widespread generic manufacturing. While some patents may exist for specific novel formulations, delivery systems, or combination therapies involving ketoprofen, these are not expected to significantly alter the core market dynamics of the unpatented active pharmaceutical ingredient.
- Original Compound Patents: Expired.
- Formulation Patents: Some expired or nearing expiration; focus on extended-release or specialized delivery systems may have seen limited, localized patent protection.
- Manufacturing Process Patents: Potential for ongoing protection of specific, efficient manufacturing methods, but not typically a barrier to generic entry for the API itself.
The lack of significant new patent filings for ketoprofen underscores its status as a well-established, off-patent drug.
What are the Regulatory Considerations for Ketoprofen?
Ketoprofen is subject to standard pharmaceutical regulations globally, which include:
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality, safety, and efficacy. Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) conduct inspections to ensure compliance.
- Marketing Authorization: Generic versions require Abbreviated New Drug Applications (ANDAs) in the U.S. or similar marketing authorization procedures in other regions, demonstrating bioequivalence to the reference listed drug.
- Pharmacovigilance: Post-market surveillance for adverse events is a continuous requirement. Regulatory agencies monitor safety data and can implement label changes or market restrictions if significant safety concerns arise.
- OTC vs. Prescription Status: In many countries, ketoprofen is available both by prescription for more severe conditions and as an OTC medication for mild to moderate pain and inflammation, with varying dosage limits and indications. For instance, in the UK, 25mg strengths are available OTC, while higher strengths require a prescription.
What are the Key Takeaways?
- Mature Market: Ketoprofen operates in a mature, highly competitive, and genericized market.
- Stable Demand: Demand is underpinned by established indications, particularly osteoarthritis and general pain management.
- Modest Growth: Projections indicate a CAGR of 2.5%, reaching approximately \$750 million by 2030, driven by aging populations and emerging market expansion.
- Price Pressure: Intense generic competition limits revenue growth and emphasizes cost-efficiency in manufacturing.
- Limited Innovation: The patent landscape is characterized by expired primary patents, with minimal investment in novel ketoprofen development.
- Safety Profile: Known NSAID-related risks necessitate careful prescribing and can lead to competition from drugs with perceived superior safety profiles.
Frequently Asked Questions
1. What is the primary reason for the limited revenue growth in the ketoprofen market?
The primary reason is the extensive genericization of the market. With most key patents expired, numerous manufacturers produce ketoprofen, leading to intense price competition and commoditization of the product.
2. How does the aging global population specifically impact ketoprofen sales?
An aging population experiences a higher incidence of musculoskeletal disorders, such as osteoarthritis and rheumatoid arthritis. These conditions often require chronic pain management, leading to sustained and increased demand for effective, affordable analgesics like ketoprofen.
3. Are there any significant new therapeutic applications for ketoprofen being developed?
Currently, there are no major new therapeutic applications for ketoprofen under active development that are expected to significantly alter its market trajectory. The focus remains on its established indications.
4. What is the difference between prescription and OTC ketoprofen?
The primary difference lies in dosage strength and approved indications. Higher strengths and formulations for more severe or chronic conditions typically require a prescription, while lower strengths for mild to moderate pain and inflammation are available over-the-counter in many regions.
5. What are the main safety concerns associated with ketoprofen use?
Like other NSAIDs, ketoprofen carries risks of gastrointestinal adverse events (e.g., ulcers, bleeding), cardiovascular complications (e.g., heart attack, stroke), and renal impairment. These risks can be dose-dependent and are more pronounced in patients with pre-existing conditions or those on long-term therapy.
Citations
[1] World Health Organization. (2022). Global report on ageing and health. Geneva: World Health Organization.
More… ↓
