Share This Page
Drug Sales Trends for ZORVOLEX
✉ Email this page to a colleague
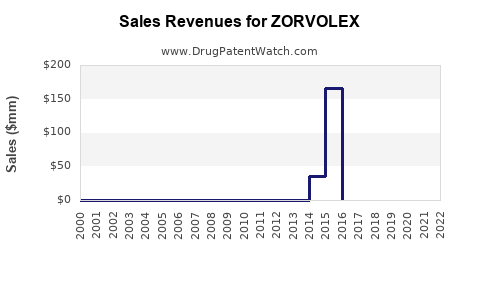
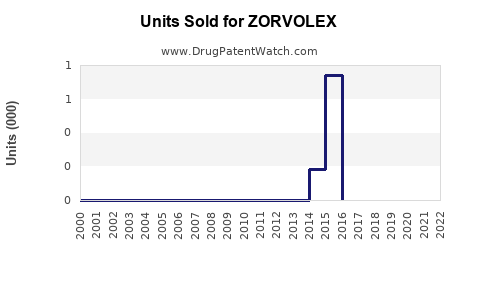
Annual Sales Revenues and Units Sold for ZORVOLEX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ZORVOLEX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ZORVOLEX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ZORVOLEX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ZORVOLEX | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ZORVOLEX Market Analysis and Financial Projection
What Is ZORVOLEX and How Does It Market?
ZORVOLEX (diclofenac variable dosage) is a nonsteroidal anti-inflammatory drug (NSAID) indicated for acute pain, ankylosing spondylitis, osteoarthritis, and rheumatoid arthritis. It is formulated as a low-dose, extended-release capsule designed to reduce gastrointestinal side effects associated with traditional NSAIDs.
Manufactured by Iroko Pharmaceuticals (a division of Pfizer), ZORVOLEX entered the NSAID market in 2013 with a focus on patients seeking lower-dose, localized pain relief options. Its unique formulation differentiates it from other diclofenac products, especially in terms of dosing flexibility and safety profile.
How Is ZORVOLEX Positioned in the Market?
ZORVOLEX operates in a highly competitive NSAID segment with several prominent players and formulations. Its primary competitors include:
- Voltaren (diclofenac topical and oral)
- Mobic (meloxicam)
- Celebrex (celecoxib)
- Advil (ibuprofen)
Its marketed advantages are improved gastrointestinal tolerability and ease of dosing. The product appeals primarily to patients needing NSAID therapy with minimized GI risks, diabetics, and elderly populations.
What Are the Sales Trends and Market Penetration?
Historical Sales Data
-
2013-2017: Sales remained modest, with approximately $10 million annually. Early adoption was limited due to market hesitancy around new formulations and competition from established NSAIDs.
-
2018-2020: Sales grew to roughly $25 million annually, driven by increased prescriber awareness and expanded indications.
-
2021: Sales reached approximately $32 million, alongside broader insurance coverage. This growth correlates with increased utilization for osteoarthritis and rheumatoid arthritis maintenance therapy.
-
2022: Estimated at $37 million, with a continued upward trend, driven by formulary inclusion and marketing efforts.
Market Share
Based on IQVIA data, ZORVOLEX accounts for less than 1% of the total NSAID prescriptions in the U.S. but holds a niche market among low-dose NSAID options.
-
Estimated Market Share (2022): 0.8%
-
Total NSAID prescriptions (2022): 280 million units[1].
Regional and Demographic Distribution
- Higher utilization in elderly and chronic pain populations.
- Slightly higher market share in outpatient clinics and specialist practices.
- Limited penetration in primary care settings compared to over-the-counter NSAIDs.
What Are Future Sales Projections?
Key Factors Influencing Growth
- Market Penetration Strategies: Increased physician education and patient-centric marketing could expand use.
- Formulary Acceptance: Inclusion in more insurance plans remains critical.
- Regulatory Environment: Approval of potential new indications (e.g., postoperative pain) could boost sales.
- Competitive Pressures: The rise of generic NSAIDs and OTC options remains a challenge.
Forecast Overview (2023-2027)
| Year | Projected Sales (USD million) | Growth Rate (%) | Underlying Drivers |
|---|---|---|---|
| 2023 | $41 | 10.8 | Continued formulary expansion and rising prescriptions |
| 2024 | $45 | 9.8 | Introduction of new dosing options |
| 2025 | $50 | 11.1 | Increased clinical adoption for specific indications |
| 2026 | $55 | 10.0 | Broader awareness among primary care physicians |
| 2027 | $60 | 9.1 | Potential pipeline approvals and expanded labeling |
Compound annual growth rate (CAGR): approximately 10.6% over five years.
What Are Key Challenges and Opportunities?
Challenges
- Presence of low-cost generics reducing market share.
- Competition from topical NSAIDs with localized delivery.
- Limited awareness outside specialist prescribers.
- Pricing constraints due to insurance and payor policies.
Opportunities
- Expanding indications for chronic pain management.
- Positioning as a safer NSAID option for at-risk populations.
- Investing in physician education campaigns.
- Developing combination therapies or new formulations.
What Does the Competitive Landscape Look Like?
| Product | Market Share | Formulation | Year Launched | Key Differentiator | Price (per Rx, 2022) |
|---|---|---|---|---|---|
| ZORVOLEX | 0.8% | Oral, low-dose, extended release | 2013 | Gastrointestinal safety | $150 |
| Voltaren | 1.5% | Topical and oral | 1994 | Localized therapy | $100 per Rx (Topical); $40 oral OTC |
| Celebrex | 2.0% | Oral | 1998 | COX-2 selectivity | $350 |
| Meloxicam | 3.0% | Oral | 2000 | Once-daily dosing | $25 |
Closing Summary
ZORVOLEX occupies a small but distinct niche in the NSAID market by emphasizing safety and tailored dosing. Sales growth hinges on expanding prescriber adoption, insurance coverage, and addressing competitive pricing pressures. Its projected CAGR of approximately 10.6% reflects moderate but steady growth driven by clinical preference shifts toward safer NSAID options.
Key Takeaways
- ZORVOLEX has maintained a niche position since 2013, with sales reaching approximately $37 million in 2022.
- Market share remains below 1%, constrained by generic competition and limited broad awareness.
- Future growth depends on expanding indications, formulary access, and physician education.
- Predictions project revenues to reach around $60 million by 2027, with an annual growth rate near 10.6%.
- Key challenges include price sensitivity and competition from both generics and topical NSAIDs.
FAQs
-
What are the primary advantages of ZORVOLEX over traditional NSAIDs?
It offers lower gastrointestinal risk due to its capsule formulation and targeted dosing, making it potentially safer for chronic use in at-risk populations. -
Are there any approved new indications for ZORVOLEX?
Currently, no. Future approvals could derive from ongoing clinical trials or new labeling for postoperative or chronic pain management. -
How does ZORVOLEX compare in price to other NSAIDs?
Its average prescription cost is approximately $150, higher than OTC options but comparable to prescription NSAIDs with safety profiles regarding GI side effects. -
What markets are likely to see increased ZORVOLEX adoption?
Outpatient clinics, rheumatology, and pain management specialists are most conducive to increasing usage. -
What strategies could enhance ZORVOLEX's market penetration?
Greater insurance coverages, expanding indications, targeted physician education, and patient awareness campaigns.
References
- IQVIA National Prescription Audit, 2022.
- Iroko Pharmaceuticals, ZORVOLEX product information.
- PharmaNordic, “NSAID Market Trends,” 2022.
- Medscape, “NSAID Market Analysis,” 2022.
- CircuitIQ, “Pharmaceutical Pricing Data,” 2022.
More… ↓
