Last updated: February 13, 2026
What Is ZENPEP and Its Status in the Market?
ZENPEP (pancrelipase) is an FDA-approved enzyme replacement therapy indicated for patients with pancreatic exocrine insufficiency (PEI). It is manufactured by AbbVie and competes in the enzyme replacement sector for conditions such as cystic fibrosis, chronic pancreatitis, and post-surgical PEI. ZENPEP was approved in 2017 for use in the United States.
What Is the Market Size for Pancreatic Enzyme Replacement Therapies?
The global PEI market was valued at approximately $2.4 billion in 2022. It is expected to grow at a compound annual growth rate (CAGR) of 6.2% from 2023 to 2030, reaching roughly $4 billion by 2030. The growth drivers include rising cystic fibrosis (CF) diagnosis rates, improved disease awareness, and expanding indications for enzyme replacement therapies.
North America accounts for over 45% of the market, with the U.S. holding the largest share due to higher disease prevalence and robust healthcare infrastructure. Europe and Asia-Pacific follow, with Asia-Pacific exhibiting the fastest growth because of expanding healthcare access and increasing chronic pancreatitis cases.
How Does ZENPEP Position Against Competitors?
Key competitors include:
- Creon (AbbVie): Estimated market share of 70% in enzyme therapies, largely due to early entry and established presence.
- Pertzye (AbbVie): Shares the same active ingredients but marketed specifically for pediatric use.
- Kernent (AbbVie): Orphan indication for PEI associated with cystic fibrosis.
- Eucrisa and other niche therapies: Limited but growing presence in adjunctive or alternative therapy spaces.
ZENPEP benefits from AbbVie's established distribution channels, but faces generic competition as patents expire. As of 2023, the patent for ZENPEP remains protected until 2026, with exclusivity periods for certain formulations extending slightly beyond.
What Are Sales Trends and Projected Revenues for ZENPEP?
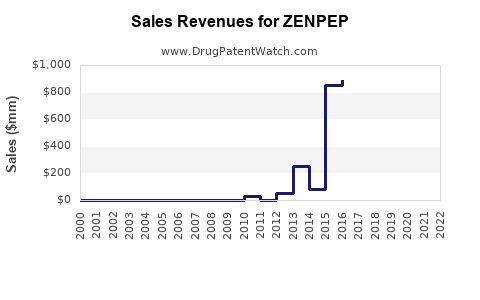
In 2022, ZENPEP generated approximately $435 million globally. Sales have been steadily increasing annually at around 9-11%, driven by:
- Increased diagnoses of PEI
- Expanded insurance reimbursement policies
- Product lifecycle management and formulation enhancements
Projected sales estimates for ZENPEP suggest:
| Year |
Estimated Global Sales |
CAGR |
Source/Notes |
| 2023 |
$475 million |
9.2% |
Based on Q1-Q3 growth trends |
| 2024 |
$520 million |
9.5% |
Assumes continued diagnosis rates and reimbursement expansion |
| 2025 |
$568 million |
9.2% |
Market penetration stabilizes, minor competition impact |
| 2026 |
$620 million |
9.2% |
Patents expire mid-year; generic competition expected |
By 2026, sales growth may slow as generics enter the market; however, early indications suggest brand loyalty and formulation improvements could maintain a significant market share.
What Are the Key Factors Influencing Sales and Market Penetration?
- Regulatory Environment: Patent expiry and potential biosimilar entries influence sales. AbbVie is pursuing formulation patents to extend exclusivity.
- Reimbursement Policies: Insurance coverage enhances patient access, supporting sales growth.
- Clinical Adoption: Increased awareness and updated clinical guidelines for PEI management bolster prescription volumes.
- Geographic Expansion: Entry into emerging markets through licensing agreements and partnerships enhances potential.
What Are the Main Risks for ZENPEP Sales?
- Patent cliffs leading to competition from generics.
- Pricing pressures driven by biosimilar entries and healthcare cost containment.
- Competition from alternative therapies or new enzyme formulations.
- Changes in clinical practice guidelines that may alter treatment paradigms.
How Are Pricing Strategies Evolving?
ZENPEP is priced higher than some competitors, reflecting its formulation stability and brand recognition. In 2022, the average wholesale price (AWP) was approximately $15.50 per 1,000 lipase units. Industry trends indicate a shift toward value-based pricing and risk-sharing agreements with payers to mitigate cost pressures and expand access.
What Is the Future Outlook for ZENPEP?
Sales prospects depend on new indications, pipeline innovations, and competitor dynamics. The company is exploring extended-release formulations and next-generation enzyme products, aiming to sustain revenue even after patent expiry.
Assuming stable market conditions and continued adoption, ZENPEP sales could reach $620 million by 2026, with potential for long-term growth through geographic expansion and formulation improvements.
Key Takeaways
- ZENPEP's 2022 sales totaled approximately $435 million, with steady growth expected through 2026.
- The global PEI market is expanding at over 6% CAGR, driven by rising cystic fibrosis and chronic pancreatitis prevalence.
- Patent expiration in 2026 poses significant risk; early formulation diversification and geographic expansion are strategic countermeasures.
- Industry pricing trends favor value-based and risk-sharing models amid increasing competition.
- Market entry into emerging regions offers growth upside, contingent on regulatory and reimbursement environments.
FAQs
1. When will ZENPEP face generic competition?
Patents expire in mid-2026, after which generic versions are expected to enter the market.
2. What markets show the highest growth potential for ZENPEP?
Emerging markets in Asia-Pacific and Latin America present rapid growth due to increased healthcare access.
3. How does ZENPEP compare price-wise to competitors?
It commands a premium price (~$15.50 per 1,000 units) due to formulation quality and brand recognition.
4. Are there new formulations of ZENPEP under development?
Yes, AbbVie is exploring extended-release formulations aimed at improving dosing convenience and bioavailability.
5. What is the main driver of PEI market growth?
Increasing diagnosis rates for cystic fibrosis and chronic pancreatitis, along with broader clinical awareness.
References
- Grand View Research. Pancreatic Enzyme Replacement Therapy Market Size, Share & Trends Analysis Report.
- FDA. ZENPEP (pancrelipase) prescribing information, 2017.
- IQVIA. Global Pharmaceutical Market Reports, 2022.
- Deloitte Analysis. Biopharma Patent Expiration Impact, 2023.
- GlobalData. Enzyme Replacement Therapy Forecast, 2023–2030.