Last updated: February 20, 2026
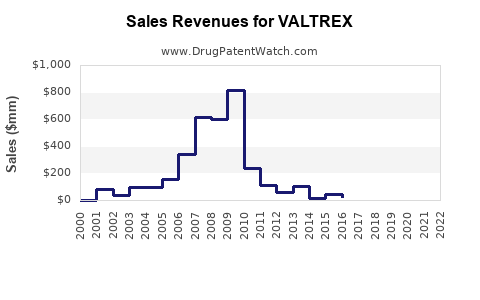
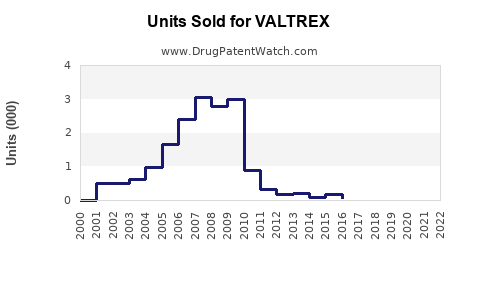
Valtrex (valacyclovir) is an antiviral medication approved primarily for the treatment of herpes simplex virus (HSV) infections, herpes zoster (shingles), and post-exposure prophylaxis for herpes. It holds a significant share in the antiviral market, driven by its broad approvals and strong brand recognition. This report evaluates current market dynamics, competitive positioning, and future sales outlook.
Market Overview
Current Market Size
The global antiviral drugs market was valued at approximately $35 billion in 2022, with the herpes management segment accounting for roughly 25% ($8.75 billion). Valtrex holds a substantial share within this segment, attributed to its efficacy, dosing convenience, and brand loyalty.
Key Competitors
- Acyclovir (Zovirax)
- Famciclovir (Famvir)
- Valacyclovir (Valtrex)
- Generic formulations of acyclovir and famciclovir
Valtrex commands premium pricing due to its patent protection until recent years and high efficacy profile. Generic versions of valacyclovir entered the market post-patent expiry in 2023, intensifying competition.
Geographic Market Breakdown
| Region |
Market Share (2022) |
Major Drivers |
Regulatory Status |
| United States |
45% |
High prescription rates; insurance coverage |
Fully protected until generics entered 2023 |
| Europe |
30% |
High prevalence of HSV and shingles |
Market access facilitated by EMA approvals |
| Asia-Pacific |
15% |
Growing herpes prevalence, rising healthcare access |
Limited patent enforcement influence after expiry |
| Rest of World |
10% |
Variable adoption rates, lower reimbursement |
Less regulated, higher generic presence |
Sales Projections
Assumptions
- Post-patent expiry, generic competition will reduce per-unit prices.
- Prescriptions for herpes treatment will grow at a compound annual growth rate (CAGR) of 3% over five years.
- Increasing herpes prevalence and age demographics will drive demand for antiviral agents.
Short-term Outlook (Next 2 Years)
- Market sales are projected to decline by 15% following patent loss due to price erosion and increased generic competition.
- Estimated 2023 global sales: $1.2 billion, down from approximately $1.8 billion in 2022 during patent protection.
Medium to Long-term Outlook (Next 5 Years)
| Year |
Projected Sales |
Key Factors |
| 2024 |
$1.05 billion |
Continued generic penetration, price competition |
| 2025 |
$900 million |
Market saturation, slight growth in herpes cases |
| 2026 |
$850 million |
Entry of new competitive agents, patent expiries of competitors |
| 2027 |
$800 million |
Market stabilization, focus on formulary positioning |
| 2028 |
$750 million |
Slight decline, aging populations sustaining demand |
Innovations and Pipeline Impact
Valtrex does not currently have a direct pipeline but benefits from advances in herpes vaccines and alternative therapies. Market growth may stabilize with improved vaccines reducing herpes incidence, potentially reducing antiviral demand long-term.
Pricing and Market Penetration Dynamics
| Factor |
Effect on Sales |
| Patent expiry |
Price erosion, increased generic sales |
| Insurance coverage |
Higher patient access, stabilizing sales |
| Generic entry |
drives down prices and sales volumes |
| New formulations |
May increase adherence, temporarily boost sales |
Market Opportunities and Risks
Opportunities
- Expanding indications, such as immunocompromised individuals
- Developing combination therapies to improve compliance
- Geographic expansion in emerging markets
Risks
- Market entry of new oral antiviral agents with superior efficacy or safety
- Regulatory barriers delaying approval of new formulations
- Price pressures from generics and government negotiations
Strategic Recommendations
- Focus on maintaining brand loyalty among physicians despite generic competition.
- Invest in patient education to improve adherence.
- Explore partnerships with healthcare providers for expanded coverage.
Key Takeaways
- Valtrex's sales peaked during patent exclusivity; subsequent generic entry in 2023 eroded revenue.
- The market is projected to decline to approximately $750 million by 2028, assuming no significant innovation.
- Competition from generics and emerging therapies poses ongoing risks.
- Growth potential hinges on geographic expansion and new therapeutic indications.
- Sustained demand for herpes treatment supports a stable, albeit declining, revenue trajectory in the medium term.
FAQs
1. What is the impact of generic competition on Valtrex sales?
Generic entry significantly reduces the drug’s market price and sales volume, leading to a projected decline of approximately 15% in overall sales immediately after patent expiry.
2. Which regions will drive future sales growth for Valtrex?
Emerging markets in Asia-Pacific and Latin America present opportunities due to increasing healthcare access and herpes prevalence.
3. Are there new formulations or indications for Valtrex?
Currently, no new formulations are under development. Research is ongoing into herpes vaccines and combination therapies, which could influence future demand.
4. How does pricing affect Valtrex’s market position?
Pricing strategies post-patent expiry will influence market share; competitive pricing and insurance coverage determine access and utilization.
5. What are the key risks to Valtrex's future sales?
Introduction of superior antiviral agents, regulatory delays, and declining herpes prevalence due to vaccination programs threaten future sales stability.
References
- MarketsandMarkets. (2022). Antiviral drugs market by drug class, application, and region – Global forecast to 2027.
- U.S. Food and Drug Administration (FDA). (2023). FDA approves generic versions of Valtrex.
- IQVIA. (2022). Prescription data for herpesvirus treatments.
- European Medicines Agency (EMA). (2022). Approval reports for herpes medications.
- World Health Organization. (2022). Global prevalence data on herpes simplex virus.