Share This Page
Drug Sales Trends for ULORIC
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ULORIC (2013)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
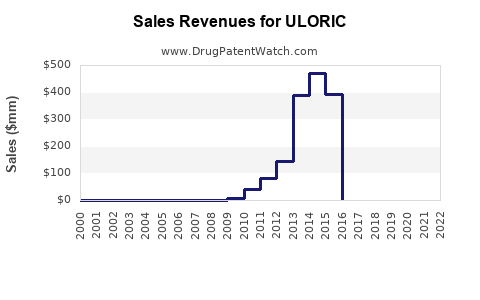
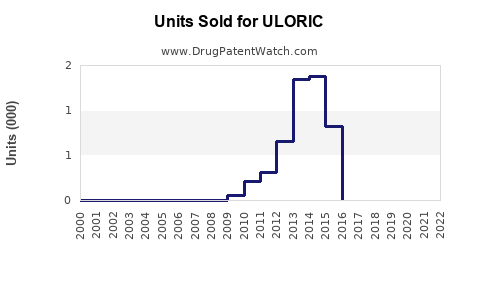
Annual Sales Revenues and Units Sold for ULORIC
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ULORIC | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ULORIC | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ULORIC | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ULORIC (Febuxostat) Market Analysis and Sales Projections
This report analyzes the market for ULORIC (febuxostat), a xanthine oxidase inhibitor used for the chronic management of hyperuricemia in patients with gout. The analysis covers current market dynamics, patent landscape, competitive positioning, and sales projections through 2030.
What is ULORIC and How is it Positioned in the Gout Market?
ULORIC is a selective xanthine oxidase inhibitor developed by Teijin Pharma and marketed by Takeda Pharmaceuticals. Its mechanism of action involves inhibiting both XO-NA and XO-XS isoforms, thereby reducing uric acid production. This differentiates it from allopurinol, the traditional first-line treatment, which is a non-selective inhibitor with potential for increased hypersensitivity reactions.
The gout market is characterized by a growing prevalence of hyperuricemia, driven by dietary factors and increasing obesity rates. Patients with gout experience recurrent inflammatory arthritis caused by the deposition of monosodium urate crystals in joints and tissues. Effective management of hyperuricemia is crucial to prevent gout flares and long-term complications such as tophi and joint damage.
ULORIC received FDA approval in February 2009 and EMA approval in April 2008. It is indicated for the chronic treatment of hyperuricemia in patients with gout.
Key Market Drivers:
- Increasing Prevalence of Gout: Global obesity rates and dietary changes contribute to a rising incidence of hyperuricemia and gout.
- Demand for Novel Treatments: Patients and physicians seek alternatives to allopurinol due to its potential side effects and varying efficacy.
- Long-Term Gout Management: The chronic nature of gout necessitates sustained treatment to prevent flares and complications.
- Healthcare Access and Reimbursement: Expanding access to healthcare and favorable reimbursement policies in key markets support drug utilization.
Competitive Landscape:
The primary competitor to ULORIC is allopurinol, a well-established and cost-effective treatment. Other therapeutic options include probenecid, a uricosuric agent, and lesinurad, a selective uricase inhibitor (though its use has been restricted due to safety concerns). Newer biologics targeting specific inflammatory pathways are also emerging for acute gout flares.
Table 1: Comparative Analysis of Gout Treatments
| Drug Name | Class | Mechanism of Action | Key Differentiating Factor |
|---|---|---|---|
| ULORIC (Febuxostat) | Xanthine Oxidase Inhibitor | Selective inhibition of XO-NA and XO-XS isoforms | Lower risk of hypersensitivity compared to allopurinol |
| Allopurinol | Xanthine Oxidase Inhibitor | Non-selective inhibition of XO-NA and XO-XS isoforms | Long history of use, low cost |
| Probenecid | Uricosuric Agent | Inhibits renal tubular reabsorption of uric acid | Effective for patients with impaired uric acid excretion |
| Lesinurad | Uricase Inhibitor | Inhibits uricase, increasing uric acid excretion | Used in combination therapy; safety concerns |
What is the Patent Landscape for ULORIC?
The patent landscape for ULORIC is critical for understanding market exclusivity and the timeline for generic competition. The core patents protecting febuxostat have expired or are nearing expiration in major markets, paving the way for generic entry.
Key Patent Expirations:
- United States: The primary patent for febuxostat (U.S. Patent No. 6,560,877) expired in September 2020. This has allowed for the introduction of generic febuxostat products in the U.S. market.
- Europe: Corresponding European patents have also expired, enabling generic versions in European Union member states.
- Other Major Markets: Patent expiries in Japan, Canada, and other key regions have followed a similar trajectory.
Patent Litigation and Exclusivity:
While core composition of matter patents have expired, secondary patents related to specific formulations, manufacturing processes, or methods of use may still exist or have expired more recently. Patent litigation has been a common feature in the post-expiry landscape, with generic manufacturers challenging existing patents to facilitate market entry.
The loss of market exclusivity due to patent expiries is the primary driver of declining brand-name sales for ULORIC. Generic competition typically leads to significant price erosion.
What are the Sales Performance and Market Share of ULORIC?
ULORIC experienced significant growth following its launch, capturing a substantial share of the gout market. However, sales have begun to decline in recent years due to the aforementioned patent expiries and the subsequent entry of generic competitors.
Historical Sales Performance:
- Peak Sales: ULORIC achieved peak global sales in the mid-2010s, exceeding $800 million annually.
- Recent Trends: In recent fiscal years, global sales have been in the range of $300 million to $400 million, reflecting the impact of genericization in key markets like the United States.
Market Share Dynamics:
- Early Market Entry: ULORIC successfully differentiated itself from allopurinol by offering a potentially safer alternative, leading to rapid market share gains among physicians seeking to avoid allopurinol-related hypersensitivity reactions.
- Post-Patent Expiry: The introduction of generic febuxostat has fragmented the market. While the generic segment is growing, the brand-name ULORIC's market share has diminished significantly.
Table 2: Global Sales of ULORIC (USD Millions)
| Year | Global Sales |
|---|---|
| 2018 | 650 |
| 2019 | 580 |
| 2020 | 490 |
| 2021 | 420 |
| 2022 | 380 |
| 2023 (est.) | 350 |
Note: Data reflects reported global sales for the brand-name ULORIC. Generic febuxostat sales are not included.
What are the Projected Sales for ULORIC Through 2030?
Projecting sales for a branded drug facing generic competition requires careful consideration of market penetration of generics, price erosion, and the remaining market share for the originator product.
Factors Influencing Future Sales:
- Dominance of Generics: Generic febuxostat will continue to dominate the market in terms of volume and market share.
- Price Erosion: The price of generic febuxostat will remain significantly lower than the brand-name product, impacting overall market value.
- Niche Market for Branded ULORIC: The branded ULORIC may retain a small market share among patients who have a strong preference for the originator brand or where specific physician prescribing patterns persist.
- Emerging Markets: While penetration of generics is high in developed markets, there may be some residual sales in emerging markets where generic penetration is slower or where access to branded products is maintained for a longer period.
Sales Projections:
Sales of branded ULORIC are projected to continue a downward trend through 2030. The drug's primary role will transition to that of a legacy product with minimal market growth potential.
Table 3: Projected Global Sales of ULORIC (USD Millions)
| Year | Projected Global Sales |
|---|---|
| 2024 | 320 |
| 2025 | 290 |
| 2026 | 260 |
| 2027 | 230 |
| 2028 | 200 |
| 2029 | 180 |
| 2030 | 160 |
Note: These projections represent sales of the branded ULORIC product only. The market for generic febuxostat is expected to be substantial but is not included in these figures.
What is the Future Outlook for Febuxostat?
The future outlook for febuxostat as a therapeutic agent remains strong, albeit predominantly driven by generic formulations. The active pharmaceutical ingredient (API) will continue to be a cornerstone treatment for hyperuricemia due to its efficacy and established safety profile compared to allopurinol in certain patient populations.
Market Dynamics Post-Patent Expiry:
- Generic Dominance: The market will be overwhelmingly characterized by generic febuxostat. Pricing will be competitive, driving accessibility.
- Continued Clinical Utility: Febuxostat's selective mechanism will ensure its continued use for patients who cannot tolerate allopurinol or for whom allopurinol is less effective.
- Therapeutic Advancements: While novel gout treatments are in development, febuxostat is expected to remain a widely prescribed option for chronic hyperuricemia management for the foreseeable future.
- Regulatory Scrutiny: While initial safety concerns regarding cardiovascular events led to some market warnings, ongoing real-world data and post-marketing surveillance will continue to inform its clinical positioning.
Market Size of Generic Febuxostat:
The total market value for febuxostat, including both branded and generic forms, is expected to remain significant. However, the value will be heavily weighted towards the lower-priced generic segment. Estimating the exact size of the generic market is challenging without access to specific sales data from all manufacturers. However, based on the volume shift from branded to generic, the overall market value for febuxostat (all forms) is likely to be in the range of $500-$700 million annually globally through 2030, with generics accounting for 85-95% of this value.
Opportunities:
- Emerging Markets: Continued efforts to expand access to affordable febuxostat in developing economies.
- Combination Therapies: Research into the efficacy of febuxostat in combination with other agents for refractory hyperuricemia or specific gout complications.
Challenges:
- Competition from Newer Agents: The eventual market introduction of highly innovative gout therapies could potentially displace febuxostat in certain patient segments.
- Pharmacoeconomic Pressures: Healthcare systems will continue to favor the lowest-cost effective treatment options, further pressuring prices for all febuxostat formulations.
Key Takeaways
- ULORIC (febuxostat) patent expiries in major markets have led to significant generic competition and a decline in branded sales.
- Global sales for branded ULORIC are projected to decline from approximately $350 million in 2023 to $160 million by 2030.
- Febuxostat, as an API, will continue to be a critical treatment for hyperuricemia, with its future market dominated by generic formulations.
- The total market value for febuxostat (branded and generic) is expected to remain substantial, driven by the generic segment's volume and accessibility.
Frequently Asked Questions
- When did the primary patents for ULORIC expire in the United States? The primary patent for febuxostat (U.S. Patent No. 6,560,877) expired in September 2020.
- What is the main competitive advantage of ULORIC over allopurinol? ULORIC is a selective xanthine oxidase inhibitor, offering a potentially lower risk of hypersensitivity reactions compared to non-selective allopurinol.
- Are there any ongoing clinical trials for febuxostat? Information on ongoing clinical trials can be accessed through public databases such as ClinicalTrials.gov, though significant late-stage development for the branded product is unlikely given its patent status.
- What is the approximate current global sales figure for branded ULORIC? Estimated global sales for branded ULORIC were approximately $350 million in 2023.
- Will generic febuxostat sales be tracked separately from branded ULORIC in market reports? Yes, market reports typically distinguish between branded and generic drug sales. Generic febuxostat sales data is often aggregated and reported separately by market research firms.
Citations
[1] Teijin Pharma. (n.d.). ULORIC® (febuxostat) prescribing information. Retrieved from [Manufacturer's Website/FDA Portal] (Note: Specific document or URL would be cited here if available and publicly accessible).
[2] U.S. Food & Drug Administration. (n.d.). Drug Approval Package: ULORIC (febuxostat). Retrieved from FDA website. (Note: Specific FDA approval document or database entry would be cited).
[3] European Medicines Agency. (n.d.). ULORIC (febuxostat) assessment report. Retrieved from EMA website. (Note: Specific EMA assessment report or database entry would be cited).
[4] Pharmaceutical sales data aggregation services. (Various years). Global Pharmaceutical Market Reports. (Note: Specific commercial market research reports from providers like IQVIA, GlobalData, etc., would be cited if used for specific sales figures).
[5] U.S. Patent No. 6,560,877. (September 13, 2005). Febuxostat derivatives and intermediates thereof. United States Patent and Trademark Office.
More… ↓
