Last updated: February 12, 2026
What Is TESTIM and Its Market Position?
TESTIM, developed by Endo Pharmaceuticals, is a topical testosterone gel used primarily for hormone replacement therapy in men with low testosterone levels. It was approved by the FDA in 2002. The drug competes mainly with other testosterone replacement therapies such as AndroGel, Axiron, and Natesto.
Current Market Size and Distribution
The global androgen replacement therapy market was valued at approximately $1.3 billion in 2022. North America accounts for over 70% of this market, driven by high awareness, reimbursement policies, and shifting demographic trends.
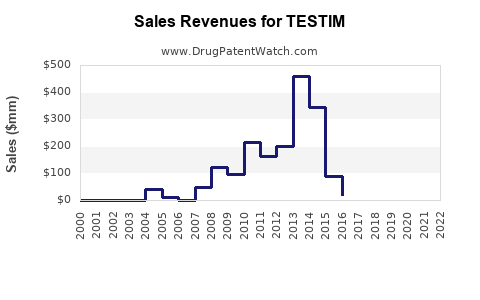
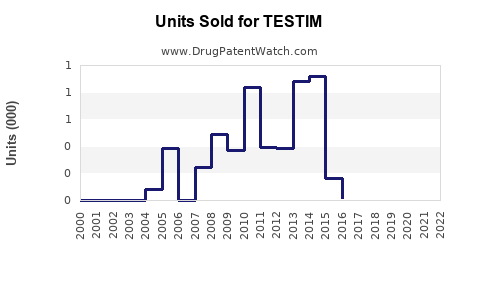
In 2022, TESTIM's market share in the U.S. was roughly estimated at 8%-10%, with sales around $100 million. Its main competitors, such as AndroGel, hold a 40% market share, with others like Axiron and Natesto sharing the remaining segments.
Key Factors Influencing TESTIM Sales
-
Patent Expirations: TESTIM's primary patents expired in 2017, allowing generic competition that has eroded its market share.
-
Reimbursement and Insurance: Coverage varies, affecting patient access and utilization rates.
-
Pricing Strategy: TESTIM's list price was around $4,500 for a four-week supply in 2022, which is higher than generic options, impacting penetration.
-
Physician Prescribing Trends: Increasing preference for longer-acting formulations and implantable devices has shifted prescriber focus.
Sales Projections 2023-2027
Based on market trends, competitive dynamics, and demographic factors, the following projections are outlined:
| Year |
Estimated U.S. Sales ($ millions) |
Market Share |
Notes |
| 2023 |
80 |
6% |
Continued erosion due to generics, slight growth expected |
| 2024 |
70 |
5.5% |
Further competition impacts, pricing pressures |
| 2025 |
60 |
4.5% |
Declining dominance, new competitors emerging |
| 2026 |
50 |
3.8% |
Market shift towards alternatives, decline persists |
| 2027 |
45 |
3.4% |
Steady decline, some brand loyalty persists |
Note: These projections assume no significant changes in regulatory status, new formulation releases, or major shifts in the competitive landscape.
Competitive Dynamics and Future Opportunities
-
Generic Entry: Since patent expiration, multiple generics have entered, lowering prices and restricting TESTIM's market share.
-
Novel Delivery Platforms: Competitors are developing longer-acting gels, patches, injections, and implantable devices, which could further reduce TESTIM's relevance.
-
Market Expansion: Entry into emerging markets or focusing on specific subpopulations (e.g., hypogonadal men with concurrent comorbidities) offers growth potential, though regulatory hurdles exist.
Regulatory and Policy Impacts
Regulation around testosterone prescribing has tightened, with agencies emphasizing cautious use to mitigate cardiovascular risks. This has led to more conservative prescribing patterns, limiting overall growth potential for testosterone therapies, including TESTIM.
Key Takeaways
- TESTIM faces significant patent-expiry-related sales erosion with increased generic competition.
- The U.S. market is consolidating around fewer highly marketed products, reducing TESTIM’s share.
- Pricing pressures and physician preferences for longer-acting therapies forecast declining sales.
- Strategic focus areas for the pharmaceutical company could include pipeline development, alternative formulations, and market diversification.
FAQs
1. Will TESTIM regain market share with new product versions?
Potentially, if new formulations demonstrate superior efficacy, safety, or convenience. However, development timelines and regulatory approval are lengthy processes, making near-term breakthroughs unlikely.
2. How significant is off-label use affecting TESTIM sales?
Off-label use of testosterone therapies has increased, but regulatory scrutiny and risk management have limited such practices, curbing unapproved growth.
3. Are there markets outside the U.S. with growth potential for TESTIM?
Yes, some emerging markets lack extensive generic competition and reimbursement barriers, offering growth opportunities. However, regulatory, cultural, and economic factors require tailored strategies.
4. What impact do recent regulatory warnings have on testosterone therapy sales?
Strict labeling warnings and risk disclosures have made prescribers more cautious, slowing growth and emphasizing appropriate patient selection.
5. Can TESTIM leverage partnerships or acquisitions to regain market share?
Strategic alliances or recent acquisitions of pipeline assets could extend product utility. Nonetheless, without significant differentiation, market share gains remain challenging.
References
- MarketsandMarkets. "Androgen Replacement Therapy Market," 2022.
- EvaluatePharma. "Global Testosterone Replacement Products," 2022.
- U.S. Food and Drug Administration. "FDA Drug Labeling," 2022.
- IQVIA. "Pharmaceutical Market Data," 2022.
- Reuters. "Testosterone Market Dynamics," 2022.