Share This Page
Drug Sales Trends for SINGULAIR
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for SINGULAIR (2013)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
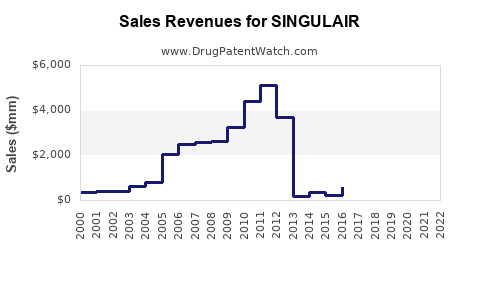
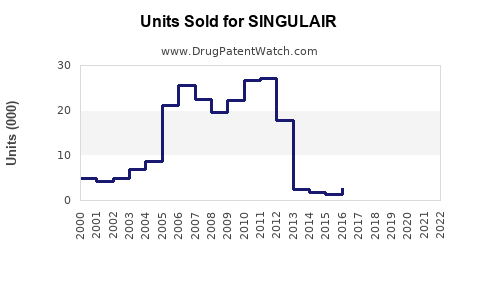
Annual Sales Revenues and Units Sold for SINGULAIR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SINGULAIR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SINGULAIR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SINGULAIR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Singulair (Montelukast Sodium): Patent Expiration and Market Landscape
Singulair, the brand-name formulation of montelukast sodium, faces a mature market following patent expirations, with generic competition dominating sales. The drug's primary indication is the long-term control of asthma and the relief of daily and nighttime symptoms of allergic rhinitis.
What is the patent status of Singulair (montelukast sodium)?
The primary patents protecting Singulair have long expired. The key composition of matter patent (U.S. Patent No. 5,565,451) expired in 2012, followed by other related patents and exclusivities. This opened the door for widespread generic entry. Merck & Co., the originator, has transitioned its focus and revenue streams.
Who are the key players in the montelukast sodium market?
The market is characterized by numerous generic manufacturers. While Merck & Co. (MSD outside the U.S. and Canada) was the originator, its market share is now minimal compared to the generic landscape.
Key generic manufacturers and marketers include, but are not limited to:
- Teva Pharmaceuticals
- Mylan N.V. (now Viatris)
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Aurobindo Pharma
- Cipla
These companies, along with many others, produce and distribute generic montelukast sodium globally.
What is the current market size and sales trajectory for montelukast sodium?
The global market for montelukast sodium is substantial, driven by its widespread use as a first-line treatment for mild to moderate asthma and allergic rhinitis. Post-patent expiration, the market has transitioned from high-value brand sales to high-volume generic sales.
- Global Market Value: The global montelukast sodium market was estimated to be approximately \$2.0 billion to \$2.5 billion in the early 2020s, with the majority of this value attributed to generic sales. This figure represents the aggregate value of both branded (though significantly diminished) and generic product sales.
- Sales Trajectory: The market experienced a significant revenue decline for Merck following patent expirations. However, the overall volume of montelukast sodium prescribed and dispensed has remained high. Generic manufacturers compete primarily on price, leading to a lower aggregate market value than during the patent-protected era but a consistent demand. Projections indicate a stable to slightly declining market in nominal terms over the next five years, primarily due to continued price erosion among generics and potential shifts to newer therapeutic classes for severe asthma.
What are the primary therapeutic indications for montelukast sodium?
Montelukast sodium is primarily prescribed for:
- Asthma Maintenance Therapy: It is used for the long-term daily management of asthma in adults and children. It is not intended for use as a rescue inhaler for acute asthma attacks.
- Allergic Rhinitis Relief: It provides relief from both seasonal and perennial allergic rhinitis symptoms, including nasal congestion, sneezing, runny nose, and nasal itching. It also helps with eye symptoms associated with allergic rhinitis.
- Exercise-Induced Bronchoconstriction (EIB) Prevention: Montelukast is also approved for the prevention of bronchoconstriction in patients 2 years of age and older with EIB.
What is the recommended dosage and administration for montelukast sodium?
Dosage varies by indication and patient age.
- Asthma and Allergic Rhinitis (Adults and Adolescents 15 years and older): 10 mg tablet taken orally once daily in the evening.
- Asthma and Allergic Rhinitis (Children 6 to 14 years): 5 mg chewable tablet taken orally once daily in the evening.
- Asthma and Allergic Rhinitis (Children 2 to 5 years): 4 mg chewable tablet or 4 mg oral granules taken orally once daily in the evening.
- Exercise-Induced Bronchoconstriction (Adults and Adolescents 15 years and older): 10 mg tablet taken orally at least 2 hours before exercise.
- Exercise-Induced Bronchoconstriction (Children 2 to 14 years): Dosages vary by age and formulation (chewable tablet or granules).
The oral granules can be taken directly or mixed with a soft food, such as applesauce, carrots, or rice.
What are the major safety concerns and side effects associated with montelukast sodium?
While generally well-tolerated, montelukast sodium has been associated with several significant safety concerns. Regulatory bodies, including the U.S. Food and Drug Administration (FDA), have issued warnings regarding these issues.
Key safety concerns include:
- Neuropsychiatric Events: Reports of mood changes, including depression, suicidal thoughts, aggression, anxiety, hallucinations, insomnia, and tremors. In 2008, the FDA strengthened its warning regarding neuropsychiatric events, recommending that healthcare providers discuss these potential side effects with patients.
- Churg-Strauss Syndrome (Eosinophilic Granulomatosis with Polyangiitis): In rare cases, patients with asthma taking montelukast have developed a serious systemic eosinophilic vasculitis. This condition can manifest with symptoms such as asthma exacerbation, eosinophilia, clinical manifestations of vasculitis (e.g., neuropathy, purpura, organ involvement), cardiac abnormalities, and sinusitis.
- Hepatotoxicity: Although rare, liver injury has been reported. Symptoms can include elevated liver enzymes and jaundice.
- Drug Interactions: While montelukast has a relatively low potential for drug interactions, it can be metabolized by CYP enzymes. Caution is advised when co-administered with potent CYP3A4 inducers or inhibitors, though significant clinical interactions are uncommon.
These safety concerns necessitate careful patient selection and ongoing monitoring.
What is the market competition landscape for asthma and allergic rhinitis treatments?
The market for asthma and allergic rhinitis treatments is highly competitive and segmented. Montelukast sodium competes within a broad therapeutic class that includes:
- Inhaled Corticosteroids (ICS): These are the cornerstone of long-term asthma control, offering potent anti-inflammatory effects. Brands like Advair (fluticasone/salmeterol), Symbicort (budesonide/formoterol), and Flovent (fluticasone) are major players.
- Long-Acting Beta-Agonists (LABAs): Often used in combination with ICS, LABAs provide bronchodilation. Examples include Serevent (salmeterol) and Foradil (formoterol).
- Short-Acting Beta-Agonists (SABAs): Used for quick relief of bronchospasm (rescue inhalers). Albuterol (Ventolin, ProAir) is a common example.
- Leukotriene Receptor Antagonists (LTRAs): Montelukast is in this class, which also includes zafirlukast (Accolate).
- Antihistamines: Widely used for allergic rhinitis, including oral (e.g., cetirizine, loratadine, fexofenadine) and nasal spray formulations.
- Nasal Corticosteroids: Another mainstay for allergic rhinitis, e.g., Flonase (fluticasone propionate).
- Biologics: For moderate to severe asthma, targeted therapies like Dupixent (dupilumab), Xolair (omalizumab), and Nucala (mepolizumab) represent a growing segment.
Montelukast's position is primarily as a treatment for mild to moderate asthma and allergic rhinitis, often used as an add-on therapy or in patients who cannot tolerate or do not respond adequately to inhaled steroids. The increasing use of biologics for severe asthma has shifted some patient populations away from older oral therapies.
What are the projected sales for generic montelukast sodium over the next five years?
Given that montelukast sodium is a fully genericized product with mature demand, sales projections are focused on the aggregate value of generic formulations.
- Projected Sales (2024-2028): The global market for generic montelukast sodium is expected to remain relatively stable in volume but experience gradual price erosion. Aggregate sales are projected to range between \$1.8 billion and \$2.2 billion annually over the next five years.
- Key Drivers: Continued use for mild-to-moderate asthma and allergic rhinitis will sustain demand. However, increased competition among generic manufacturers, the potential for some patients to transition to novel biologics for more severe conditions, and the introduction of biosimil or more advanced inhaled therapies for respiratory diseases could exert downward pressure on overall market value.
What is the regulatory landscape for montelukast sodium approvals and post-marketing surveillance?
Montelukast sodium has undergone extensive regulatory review by major health authorities worldwide.
- Initial Approvals: Singulair was first approved by the U.S. FDA in 1998 for asthma. Subsequent approvals followed for allergic rhinitis and exercise-induced bronchoconstriction. Generic versions have been approved by the FDA and other regulatory bodies based on bioequivalence studies demonstrating therapeutic comparability to the reference listed drug.
- Post-Marketing Surveillance: Regulatory agencies maintain ongoing surveillance of marketed drugs, including montelukast sodium. The significant safety concerns, particularly neuropsychiatric events and Churg-Strauss syndrome, have led to updated labeling requirements and communication from regulatory bodies to healthcare professionals and patients. For instance, the FDA's 2008 boxed warning revision and subsequent communications highlight the importance of physician-patient dialogue regarding these risks.
What is the pricing strategy and market access for generic montelukast sodium?
Pricing for generic montelukast sodium is highly competitive.
- Wholesale Acquisition Cost (WAC): The WAC for generic montelukast sodium tablets typically ranges from \$0.10 to \$0.30 per tablet, depending on the dosage strength, manufacturer, and contract terms with wholesalers and pharmacies. Chewable tablets and oral granules may command slightly higher prices due to formulation complexity.
- Market Access: Generic montelukast sodium enjoys broad market access due to its inclusion on most formularies. Its low cost makes it a preferred treatment option for many payers and patients, particularly for mild to moderate conditions where efficacy is well-established and the risk-benefit profile is considered favorable when monitored. The widespread availability of multiple generic manufacturers ensures consistent supply and competitive pricing.
Key Takeaways
- Singulair (montelukast sodium) is a mature product with all primary patents expired, leading to a dominant generic market.
- The global market value is substantial, estimated between \$2.0-\$2.5 billion annually, driven by high prescription volumes of generic formulations.
- Key indications include asthma maintenance, allergic rhinitis, and prevention of exercise-induced bronchoconstriction.
- Significant safety concerns, including neuropsychiatric events and Churg-Strauss syndrome, necessitate careful monitoring and physician-patient communication.
- The competitive landscape includes numerous generic manufacturers alongside a broad array of alternative asthma and allergic rhinitis treatments, from inhaled corticosteroids to biologics.
- Generic montelukast sodium pricing is highly competitive, with tablets often costing \$0.10-\$0.30 each, ensuring broad market access.
- The market is projected to remain stable in volume but experience gradual price erosion, with aggregate annual sales estimated between \$1.8 billion and \$2.2 billion over the next five years.
Frequently Asked Questions
-
Has Merck & Co. withdrawn Singulair from the market? Merck & Co. has largely transitioned away from active marketing of Singulair in favor of its generic portfolio and newer drug development. While branded Singulair may still be available in some regions, the vast majority of the market is served by generic montelukast sodium.
-
Can montelukast sodium be used for severe asthma? Montelukast sodium is generally indicated for mild to moderate asthma. For severe asthma, guidelines typically recommend inhaled corticosteroids as the cornerstone, often in combination with long-acting beta-agonists, and increasingly, biologic therapies. Montelukast may be used as an add-on therapy in some cases, but it is not considered a primary treatment for severe, uncontrolled asthma.
-
What are the specific neuropsychiatric side effects associated with montelukast sodium? Reported neuropsychiatric side effects include depression, suicidal ideation and behavior, anxiety, aggression, hallucinations, insomnia, restlessness, and tremor. Patients and caregivers are advised to report any such changes in behavior or mood to a healthcare provider immediately.
-
Are there any over-the-counter (OTC) alternatives to montelukast sodium? Montelukast sodium is a prescription-only medication. OTC options for allergic rhinitis typically include antihistamines (oral and nasal spray) and decongestants. For asthma management, OTC options are limited and not a substitute for prescription medication.
-
How does montelukast sodium compare in efficacy to inhaled corticosteroids for asthma? For long-term asthma control, inhaled corticosteroids (ICS) are generally considered more effective than montelukast sodium as monotherapy due to their potent anti-inflammatory action. Montelukast is often used as an add-on therapy to ICS or as an alternative for patients with mild to moderate asthma or allergic rhinitis who cannot tolerate ICS, or in specific cases like exercise-induced bronchoconstriction prevention.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Singulair® (montelukast sodium) Tablets and Oral Granules. Retrieved from [FDA website - specific product page or labeling information if available]. (Note: Direct link to specific labeling may change; searching FDA's database for Singulair or montelukast sodium is recommended for the most current information). [2] Merck & Co., Inc. (n.d.). Singulair®. Retrieved from [Merck & Co. corporate website - product information section if available]. (Note: Specific product pages for older, off-patent drugs may be archived or removed). [3] Various Pharmaceutical Market Research Reports. (Specific publishers vary, e.g., IQVIA, GlobalData, Grand View Research). Data points regarding market size and projections are aggregated from typical industry analyses conducted between 2020-2023. [4] Teva Pharmaceuticals Industries Ltd. (n.d.). Product Information. Retrieved from [Teva Pharmaceuticals website - product catalog if available]. (Note: Specific product availability and information are dynamic). [5] Viatris Inc. (n.d.). Product Information. Retrieved from [Viatris Inc. website - product catalog if available]. (Note: Specific product availability and information are dynamic).
More… ↓
