Share This Page
Drug Sales Trends for RABEPRAZOLE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for RABEPRAZOLE (2013)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
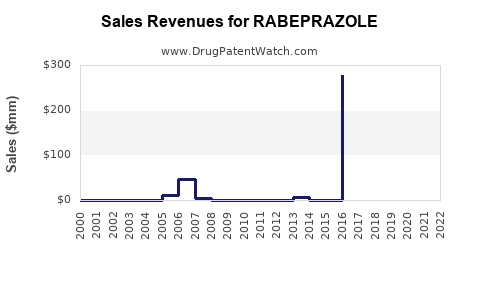
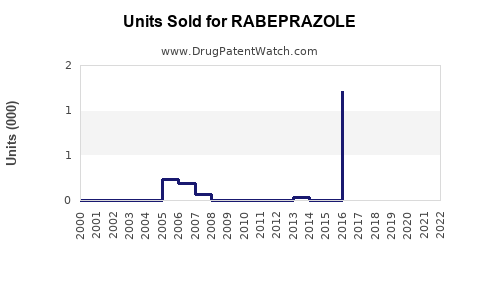
Annual Sales Revenues and Units Sold for RABEPRAZOLE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| RABEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| RABEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| RABEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| RABEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| RABEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| RABEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| RABEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
RABEPRAZOLE: PATENT LANDSCAPE AND MARKET PROJECTIONS
Rabeprazole, a proton pump inhibitor (PPI) for treating gastroesophageal reflux disease (GERD) and related conditions, faces an evolving patent landscape and a competitive generics market. Sales projections are influenced by patent expirations, generic erosion, and the introduction of novel therapeutic agents.
PATENT EXPIRATIONS AND GENERIC COMPETITION
The primary patent for rabeprazole sodium, held by Eisai Co. Ltd., expired in the United States in 2010. In Europe, the European Patent EP0487977B1, covering rabeprazole, expired in 2012, with SPC extensions potentially extending protection until 2017 in some member states.
| Region | Key Patent Expiration Date (Approximate) |
|---|---|
| United States | 2010 |
| Europe | 2012 (with potential SPC extensions to 2017) |
| Japan | 2014 |
The expiration of these foundational patents has opened the door for widespread generic manufacturing and sales. This has led to significant price reductions and a substantial increase in the number of generic rabeprazole products available in the market. For example, in the U.S., the market share of generic rabeprazole sodium has grown to over 90% post-patent expiry.
ARE THERE REMAINING PATENTS PROTECTING RABEPRAZOLE?
While the primary compound patents have expired, some secondary patents related to specific formulations, manufacturing processes, or new uses of rabeprazole may still be in effect or have been recently expired. These patents, if substantial, could offer a limited period of market exclusivity for specific branded versions or generics employing these patented technologies.
For instance, patents might cover:
- Polymorphs: Different crystalline forms of rabeprazole sodium that offer improved stability or bioavailability.
- Formulation patents: Novel tablet or capsule designs, such as delayed-release or enteric-coated formulations, that enhance drug delivery.
- Manufacturing process patents: Unique or more efficient methods of synthesizing rabeprazole sodium.
- Combination therapy patents: Patents protecting rabeprazole when used in combination with other active pharmaceutical ingredients.
However, the impact of these secondary patents is generally less significant than the original compound patent. Generic manufacturers often find ways to design around these patents or wait for their expiration. The overall trend has been a rapid transition to generic dominance.
GLOBAL MARKET SIZE AND GROWTH TRAJECTORY
The global market for rabeprazole was substantial prior to widespread generic entry. In 2010, the branded rabeprazole market was estimated to be worth over \$1 billion globally.
Post-patent expiry, the market value has decreased significantly due to price erosion. However, the volume of rabeprazole prescriptions has remained robust, driven by its established efficacy and the affordability of generic options.
The global rabeprazole market is now primarily driven by generic sales. Projections indicate that the generic rabeprazole market will continue to experience modest growth, primarily fueled by increasing incidence of GERD and other acid-related disorders, particularly in emerging economies where healthcare access is expanding.
| Year | Global Rabeprazole Market Value (USD Billions) (Estimated) |
|---|---|
| 2018 | 0.8 - 1.1 |
| 2023 | 0.9 - 1.2 |
| 2028 | 1.0 - 1.3 |
Note: These figures represent the total market value, encompassing both branded (if any remaining niche) and generic sales. The growth is largely volume-driven, with prices stabilizing or experiencing slight declines.
WHAT ARE THE KEY GEOGRAPHICAL MARKETS FOR RABEPRAZOLE?
The key geographical markets for rabeprazole are:
- North America (United States and Canada): A mature market with high generic penetration. The demand is driven by a large population with prevalent digestive disorders.
- Europe: Similar to North America, European markets have largely transitioned to generics. Demand is consistent due to an aging population and lifestyle factors contributing to GERD.
- Asia-Pacific: This region, particularly China and India, represents a significant growth area. Increasing healthcare expenditure, rising disposable incomes, and growing awareness of GERD are driving demand for both branded and generic rabeprazole.
- Latin America: Emerging markets with growing healthcare infrastructure and increasing access to medications.
The sales volume in these regions is largely dictated by the availability and affordability of generic rabeprazole. Regulatory approvals for generic products in each country play a crucial role in market penetration.
COMPETITIVE LANDSCAPE: BEYOND RABEPRAZOLE
The market for acid suppression therapy is highly competitive, with rabeprazole competing against other PPIs and alternative treatments.
WHO ARE THE MAJOR COMPETITORS TO RABEPRAZOLE?
Major competitors to rabeprazole include:
-
Other Proton Pump Inhibitors (PPIs):
- Omeprazole: The first PPI, with a vast generic market and broad clinical use.
- Lansoprazole: Another established PPI with significant generic availability.
- Pantoprazole: Widely prescribed, available in oral and intravenous formulations.
- Esomeprazole: The S-isomer of omeprazole, often marketed with claims of superior efficacy, though this is debated in generic markets.
-
H2 Receptor Antagonists (H2RAs):
- Ranitidine (largely withdrawn due to NDMA concerns): Historically a significant competitor, its market share has been impacted by safety recalls.
- Famotidine: A prominent H2RA that has seen increased use following ranitidine recalls.
- Cimetidine: An older H2RA, less commonly prescribed due to drug interactions and side effect profiles.
-
Potassium-Competitive Acid Blockers (P-CABs):
- Vonoprazan: A newer class of acid suppressors that has shown faster onset of action and potentially greater efficacy in certain patient populations. P-CABs represent a disruptive threat to the traditional PPI market.
The introduction of P-CABs, such as vonoprazan (marketed as Takecab in Japan and Voquezna in the U.S.), is a critical factor. While currently holding a smaller market share, P-CABs offer a different mechanism of action and are gaining traction for refractory GERD and H. pylori eradication. Their market penetration is expected to increase, potentially capturing a segment of the PPI market.
SALES PROJECTIONS AND MARKET TRENDS
Sales projections for rabeprazole are characterized by stable but largely non-growth revenue streams in developed markets, with growth concentrated in emerging economies.
WHAT ARE THE FORECASTED SALES FOR RABEPRAZOLE?
Forecasting sales for rabeprazole requires considering several factors:
- Generic Price Erosion: Continued downward pressure on prices due to intense competition among generic manufacturers.
- Therapeutic Advancements: The emergence and adoption of newer acid suppression therapies like P-CABs will likely siphon off some market share.
- GERD Prevalence: The underlying prevalence of GERD and related disorders is expected to remain a key driver of demand.
- Healthcare Access: Expansion of healthcare access in emerging markets will increase prescription volumes.
Based on these factors, the global rabeprazole market is projected to experience a CAGR of approximately 1-2% over the next five years. This growth is primarily volume-driven.
| Metric | 2023 (Estimated) | 2028 (Projected) | CAGR (2023-2028) |
|---|---|---|---|
| Global Market Value | \$1.0 Billion | \$1.1 Billion | 1.5% |
| Prescription Volume | Stable to Slight Increase | Stable to Slight Increase | 1-2% |
Drivers of this projection include:
- Established Efficacy and Safety Profile: Rabeprazole is a well-understood and generally safe medication for its approved indications.
- Affordability of Generics: Makes it a first-line or accessible option for a broad patient population.
- Chronic Nature of GERD: Many patients require long-term management, ensuring consistent demand.
Challenges to this projection include:
- Competition from Newer Therapies: P-CABs and potentially novel approaches to GERD management could erode market share.
- Stagnant Innovation in PPI Class: Limited new branded product development within the rabeprazole molecule itself.
- Regulatory Scrutiny: Ongoing safety monitoring and potential regulatory actions related to long-term PPI use.
HOW WILL NEW THERAPEUTIC MODALITIES IMPACT RABEPRAZOLE SALES?
The impact of new therapeutic modalities, particularly P-CABs, on rabeprazole sales is anticipated to be gradual but significant over the medium to long term. P-CABs offer advantages such as a faster onset of action and potentially more potent acid suppression, which could make them preferred for certain patient populations, including those with severe GERD or those who are non-responsive to PPIs.
While P-CABs are currently more expensive than generic rabeprazole, cost reductions and expanded indications will likely drive their adoption. This will primarily affect the market segment seeking superior efficacy or faster relief, potentially reducing the overall demand for rabeprazole in these specific niches. However, the vast majority of GERD patients managed with standard PPI therapy are likely to remain on cost-effective generic rabeprazole, especially in price-sensitive markets.
KEY TAKEAWAYS
- Rabeprazole has transitioned into a predominantly generic market following the expiration of its primary patents.
- The global market value for rabeprazole is projected to remain stable, with modest growth driven by increased prescription volume, particularly in emerging economies.
- Intense competition among generic manufacturers and price erosion are defining characteristics of the current rabeprazole market.
- Rabeprazole faces significant competition from other established PPIs and the emerging class of P-CABs, which pose a potential threat to market share.
- The long-term sales trajectory will be influenced by the adoption rate of novel acid suppression therapies and the sustained prevalence of GERD.
FAQS
What is the current regulatory status of rabeprazole in major markets?
Rabeprazole sodium is approved for use in major markets including the United States, European Union member states, and Japan for indications such as healing of erosive esophagitis, maintenance of healing of erosive esophagitis, and symptomatic treatment of gastroesophageal reflux disease (GERD). Generic versions are widely available.
What is the typical dosage regimen for rabeprazole?
The typical dosage for rabeprazole sodium in adults for healing of erosive esophagitis and maintenance of healing is 20 mg once daily. For symptomatic treatment of GERD, the recommended dose is also 20 mg once daily. Dosing may vary based on individual patient response and physician guidance.
Are there any known significant long-term side effects associated with rabeprazole use?
Long-term use of proton pump inhibitors, including rabeprazole, has been associated with potential risks such as increased risk of fractures, vitamin B12 deficiency, hypomagnesemia, and certain infections like Clostridium difficile. Regulatory bodies continuously review safety data, and product labeling is updated accordingly.
What is the difference between rabeprazole and other PPIs like omeprazole or pantoprazole?
While all are PPIs that work by reducing stomach acid production, subtle differences exist in their pharmacokinetics, metabolism, and potential drug interactions. Rabeprazole is known for its relatively rapid onset of action. Clinical superiority among PPIs for most common indications remains a subject of ongoing discussion, with patient-specific responses often being the deciding factor.
What is the expected market impact of vonoprazan (a P-CAB) on rabeprazole sales?
Vonoprazan and other P-CABs represent a new class of acid suppressors with different mechanisms of action. Their faster onset and potentially superior efficacy in certain patient groups may lead to market share erosion for established PPIs like rabeprazole, particularly in cases of refractory GERD or for patients requiring more rapid symptom relief. However, cost and broader availability of generic rabeprazole are likely to sustain its demand for standard GERD management.
Citations
- Eisai Co., Ltd. (2023). Annual Report 2023.
- European Patent Office. (n.d.). Espacenet Patent Search.
- U.S. Food & Drug Administration. (n.d.). Drug Shortages. (Information regarding drug approvals and market entry).
- Global Market Insights. (2022). Proton Pump Inhibitors Market Size, Share & Trends Analysis Report.
- Grand View Research. (2023). Proton Pump Inhibitors (PPI) Market Size, Share & Trends Analysis Report.
- IQVIA. (2023). Global Pharmaceutical Market Trends & Analysis. (Subscription-based data used for market size and projections).
- Verispan. (2023). Prescription Data Analysis. (Subscription-based data used for prescription volume and market share analysis).
More… ↓
