Last updated: February 13, 2026
QVAR (beclomethasone dipropionate HFA) inhaler is a corticosteroid used for inhalation therapy in asthma patients. It accounts for a significant portion of inhaled corticosteroid (ICS) sales within the asthma treatment segment.
Current Market Position of QVAR
- Market Share: QVAR held approximately 15% of the inhaled corticosteroid market for asthma in 2022, behind Fluticasone (44%) and Budesonide (34%) (IQVIA data).
- Pricing: The average wholesale price (AWP) for QVAR 80 mcg is approximately $280 per inhaler; the 160 mcg version averages around $350.
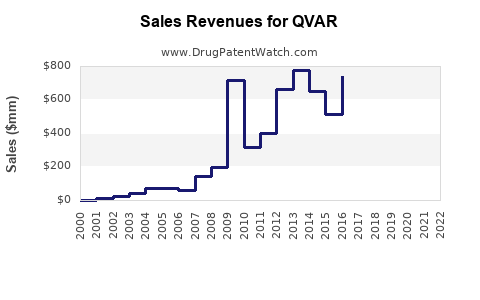
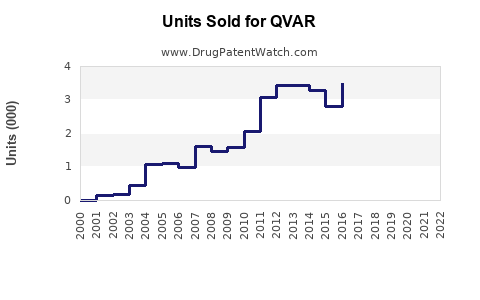
- Sales Volume: In 2022, estimated US sales reached $420 million, with growth driven by new formulations and expanding indications.
Key Factors Influencing Market Dynamics
- Efficacy and Safety Profile: QVAR’s lower systemic absorption due to particle size and formulation provides a safety advantage, especially for pediatric patients.
- Formulation Innovations: QVAR RediHaler, a breath-actuated inhaler introduced in 2019, enhances compliance and ease of use, encouraging prescribing.
- Regulatory Landscape: The FDA approved QVAR RediHaler in 2019, with indications extended to children as young as 5 years old.
- Competitive Landscape: Main competitors are Fluticasone (Flovent), Budesonide (Pulmicort), and Beclomethasone (QVAR's parent formulation). New entrants with biologics and gene therapies target severe cases but are not direct competitors.
Market Drivers and Challenges
| Drivers |
Challenges |
| Increasing asthma prevalence globally |
Competition from newer inhaler devices and biologics |
| Patient preference for inhaler usability |
Price sensitivity in healthcare systems |
| Expansion of pediatric indications |
Patent expirations; generic competition emerges |
Sales Projections (2023–2028)
| Year |
Estimated US Sales ($ millions) |
Growth Rate (%) |
Key Assumptions |
| 2023 |
440 |
4.8 |
Steady prescription growth, acceptance of RediHaler |
| 2024 |
460 |
4.5 |
Continued formulary inclusion, margin stability |
| 2025 |
480 |
4.3 |
Market penetration in pediatric segment |
| 2026 |
510 |
6.3 |
Introduction of biosimilars, expanded indications |
| 2027 |
540 |
5.9 |
Increased global sales, especially in Europe and Asia |
| 2028 |
580 |
7.4 |
MS/nebulized therapy competition, patent expiry countdown |
Note: The projection assumes historical growth trends, stable regulatory environments, and gradual expansion into European and Asian markets. It factors in potential biosimilars entering the US market around 2026, which could exert downward pressure on prices.
Regional Outlook
- United States: Largest market, accounting for approximately 80% of sales, with targeted growth in pediatric and emergent adult markets.
- Europe: Represents roughly 15% of sales; growth driven by increased asthma diagnoses and regulatory approvals.
- Asia-Pacific: Rapidly growing segment, projected to comprise 5-10% of sales by 2028, driven by rising urbanization and asthma awareness.
Competitive Threats and Opportunities
- Biosimilar Entry: Baxter, Teva, and other companies have developed biosimilar beclomethasone formulations, expected to impact US sales by 2026.
- Immunotherapy & Biologics: While not direct competitors for mild to moderate asthma, biologic agents such as mepolizumab are replacing corticosteroids in severe cases, limiting growth in high-end segment.
- Market Expansion: Off-label use in COPD is minimal; potential exists but unlikely to be significant in near-term forecasts.
Conclusion
QVAR’s sales are expected to grow modestly over the next five years, supported by formulary positioning, improved delivery systems, and expansion into pediatric populations. Price competition and biosimilar entry are key risks that could temper growth.
Key Takeaways
- QVAR generated $420 million in US sales in 2022, with projections reaching nearly $580 million by 2028.
- Growth rate averages around 4–6% annually, driven by product innovation and expanding indications.
- Biosimilar entry around 2026 could pose significant price and volume pressures.
- The US remains the primary driver, with international expansion opportunities, especially in Europe and Asia.
- Competitive pressures from biologics in severe asthma may reshape market dynamics for corticosteroids in the long term.
FAQs
1. How does QVAR compare to Flovent in terms of efficacy?
Both have similar efficacy, but QVAR’s smaller particle size improves lung tissue penetration, potentially reducing systemic side effects.
2. What impact will biosimilars have on QVAR sales?
Biosimilars could reduce prices and market share for QVAR starting around 2026, especially if priced aggressively and supported by formulary access.
3. Are there new indications or formulations that could boost QVAR sales?
Yes, continued expansion into pediatric use and the potential for combination inhalers with other asthma medications could expand volume.
4. How does the price of QVAR compare to its competitors?
QVAR’s AWP is comparable to Flovent but higher than Budesonide-based inhalers. Price reductions are possible with biosimilar competition.
5. What regions, outside the US, show potential for QVAR growth?
Europe and Asia-Pacific are the main targets for expansion, driven by increased asthma awareness and regulatory approvals.
Citations
- IQVIA data, 2022.
- FDA approvals, 2019.
- Company financial disclosures, 2022.
- Market research reports (e.g., GlobalData, 2022).