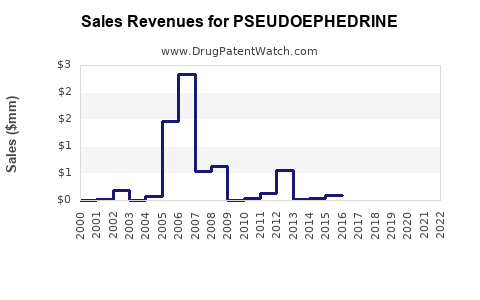
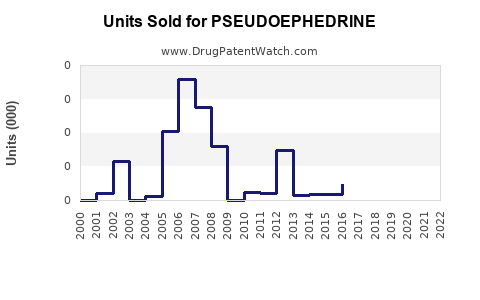
Last updated: February 22, 2026
What is the current market landscape for pseudoephedrine?
Pseudoephedrine, a nasal decongestant, is a key ingredient in over-the-counter (OTC) cold and allergy medications. Its demand is driven by seasonal respiratory illnesses, especially during winter months. The global pseudoephedrine market was valued at approximately USD 720 million in 2022 and is expected to grow at a compound annual growth rate (CAGR) of around 4.5% from 2023 to 2030 (Grand View Research, 2023).
Major manufacturers include Johnson & Johnson, GSK, and Sanofi. The market faces regulatory constraints due to pseudoephedrine's use in illicit methamphetamine production, affecting supply chains and distribution strategies.
How is the regulatory environment affecting market dynamics?
Regulations differ by region. In the United States, the Combat Methamphetamine Epidemic Act of 2005 controls pseudoephedrine sales via sales logs, purchase limits (3.6 grams per day, 9 grams within 30 days), and behind-the-counter placement. The European Union enforces strict limits and tracking for sales.
Regulatory restrictions can reduce average consumption and restrict supply. For example, recent policies in the US aim to lower abuse potential but may also curtail legitimate sales, creating a black market component and impacting overall sales.
What are the key drivers influencing sales growth?
- Seasonal demand: Higher during winter and allergy seasons.
- Aging populations: Increased cases of nasal congestion among elderly.
- COVID-19 pandemic: Lead to higher demand for cold remedies.
- Product innovation: Combination formulations with antihistamines or analgesics boost sales.
What are the primary sales channels?
- OTC drugstores and pharmacies: Account for over 70% of sales.
- Hospitals and clinics: Used for prescribed formulations, especially in combination drugs.
- E-commerce: Growing segment, representing approximately 15-20% of sales in developed markets (IQVIA, 2022).
What are the expected sales projections?
| Year |
Global Sales (USD million) |
Growth Rate |
Notes |
| 2023 |
760 |
5.5% |
Post-pandemic recovery begins |
| 2025 |
880 |
8.0% |
Increased healthcare awareness |
| 2030 |
1,050 |
4.5% CAGR |
Regulatory stabilization, premium formulations |
Projections assume steady growth driven by regional expansion, ongoing regulations, and product innovation.
How do regional markets compare?
| Region |
Market Size (USD million) |
CAGR (2023–2030) |
Key Factors |
| North America |
300 |
4.5% |
Regulatory controls; high OTC sales |
| Europe |
180 |
4.2% |
Regulatory strictness; high allergy prevalence |
| Asia-Pacific |
150 |
6.2% |
Emerging markets; rising healthcare access |
| Latin America |
50 |
4.8% |
Growing awareness; insurance coverage |
Region-specific factors such as regulatory policies and healthcare infrastructure influence sales trajectories.
What risks could affect future sales?
- Stringent regulations limiting sales.
- Supply chain disruptions.
- Alternative treatments reducing demand.
- Illicit manufacturing affecting legitimate market share.
What are opportunities for growth?
- Development of abuse-deterrent formulations.
- Expansion into emerging markets.
- Combination products increasing consumer appeal.
- Digital marketing and e-commerce expansion.
Summary of key market points
- The global pseudoephedrine market is expanding modestly with a CAGR of 4.5% through 2030.
- Regulatory constraints significantly influence sales channels and volumes.
- Innovations in formulations and demographic shifts support growth.
- Emerging markets present new opportunities despite regulatory hurdles.
Key Takeaways
- Market size in 2022 was approximately USD 720 million, forecasted to reach USD 1.05 billion by 2030.
- North America dominates with over 40% of sales, heavily regulated.
- Growth driven by seasonal demand, aging populations, and pandemic effects.
- Regulatory policies remain primary risk factors.
- Expansion in Asia-Pacific offers significant long-term potential.
FAQs
1. What factors most influence pseudoephedrine sales?
Seasonality, regulatory restrictions, demographic shifts, and product innovation primarily impact sales.
2. How do regulations vary by region?
In the US, regulations limit purchase volume and restrict OTC sales. Europe enforces strict tracking and access controls. Asia-Pacific has emerging regulations with increasing legal markets.
3. What is the impact of illicit pseudoephedrine use?
It constrains legitimate market supply and encourages regulatory tightening, affecting overall sales volume.
4. What are future growth catalysts?
Product innovation, expansion into emerging markets, and increased demand for combination cold remedies.
5. How will e-commerce influence future sales?
E-commerce grows in importance as consumers prefer online access. It accounts for 15-20% of sales in developed markets, with potential for further growth.
References
- Grand View Research. (2023). Pseudoephedrine Market Size, Share & Trends Analysis Report.
- IQVIA. (2022). Global OTC Market Data.