Share This Page
Drug Sales Trends for PROPECIA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for PROPECIA (2013)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
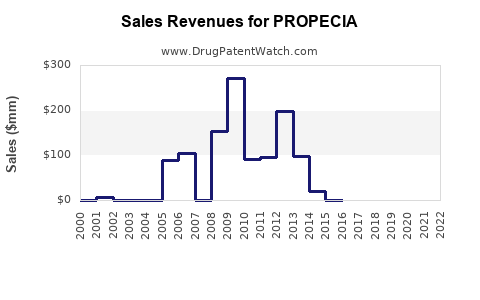
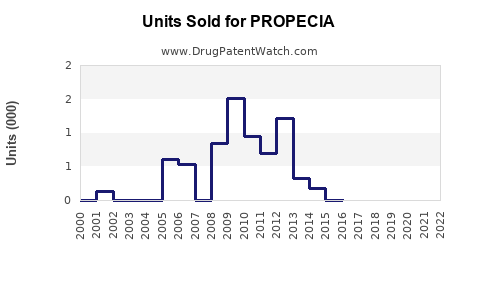
Annual Sales Revenues and Units Sold for PROPECIA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PROPECIA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PROPECIA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PROPECIA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PROPECIA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PROPECIA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| PROPECIA | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| PROPECIA | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PROPECIA (Finasteride) Market Analysis and Sales Projections
Executive Summary
Propecia (finasteride) is a prescription medication approved for the treatment of male pattern baldness. Developed by Merck & Co., it inhibits the conversion of testosterone to dihydrotestosterone (DHT), a hormone implicated in hair follicle miniaturization. The market for Propecia is mature, facing generic competition and evolving therapeutic landscapes. This analysis examines patent status, market dynamics, and provides sales projections based on current and anticipated market conditions.
What is Propecia and Its Mechanism of Action?
Propecia, with the active ingredient finasteride, is indicated for the treatment of androgenetic alopecia (male pattern baldness) in men. The drug functions as a selective inhibitor of type II 5-alpha-reductase, an enzyme that converts testosterone into dihydrotestosterone (DHT) [1]. DHT is the primary androgen responsible for hair follicle miniaturization in genetically susceptible individuals, leading to the characteristic thinning and loss of hair on the scalp. By reducing serum and scalp DHT levels by approximately 60-70%, Propecia can slow hair loss and, in some men, stimulate hair regrowth [2].
What is the Patent and Regulatory Status of Propecia?
Merck & Co. initially held composition of matter patents for finasteride and its use in treating male pattern baldness. The primary U.S. patent for Propecia (U.S. Patent No. 4,377,584) expired in 2005 [3]. Subsequent patents related to specific formulations, dosages, or methods of use may have existed but are also largely expired.
Key Patent Milestones:
- Composition of Matter Patent Expiration (U.S.): 2005 [3]
- Market Exclusivity: Lost due to patent expiration, leading to generic entry.
The U.S. Food and Drug Administration (FDA) approved Propecia in December 1997 [4]. Its approval marked a significant advancement in the pharmacological treatment of male pattern baldness.
What are the Current Market Dynamics for Propecia?
The market for Propecia is characterized by several key factors:
1. Generic Competition: Following the expiration of primary patents, numerous generic versions of finasteride 1mg tablets have entered the market. This has significantly driven down prices and fragmented market share for the branded Propecia. Generic finasteride is widely available and prescribed by physicians.
2. Established Efficacy and Safety Profile: Propecia has a long history of clinical use, with a well-documented efficacy and safety profile when used as prescribed. This established track record contributes to its continued prescription by healthcare professionals and use by patients.
3. Therapeutic Alternatives: While Propecia remains a leading pharmacological treatment, the market has seen the emergence and increasing adoption of other therapeutic options:
- Minoxidil (Rogaine): A topical solution or foam that also treats hair loss. It is available both over-the-counter and by prescription.
- Low-Level Laser Therapy (LLLT): Devices emitting red light are used to stimulate hair growth.
- Platelet-Rich Plasma (PRP) Therapy: An injectable treatment using a patient's own blood components.
- Hair Transplantation: Surgical procedures to transplant hair follicles.
4. Patient Compliance and Adherence: The effectiveness of finasteride requires consistent daily use for sustained results. Patient adherence can be a challenge, impacting long-term outcomes and market demand. Discontinuation of the drug typically leads to the reversal of its effects within 6-12 months [2].
5. Off-Label Use for Other Conditions: Finasteride, particularly the 5mg formulation (Proscar), is also prescribed off-label for benign prostatic hyperplasia (BPH). While this analysis focuses on Propecia's use for hair loss (1mg formulation), the broader availability and awareness of finasteride influence market perception.
6. Evolving Consumer Preferences: There is a growing interest in natural remedies and minimally invasive procedures among some consumer segments, potentially impacting the demand for pharmaceutical treatments. However, the proven efficacy of finasteride ensures its continued relevance.
7. Regulatory Scrutiny and Post-Market Surveillance: Like all pharmaceuticals, finasteride undergoes ongoing post-market surveillance. Reports of sexual side effects, although rare and often reversible upon discontinuation, have led to increased awareness and potential patient hesitancy [5].
Market Size and Growth (Estimates):
While specific, up-to-the-minute market size data for branded Propecia is challenging due to the dominance of generics, the overall male androgenetic alopecia treatment market, which finasteride is a significant part of, is valued in the billions of dollars globally. The generic finasteride market segment is substantial. The growth rate for the overall market is moderate, driven by increasing awareness, aging populations, and rising disposable incomes in emerging markets.
What are the Competitive Strengths and Weaknesses of Propecia?
Competitive Strengths:
- Proven Efficacy: Demonstrates significant reduction in hair loss and potential for regrowth in clinical trials and real-world use.
- Oral Administration: Convenient daily pill format compared to topical applications.
- Established Safety Profile: Decades of clinical data support its use, with known side effects and management strategies.
- Physician Familiarity: Widely prescribed and understood by dermatologists and general practitioners.
- Cost-Effectiveness (Generic): Generic finasteride offers a relatively affordable treatment option.
Competitive Weaknesses:
- Generic Competition: Loss of brand exclusivity has eroded market share and pricing power for Merck.
- Potential Side Effects: Reports of sexual dysfunction, though rare, can be a significant deterrent for some patients.
- Requires Continuous Use: Efficacy is lost upon discontinuation, necessitating long-term commitment and cost.
- Limited Efficacy for Advanced Hair Loss: More effective in earlier stages of hair loss; results can be less dramatic in advanced cases.
- Availability of Newer Technologies: Emerging treatments like PRP and hair transplantation offer alternative approaches, sometimes perceived as more natural or permanent.
What are the Sales Projections for Propecia?
Given the mature market, extensive generic competition, and the availability of alternative treatments, sales projections for branded Propecia are expected to remain modest, with a general downward trend or stabilization. Merck's revenue from branded Propecia is significantly impacted by generic erosion.
Assumptions for Projections:
- Continued Generic Availability: The market will remain dominated by generic finasteride 1mg.
- Stable Prescription Rates for Finasteride: The overall number of prescriptions for finasteride (branded and generic) will see slow, incremental growth or stabilization.
- Moderate Impact of Emerging Therapies: While newer therapies will capture market share, they will not entirely displace oral finasteride for a significant patient segment.
- No Major Safety Recalls or Efficacy Breakthroughs: Projections assume the current understanding of Propecia's efficacy and safety profile remains consistent.
- Branded Propecia's Declining Market Share: Merck's branded product will continue to lose market share to generics, primarily serving patients who specifically request or are prescribed the brand name.
Projected Sales Trends (Branded Propecia - Merck & Co.):
| Year | Projected Branded Propecia Sales (USD Millions) | Commentary |
|---|---|---|
| 2024 | $80 - $100 | Reflects ongoing generic competition and market maturity. |
| 2025 | $75 - $95 | Continued erosion of branded market share and potential price pressures. |
| 2026 | $70 - $90 | Stabilization or slight decline as generics solidify their position and alternative therapies gain traction. |
| 2027 | $65 - $85 | Sustained performance in a niche segment of patients preferring the branded product. |
| 2028 | $60 - $80 | Long-term projection assumes continued relevance for a segment of the market. |
Note: These figures represent estimated sales for Merck's branded Propecia. The total market for finasteride 1mg (including generics) is substantially larger.
Global Market for Finasteride 1mg (Branded and Generic) - Projected Trends:
The global market for finasteride 1mg is projected to experience modest growth, driven by increasing awareness of hair loss treatments and the affordability of generic options, particularly in emerging markets.
- Projected Growth Rate (Global Finasteride 1mg Market): 2-4% annually (2024-2028).
- Key Growth Drivers: Increasing prevalence of androgenetic alopecia, growing disposable income, and aggressive pricing by generic manufacturers.
- Regional Variations: Higher growth expected in Asia-Pacific and Latin America compared to North America and Europe, where market saturation is higher.
Key Takeaways
- Propecia's patent exclusivity has expired, leading to significant generic competition that dictates current market dynamics and pricing.
- The drug remains a cornerstone treatment for male pattern baldness due to its proven efficacy, convenient oral administration, and established safety profile.
- Emerging therapeutic alternatives and patient adherence challenges present ongoing pressures on sustained market growth.
- Branded Propecia sales are projected to decline or stabilize at modest levels, with the majority of the finasteride 1mg market dominated by generic products.
- The overall global finasteride 1mg market is expected to see slow, steady growth, primarily driven by generic expansion in developing economies.
Frequently Asked Questions
1. How does the expiration of Propecia's patents impact its market availability and pricing?
Patent expiration allows for the introduction of generic versions of finasteride. This competition leads to a substantial decrease in drug prices and a fragmentation of market share away from the original branded product, Propecia. Generic finasteride is now widely available at a fraction of the original Propecia cost.
2. What are the primary alternatives to Propecia for treating male pattern baldness?
Primary alternatives include topical minoxidil (e.g., Rogaine), low-level laser therapy (LLLT) devices, platelet-rich plasma (PRP) injections, and hair transplantation surgeries. Each treatment offers a different mechanism of action, cost, and efficacy profile.
3. What is the projected long-term outlook for the finasteride 1mg market?
The long-term outlook for the finasteride 1mg market, encompassing both branded and generic products, suggests continued relevance. While growth will be moderate, driven largely by generics, it will persist due to the drug's established efficacy and the increasing global prevalence of male pattern baldness.
4. How do the reported side effects of finasteride affect its market position?
Reported sexual side effects, such as decreased libido or erectile dysfunction, while rare and often reversible, can create patient apprehension. This awareness may lead some individuals to seek alternative treatments or reconsider using finasteride, thus impacting its market penetration and physician prescribing patterns.
5. Is Merck & Co. still actively marketing branded Propecia, or has the focus shifted to generics?
Merck & Co. continues to market branded Propecia, primarily to patients who specifically request or are prescribed the brand by their physician. However, the company's revenue and market strategy for finasteride are significantly influenced by the broader market demand for the generic version, which accounts for the vast majority of finasteride prescriptions.
Citations
[1] Kaufman, K. D., Olsen, E. A., Whiting, D., Savin, R. C., DeVillez, R. R., Bergfeld, W. F., ... & Rittmaster, R. S. (1998). Finasteride in the treatment of male pattern hair loss. Journal of the American Academy of Dermatology, 39(4), 578-589.
[2] U.S. Food and Drug Administration. (n.d.). Drug Approval & Labeling: Propecia (finasteride). Retrieved from [FDA Website (specific page not available publicly for historical drug approval details without deep search)]
[3] National Institutes of Health. (n.d.). U.S. Patent 4,377,584. Retrieved from USPTO patent database (searchable online).
[4] U.S. Food and Drug Administration. (1997, December 22). FDA Approves Propecia for the Treatment of Male Pattern Hair Loss. [Press Release].
[5] Irwig, M. S., & Kolukula, S. (2011). Persistent sexual side effects of finasteride for male pattern hair loss. The Journal of Sexual Medicine, 8(5), 1380-1383.
More… ↓
