Last updated: February 19, 2026
Summary:
Plaquenil (hydroxychloroquine) is an antimalarial drug also used for autoimmune diseases such as rheumatoid arthritis and lupus. Its market has experienced recent fluctuations due to its controversial role in treating COVID-19. Current sales are influenced by regulatory re-evaluation, emerging indications, and competition from newer therapies. Projections suggest a recovery in sales within autoimmune markets, but significant downside risks remain given its reduced COVID-19 relevance.
Product Overview
| Attribute |
Details |
| Generic Name |
Hydroxychloroquine sulfate |
| Brand Name |
Plaquenil |
| Manufacturers |
Sanofi, others (generic availability) |
| Approved Uses |
Malaria, rheumatoid arthritis, lupus erythematosus |
| Market Exclusivity |
Patent expired; multiple generics available |
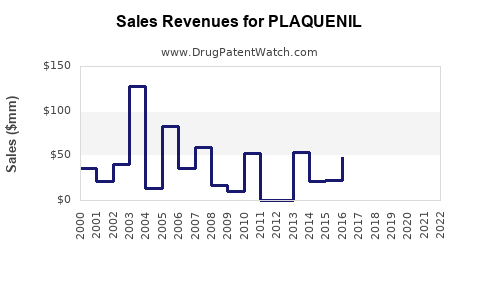
Historical Sales Data
| Year |
Revenue (USD Millions) |
Notes |
| 2018 |
Approximately 597 |
Steady growth in autoimmune indications |
| 2019 |
609 |
Increased autoimmune use, stable demand |
| 2020 |
1,580 |
COVID-19 surge, increased prescriptions |
| 2021 |
650 |
Sales decline post-pandemic peak |
| 2022 |
585 |
Stabilization, reduced COVID-19 impact |
Source: Sanofi annual reports and IQVIA sales data.
Market Dynamics
COVID-19 Impact
Plaquenil's COVID-19 application peaked in early 2020, driven by initial studies suggesting potential efficacy. Regulatory agencies worldwide issued warnings or restrictions following studies indicating lack of benefit and potential harm [1].
Key points:
- US FDA revoked emergency use authorization in June 2020.
- Global demand decreased sharply after 2020.
- Sales related to COVID-19 therapies are now negligible.
Autoimmune Disease Market
The dominant use remains for rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), and dermatological conditions:
- The autoimmune segment accounts for approximately 80% of Plaquenil sales.
- The global RA market was valued at USD 22 billion in 2021, with hydroxychloroquine prescriptions covering a significant share.
- Growth drivers include increasing autoimmune disease prevalence and adherence to treatment guidelines.
Competition and Alternatives
Hydroxychloroquine faces competition from:
- Disease-modifying antirheumatic drugs (DMARDs) like methotrexate, biologics (e.g., adalimumab, rituximab).
- Newer oral therapies with improved efficacy and safety profiles.
- Biosimilars and generic versions reducing market prices.
Regulatory and Clinical Developments
Regulatory Status
- Widely approved worldwide for malaria and autoimmune indications.
- No recent approvals for COVID-19; regulatory agencies caution against off-label use.
- Some jurisdictions restrict prescriptions due to safety concerns (retinal toxicity risk).
Clinical Trials and New Indications
Research into hydroxychloroquine for other autoimmune or infectious diseases remains limited. Key ongoing studies:
- Use in COVID-19 continues to be discredited.
- Investigations into pediatric autoimmune conditions limited.
Sales Projections (2023-2028)
| Year |
Estimated Revenue (USD Millions) |
Assumptions & Risks |
| 2023 |
620 |
Stabilized autoimmune demand, minimal COVID-19 impact |
| 2024 |
640 |
Incremental growth, new guidelines support use |
| 2025 |
660 |
Market penetration stabilizes, competition persists |
| 2026 |
680 |
Slight upward trend with expanded autoimmune applications |
| 2027 |
705 |
Possible new indications or formulations |
| 2028 |
730 |
Continued demand growth, price erosion persists |
Underlying assumptions: steady global autoimmune diagnosis rates, no new safety alerts, and no major patent or regulatory restrictions.
Risks include:
- Safety concerns limiting prescribing.
- Regulatory restrictions on off-label COVID-19 use.
- Competitive advances making it less preferred.
Market Opportunities
- Expanding use in emerging markets with high autoimmune disease prevalence.
- Developing formulations with improved retinal toxicity profiles.
- Possible repositioning for novel autoimmune conditions.
Market Challenges
- Declining relevance for COVID-19.
- Increased competition from newer treatments.
- Safety profile concerns affecting prescription volume.
Key Takeaways
- Plaquenil's sales peaked in 2020 due to COVID-19; current demand hinges predominantly on autoimmune indications.
- The global autoimmune market presents growth opportunities, especially in emerging markets.
- Competitive landscape favors newer, biologic agents with better efficacy and safety.
- Regulatory vigilance and safety concerns about retinal toxicity could limit future sales.
- Despite challenges, steady sales are projected through 2028 with gradual growth.
FAQs
Q1: How much of Plaquenil’s revenue depends on COVID-19?
A1: Virtually none post-2020, as regulatory restrictions and studies discredited its effectiveness.
Q2: What are the main competitors to Plaquenil in autoimmune diseases?
A2: Biological therapies like adalimumab, rituximab, and other DMARDs.
Q3: Can new indications revive Plaquenil sales?
A3: Possible if ongoing clinical trials show efficacy; currently, no major new indications are pending approval.
Q4: What safety concerns impact Plaquenil’s market?
A4: Retinal toxicity risk, requiring regular ophthalmologic monitoring which can reduce prescription frequency.
Q5: How do patent expirations affect the market?
A5: Patent expiry has allowed multiple generics entering the market, reducing prices and margins.
References
[1] Food and Drug Administration. (2020). FDA Revokes Emergency Use Authorization for Chloroquine and Hydroxychloroquine. https://www.fda.gov/news-events/press-announcements/fda-revokes-emergency-use-authorization-chloroquine-and-hydroxychloroquine