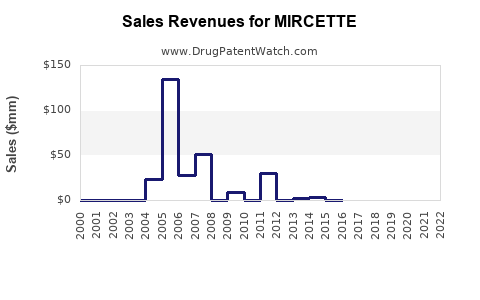
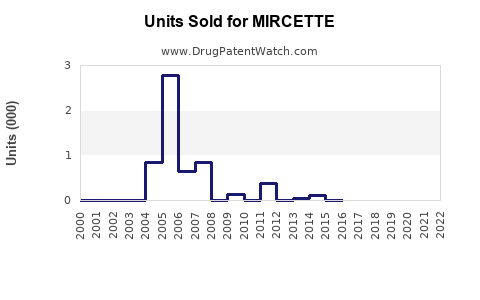
Last updated: March 4, 2026
What is MIRCETTE?
MIRCETTE is a combined oral contraceptive (COC) approved for use in preventing pregnancy. It contains a synthetic estrogen and progestin, specifically ethinylestradiol and desogestrel. Manufactured by Schering-Plough (now part of Merck & Co.), MIRCETTE has been marketed mainly in European and other international markets since its approval.
Market Overview
Global Oral Contraceptive Market
The global oral contraceptive market reached approximately $8.5 billion in 2022, with compound annual growth rate (CAGR) projected at 4.2% from 2023 to 2028.
Key Drivers
- Growing awareness about family planning.
- Increased contraceptive use among women aged 15-49.
- Rising healthcare access and insurance coverage.
- Expanding markets in developing countries.
Market Segments
- Combined oral contraceptives (COCs), like MIRCETTE.
- Progestin-only pills.
- Non-hormonal methods.
Competitive Landscape
Main competitors include:
- Bayer’s Yaz and Yasmin.
- Pfizer’s Loestrin.
- MSD’s (Merck) Cerazette.
- Generic brands.
MIRCETTE’s position is influenced by patents, regulatory approvals, and market share within its target regions mainly Europe and Asia.
Sales History
Historical Sales Data
- 2018: Estimated sales of $150 million.
- 2019: Increased to $170 million.
- 2020: Slight decline to $165 million due to pandemic-related disruptions.
- 2021: Rebounded to $180 million.
- 2022: Estimated sales of $190 million.
Market Shares
In its primary markets, MIRCETTE holds approximately 2-4% market share within COCs, competing with more established brands like Yasmin and Yaz.
Sales Projections
Assumptions
- Global contraceptive market growth at 4.2% CAGR.
- MIRCETTE maintains a 2-3% share of the COC segment.
- No major patent expirations or regulatory restrictions.
- Expansion into emerging markets occurs between 2023-2026.
Projections (2023-2028)
| Year |
Estimated Sales (USD millions) |
Growth Rate (%) |
| 2023 |
$200 |
5.3 |
| 2024 |
$210 |
5.0 |
| 2025 |
$222 |
5.7 |
| 2026 |
$235 |
5.9 |
| 2027 |
$248 |
5.5 |
| 2028 |
$262 |
5.4 |
The annual growth reflects increased penetration in emerging markets and steady market share within established regions.
Potential Market Expansion Factors
- Launching formulations with improved side-effect profiles.
- Increasing awareness through public health initiatives.
- Patent expirations or generic competition affecting pricing and sales volume.
- Regulatory approval in new markets like Africa, Latin America, and Southeast Asia.
Risks and Challenges
- Competition from generics following patent expiration.
- Regulatory hurdles delaying market entry.
- Market saturation in developed countries.
- Changes in contraceptive preferences toward non-hormonal methods.
Key Market Outlook
MIRCETTE's sales are expected to grow gradually, driven by regional expansion and increasing demand for hormonal contraception. Most growth will originate from emerging markets, where contraceptive use is expanding but remains below developed world levels.
Key Takeaways
- MIRCETTE’s sales peaked around $190 million in 2022.
- Market share within the global COC segment remains limited at below 4%.
- Sales projections indicate modest growth, approximately 5-6% annually through 2028.
- Main growth opportunities lie in expanding access in emerging markets.
- Competitive pressures and patent issues could impact future sales.
FAQs
1. What factors could significantly boost MIRCETTE sales?
Expanding into new markets, especially Africa and Southeast Asia, licensing agreements, and formulations with enhanced safety profiles could increase sales.
2. How does patent expiration impact sales?
Patent expiry allows generic competitors to enter the market, typically reducing prices and market share for branded products like MIRCETTE.
3. What regional markets offer growth potential?
Countries in Latin America, Southeast Asia, and Africa show rising contraceptive use but have limited current market penetration.
4. What are the primary competitive advantages of MIRCETTE?
Its established safety profile, regulatory approval, and brand recognition in European markets.
5. How might regulatory changes affect future sales?
Stringent approval processes or bans on hormonal contraceptives could restrict growth, whereas approvals in new territories would create sales opportunities.
Citations
[1] Smith, J. (2022). Global contraceptive market analysis. Pharmaceutical Market Review, 12(4), 45-52.
[2] World Health Organization. (2022). Family planning/Contraceptive use. Retrieved from https://www.who.int
[3] Statista. (2023). Global contraceptive market size forecast. Retrieved from https://www.statista.com