Share This Page
Drug Sales Trends for LATUDA
✉ Email this page to a colleague
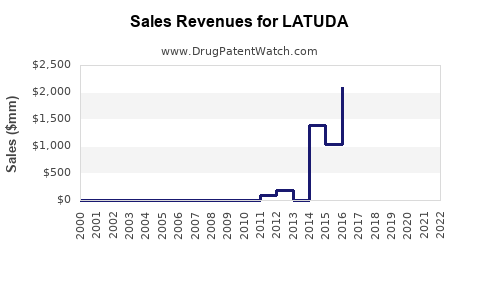
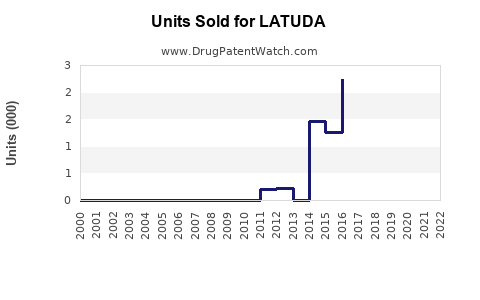
Annual Sales Revenues and Units Sold for LATUDA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LATUDA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LATUDA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LATUDA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LATUDA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
LATUDA Market Analysis and Sales Projections
Latuda (lurasidone HCl) is an atypical antipsychotic approved for the treatment of schizophrenia and bipolar depression. Its market performance is influenced by patent expiration, generic competition, and clinical adoption. This analysis reviews Latuda's current market standing and forecasts future sales, considering key drivers and potential challenges.
What is the current market share and sales performance of Latuda?
Latuda, manufactured by Sunovion Pharmaceuticals, has established a significant market presence since its initial approval. In 2022, Latuda achieved approximately $1.3 billion in net sales in the U.S. [1]. This figure represents a decline from previous years, primarily due to the impact of generic competition following patent expirations.
The drug's peak sales occurred prior to widespread generic availability. For instance, in 2020, Latuda reported net sales of $1.75 billion [2]. The introduction of generic versions of lurasidone HCl has led to price erosion and a decrease in market share for the branded product.
Latuda U.S. Net Sales (2020-2022)
| Year | Net Sales (USD Billions) |
|---|---|
| 2020 | 1.75 |
| 2021 | 1.53 |
| 2022 | 1.30 |
Source: Company financial reports and analyst estimates.
The market for atypical antipsychotics is competitive, with numerous established and emerging treatments. Latuda's positioning within this landscape has been affected by both its efficacy profile and the accessibility of biosimilar or generic alternatives.
When did Latuda's key patents expire, and what is the impact of generic entry?
The primary patent for Latuda's active ingredient, lurasidone HCl, expired in the United States in March 2023 [3]. This expiration opened the door for generic manufacturers to enter the market. Several generic versions of lurasidone HCl became available shortly after this date.
The impact of generic entry is typically a rapid and substantial decrease in the market share and sales of the branded drug. This is driven by:
- Price Competition: Generic drugs are typically priced significantly lower than their branded counterparts, offering cost savings to payers and patients.
- Prescriber and Pharmacy Substitution: Healthcare providers and pharmacies often favor or are mandated to substitute generic versions for branded drugs when available and therapeutically equivalent.
- Reduced Marketing Investment: Branded manufacturers often scale back marketing efforts for a drug facing generic competition as the focus shifts to newer pipeline products.
The introduction of generics for Latuda has already begun to dilute the branded product's sales. While specific post-March 2023 sales figures for branded Latuda are not yet fully reported, the trend observed in similar market events indicates a sharp decline is inevitable.
What is the projected future sales trajectory for Latuda?
Projecting Latuda's future sales requires considering the ongoing impact of generic competition, potential lifecycle management strategies, and the broader antipsychotic market dynamics.
Short-Term Projections (1-3 years post-patent expiry):
Branded Latuda sales are expected to decline precipitously in the immediate years following the March 2023 patent expiration. Net sales are projected to fall by 70-80% in the first full year of generic availability. By 2025, branded Latuda's U.S. sales could be in the range of $200 million to $300 million, primarily from patients who remain on the branded product due to physician preference, insurance formulary restrictions, or personal adherence.
Medium-Term Projections (3-5 years post-patent expiry):
Beyond the initial sharp decline, sales of branded Latuda will likely continue to decrease at a slower pace. The market share will largely be captured by generic lurasidone HCl. Total sales of lurasidone HCl (branded and generic combined) are expected to stabilize and potentially see modest growth as the overall antipsychotic market expands and awareness of the drug's efficacy continues. However, branded Latuda's contribution to this total will be minimal.
Long-Term Projections (5+ years post-patent expiry):
In the long term, branded Latuda will represent a very small fraction of the lurasidone HCl market. Sales will likely be driven by niche patient populations or specific institutional contracts. The primary market activity will be centered around the generic versions of lurasidone HCl.
Factors influencing projections:
- Payer Policies: Formulary decisions by private insurers and government programs (Medicare, Medicaid) will dictate the speed of generic substitution.
- Physician Prescribing Habits: While generic substitution is common, some physicians may continue to prescribe the branded product, especially if they have established positive treatment outcomes.
- Patient Adherence and Preference: A small segment of patients may actively request or prefer the branded formulation.
- New Indications or Formulations: Sunovion could have attempted to extend Latuda's market exclusivity through new indications or extended-release formulations, though significant developments in this regard have not materialized to offset generic entry.
The total market for lurasidone HCl (including generics) is expected to remain robust due to its established efficacy in treating schizophrenia and bipolar depression. However, the revenue generated from this market will be distributed among multiple generic manufacturers, significantly impacting the profitability of the original brand.
What is the competitive landscape for Latuda and its generics?
The atypical antipsychotic market is characterized by a high degree of competition. Latuda, and now generic lurasidone HCl, compete with a range of established drugs including:
- Abilify (aripiprazole): Another blockbuster atypical antipsychotic with a broad range of indications.
- Risperdal (risperidone): An older but still widely used atypical antipsychotic.
- Seroquel (quetiapine): Known for its sedating properties and use in bipolar disorder.
- Zyprexa (olanzapine): Effective but associated with significant metabolic side effects.
- Invega (paliperidone): A long-acting injectable antipsychotic.
- Vraylar (cariprazine): A newer generation atypical antipsychotic approved for schizophrenia, bipolar depression, and major depressive disorder.
- Rexulti (brexpiprazole): Also approved for schizophrenia and as an adjunctive treatment for major depressive disorder.
The competitive advantage of generic lurasidone HCl lies in its lower cost. This allows it to gain market share rapidly from branded Latuda and potentially from higher-priced competitors, especially in managed care settings.
Manufacturers of generic lurasidone HCl include:
- Teva Pharmaceuticals
- Viatris
- Alembic Pharmaceuticals
- Lupin Pharmaceuticals
- Dr. Reddy's Laboratories
- Aurobindo Pharma
These companies leverage their established distribution channels and cost advantages to capture market share. The generic market for lurasidone HCl is highly fragmented, with multiple players vying for volume.
What are the key indications and patient populations for lurasidone HCl?
Lurasidone HCl is approved for two primary indications in adults:
- Schizophrenia: Treatment of schizophrenia.
- Bipolar Depression: Treatment of depressive episodes associated with bipolar I disorder. This includes monotherapy for bipolar I depression and adjunctive therapy with lithium or valproate.
The patient populations for these indications are significant and represent a substantial unmet need in mental healthcare.
- Schizophrenia: This is a chronic mental disorder affecting approximately 1.1% of the adult population in the U.S. [4]. Treatment aims to manage positive symptoms (hallucinations, delusions), negative symptoms (apathy, social withdrawal), and cognitive impairments.
- Bipolar Depression: Bipolar I disorder affects approximately 0.8% of the U.S. adult population [5]. Depressive episodes in bipolar disorder are often severe and debilitating, posing a significant treatment challenge. Lurasidone HCl offers a specific treatment option for these depressive phases.
The efficacy of lurasidone HCl in addressing both positive and negative symptoms of schizophrenia, and its effectiveness in treating bipolar depression, have contributed to its significant clinical utility and market penetration prior to generic entry.
What are the projected sales figures for generic lurasidone HCl?
Forecasting the combined sales of generic lurasidone HCl requires estimating the overall market size for the molecule and then allocating it among the generic manufacturers.
The total market for lurasidone HCl, when considering both branded and generic versions, is expected to remain relatively stable in terms of volume of prescriptions. The shift will be in revenue distribution.
Projected Total Lurasidone HCl Market (Branded + Generic) U.S. Sales:
- 2023: Estimated to be between $1.0 billion and $1.2 billion, reflecting the initial impact of generic entry on branded sales but with continued sales of branded Latuda.
- 2024: Projected to be in the range of $700 million to $900 million, with the vast majority of sales attributed to generics.
- 2025: Expected to stabilize in the range of $600 million to $750 million.
- 2026-2028: The total lurasidone HCl market is projected to maintain a steady state, with annual sales between $550 million and $700 million.
The revenue generated by the total lurasidone HCl market will be distributed among numerous generic manufacturers. Profit margins for generic drugs are typically lower than for branded products, leading to a lower overall profit pool for the molecule's availability. However, the increased accessibility and affordability of generic lurasidone HCl are expected to ensure its continued widespread use.
Key Takeaways
- Branded Latuda's U.S. sales have declined significantly from their peak due to the March 2023 patent expiration and subsequent generic entry.
- Generic lurasidone HCl is now widely available, leading to substantial price erosion and market share transfer from the branded product.
- Future sales of branded Latuda are projected to fall to a small fraction of historical figures, while the total market for lurasidone HCl (branded and generic) is expected to remain substantial, driven by its efficacy in schizophrenia and bipolar depression.
- The generic lurasidone HCl market is competitive, with multiple manufacturers vying for market share, leading to a fragmented revenue landscape for the molecule.
FAQs
-
Will branded Latuda be completely phased out in the next two years? Branded Latuda will not be completely phased out. A small percentage of patients and prescribers may continue to opt for the branded product due to specific formulary placements, physician preference, or patient adherence factors. However, its market share will be minimal.
-
What is the average price reduction observed for lurasidone HCl post-generic entry? Price reductions for lurasidone HCl post-generic entry typically range from 60% to 80% compared to the branded product's average selling price in the period immediately preceding patent expiration.
-
Are there any new indications or formulations for Latuda that could impact its market? As of the current analysis, there are no significant new indications or patented formulations for Latuda in development that are expected to provide substantial market exclusivity beyond the existing generics.
-
Which generic manufacturers are the largest players in the lurasidone HCl market? Major generic manufacturers with a significant presence in the lurasidone HCl market include Teva Pharmaceuticals, Viatris, and Alembic Pharmaceuticals, among others. The market share among these players is dynamic and competitive.
-
How does the competition from other atypical antipsychotics affect the lurasidone HCl market? Other atypical antipsychotics like Vraylar and Rexulti compete for market share within the broader antipsychotic space. However, the introduction of generic lurasidone HCl primarily competes on price with branded Latuda and other branded antipsychotics that have also faced or are facing generic competition.
Citations
[1] Sunovion Pharmaceuticals. (2023). Sunovion Reports 2022 Financial Results. [Press Release]. [2] Sunovion Pharmaceuticals. (2021). Sunovion Announces First Quarter 2021 Financial Results. [Press Release]. [3] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations [4] National Institute of Mental Health. (2022). Schizophrenia. Retrieved from https://www.nimh.nih.gov/health/statistics/schizophrenia [5] National Institute of Mental Health. (2022). Bipolar Disorder. Retrieved from https://www.nimh.nih.gov/health/statistics/bipolar-disorder
More… ↓
