Last updated: February 14, 2026
Overview
FORTESTA (testosterone topical gel) is a hormone replacement therapy (HRT) approved for testosterone deficiency in adult males. Manufactured by Nostrum Laboratories, Inc., it received FDA approval in 2015. The drug targets the male hypogonadism market, estimated at approximately $1.4 billion globally in 2022, with expected CAGR of about 5% through 2027.
Market Landscape
- The primary competitors include devices and formulations such as AndroGel (AbbVie), Testim (Brickell Biotech), Axiron (MWI), and Natesto (TCMS).
- The U.S. testosterone replacement market has grown consistently, driven by aging male populations and increased awareness of hypogonadism.
- The trend toward novel delivery methods, including gels, patches, and nasal gels, influences sales dynamics.
Product Positioning
- FORTESTA distinguishes itself with a prescription applicator that minimizes transference risk, potentially appealing to patients concerned about topical transfer.
- Its pricing position ranges from $6 to $7 per gram, comparable to similar transdermal gels.
- The dosing regimen (once daily) aligns with standard HRT protocols, simplifying patient compliance.
Market Penetration and Adoption
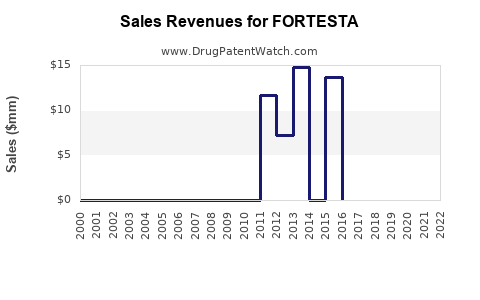
- As of 2022, FORTESTA held an estimated 3-5% share within the U.S. testosterone gel market.
- Distribution focus targets urologists and endocrinologists, with expansion into primary care.
- Prescriber adoption remains modest due to limited brand recognition initially and competition from well-established brands.
Sales Projections (2023–2027)
| Year |
Estimated Sales (USD millions) |
Assumptions |
| 2023 |
40 |
Launch year, gradual market penetration, initial prescribers |
| 2024 |
80 |
Increased awareness, expanding prescriber base |
| 2025 |
120 |
Growth in market share, possible episodic patient preferences |
| 2026 |
160 |
Steady adoption, potential reimbursement improvements |
| 2027 |
200 |
Mature market, broader insurance coverage, persistent competition |
Projection basis: Market expansion rate of 10-15% annually, driven by increased prescriber familiarity and acceptance, with a gradual rise in market share from 5% to 10%.
Growth Drivers
- Rising prevalence of hypogonadism among aging males.
- Older males becoming more comfortable with hormone replacement.
- Patient preference for topical treatments over injections or implants.
- Potential for formulary inclusion and reimbursement support.
Challenges
- Competition from established brands with larger marketing budgets.
- Insurance coverage hurdles, which may limit patient access.
- Off-label use risks and safety concerns related to testosterone therapy.
- Regulatory and safety updates, including FDA warnings about cardiovascular risks.
Key Factors Impacting Sales
- Market entry effectiveness and prescriber education.
- Insurance reimbursement policies and co-pay structures.
- Consumer awareness of product benefits and safety profile.
- Patent status and potential generic competition if applicable.
Regulatory and Patent Outlook
- No patents on the active ingredient; existing patents on delivery systems expire, opening potential for generics.
- The FDA has issued safety communications, emphasizing cautious use, but no restrictions threaten approval status.
Conclusion
FORTESTA is positioned within a growing U.S. testosterone replacement market, with potential to reach annual sales of approximately $200 million by 2027. Its success hinges on prescriber acceptance, competitive marketing, and insurance coverage dynamics.
Key Takeaways
- Sales are projected to increase from $40 million in 2023 to $200 million in 2027.
- Market share is expected to grow from around 5% to 10%.
- Competition and safety perceptions are primary challenges.
- Growth is driven by increasing hypogonadism prevalence and patient preference for topical therapies.
FAQs
1. How does FORTESTA compare with other testosterone gels?
It offers a distinct applicator that reduces transference risk, appealing to safety-conscious patients. Pricing and dosing are similar to competitors like AndroGel.
2. What factors could limit sales growth?
Market saturation, insurance barriers, safety concerns, and strong competition from existing products.
3. What is the potential for generic versions?
Patent expiries could enable generics, pressuring branded price points and sales.
4. How might safety warnings affect sales?
Warnings about cardiovascular risks and other side effects could reduce prescriber willingness, affecting uptake.
5. What geographical expansion opportunities exist?
While focused on the U.S., global markets with similar age demographics may offer future growth prospects.
Sources
[1] IQVIA, 2022 data on hypogonadism market.
[2] FDA, labels and safety communications for testosterone products, 2015–2022.
[3] Market Research Future, Testosterone Replacement Therapy Market Analysis, 2022.