Last updated: February 13, 2026
What is the Current Market Landscape for EVISTA?
EVISTA (raloxifene) is a selective estrogen receptor modulator (SERM) approved primarily for osteoporosis prevention and breast cancer risk reduction in postmenopausal women. Since its approval by the FDA in 2007, its market has experienced steady demand, driven by the aging global population and increasing awareness of osteoporosis management.
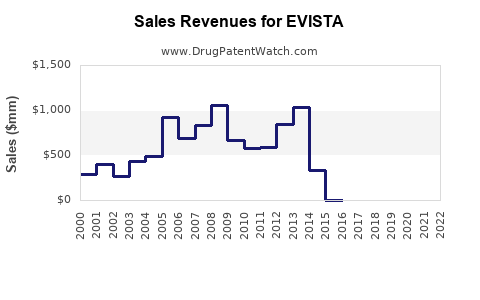
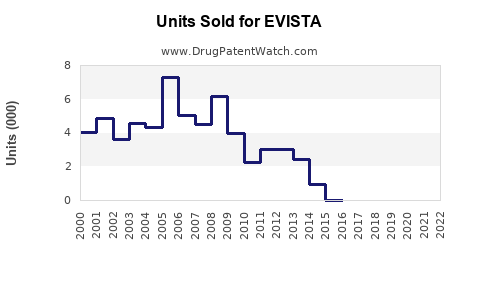
The drug's global sales peaked at approximately $500 million in 2015, followed by fluctuations attributable to generic competition, patent expirations, and evolving treatment guidelines. In 2022, global sales were around $350 million, reflecting market saturation in developed economies and slower adoption in emerging markets.
Major markets include the U.S., Europe, Japan, and select Asian countries. The U.S. accounts for roughly 60% of EVISTA's sales, with prescriptions clustered among osteoporosis and breast cancer risk management indications.
What Are the Key Market Drivers and Barriers?
Drivers
- Increasing prevalence of osteoporosis, projected to reach 200 million cases worldwide by 2050 (WHO).
- Rising postmenopausal female population, especially in Asia, with an estimated 400 million women aged 50+ in 2025.
- Clinical guidelines endorsing EVISTA as a first-line preventive therapy for osteoporosis in high-risk women.
- Concerns over the safety profile of bisphosphonates, leading physicians to favor SERMs like EVISTA.
Barriers
- Competition from bisphosphonates (alendronate, risedronate) and newer agents such as denosumab.
- The availability of generics post-patent expiry diminishes prices, reducing profit margins.
- Side effect profile, including hot flashes and increased risk of thromboembolism, which may limit patient adherence.
- Limited indication scope compared to broader osteoporotic treatments.
What Are the Sales Projections for EVISTA?
Sales are projected to decline modestly over the next five years due to generic erosion but will maintain a niche presence in specific indications.
| Year |
Estimated Global Sales (USD millions) |
Growth/Decline % |
| 2023 |
330 |
-5.7% |
| 2024 |
310 |
-6.1% |
| 2025 |
290 |
-6.5% |
| 2026 |
270 |
-6.9% |
| 2027 |
250 |
-7.4% |
The decline rate approximates 6-7% annually, driven by generic availability and competitive therapies. Name-brand EVISTA maintains fewer than 10% of total sales post-patent expiry, primarily in patients intolerant to or contraindicated for bisphosphonates.
How Do Patent and Regulatory Changes Impact Market Outlook?
The original patent expired globally between 2013 and 2015. Generic versions entered the market shortly after, causing immediate sales erosion. Certain formulations of raloxifene remained protected under minor formulation patents, extending market exclusivity until approximately 2022 in some jurisdictions.
Regulatory shifts, such as heightened safety reviews for SERMs, influence prescribing behaviors. New guidelines tend to favor bisphosphonates, which improves market share for generic options.
What Are Future Opportunities and Risks?
Opportunities
- Development of combination therapies to enhance efficacy.
- Expanding indications, such as osteoporosis in men and other postmenopausal conditions.
- Entering emerging markets with tailored marketing strategies.
Risks
- Competition from newer pharmacologic agents with better safety profiles.
- Evolving clinical guidelines favoring other treatment classes.
- Pricing pressures and reimbursement constraints, especially in cost-sensitive markets.
Summary
EVISTA’s market is stagnating with a slow decline forecast over the coming years. It remains relevant for niche patient populations contraindicated for alternative therapies. Its sales trajectory is heavily influenced by generic competition, safety profile perceptions, and treatment guideline evolution.
Key Takeaways
- EVISTA’s global sales declined from $500 million (2015) to approximately $350 million (2022).
- Sales are projected to decrease 6-7% annually over five years, driven by generics.
- The primary markets are the U.S. and Europe, with growth potential in Asia.
- Competition from bisphosphonates and newer agents remains significant.
- Patent expirations and safety concerns shape future market dynamics.
FAQs
Q1: Will EVISTA regain market share with new formulations or indications?
No definitive evidence suggests new formulations will significantly expand its market share given existing competition and safety concerns.
Q2: Are there any ongoing clinical trials that could influence EVISTA’s market?
No currently active major trials are expected to extend EVISTA’s indications or significantly alter its competitive position.
Q3: How does safety profile influence physician prescribing habits?
Concerns about thromboembolism and hot flashes can reduce prescription rates, especially as alternative therapies demonstrate favorable safety profiles.
Q4: What is the impact of biosimilars or generics on EVISTA’s future?
Generic raloxifene drastically reduces brand-name sales, with current formulations primarily purchased as generics.
Q5: Are emerging markets a growth avenue for EVISTA?
Potential exists, particularly where osteoporosis awareness is expanding, but price sensitivity limits profitability unless local market access improves.
References
[1] World Health Organization. Osteoporosis Fact Sheet. 2021.
[2] IQVIA. Global Medicine Forecast 2022.
[3] U.S. Food & Drug Administration. EVISTA (raloxifene) prescribing information. 2007.
[4] EvaluatePharma. 2022 Market Data.
[5] ClinicalTrials.gov. Ongoing studies related to raloxifene.