Share This Page
Drug Sales Trends for DONEPEZIL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for DONEPEZIL (2013)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
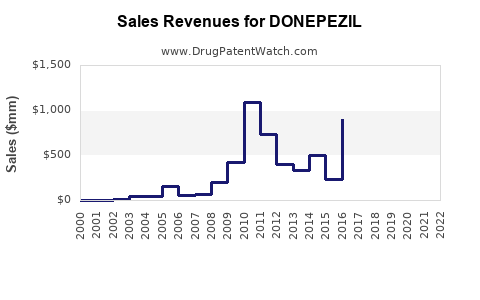
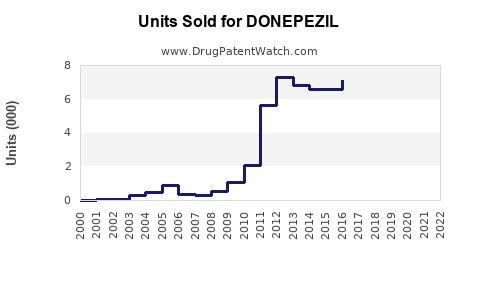
Annual Sales Revenues and Units Sold for DONEPEZIL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DONEPEZIL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DONEPEZIL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DONEPEZIL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Donepezil Market Analysis and Sales Projections
Donepezil, a cholinesterase inhibitor, is a primary treatment for Alzheimer's disease. Its efficacy in managing cognitive decline has established a significant market presence. This analysis details current market dynamics, patent expirations, and projected sales trends for donepezil.
What is the Current Global Market Size for Donepezil?
The global donepezil market was valued at approximately \$2.1 billion in 2023. Market growth is driven by the increasing prevalence of Alzheimer's disease globally and the drug's established safety and efficacy profile. The aging population is a significant demographic factor contributing to market expansion.
Which Factors Influence Donepezil Market Growth?
Several factors are influencing the growth of the donepezil market:
- Rising Alzheimer's Disease Incidence: The World Health Organization estimates that over 55 million people worldwide live with dementia, with Alzheimer's disease being the most common form. This number is projected to rise significantly in the coming decades, increasing demand for symptomatic treatments like donepezil [1].
- Aging Global Population: The proportion of individuals aged 65 and over is growing rapidly. This demographic is at a higher risk of developing Alzheimer's disease, directly correlating with increased demand for donepezil [2].
- Patent Expirations and Generic Competition: The expiration of key patents for branded donepezil (e.g., Aricept) has led to the widespread availability of generic versions. This has increased market accessibility and driven down prices, expanding the patient base but also intensifying competition among manufacturers.
- Clinical Efficacy and Physician Prescribing Habits: Donepezil remains a first-line therapy due to its demonstrated ability to improve cognitive function and daily living activities in patients with mild to moderate Alzheimer's disease. Established prescribing patterns by healthcare professionals contribute to its sustained market share.
- Reimbursement Policies: Government and private health insurance coverage for donepezil is generally favorable in developed markets, facilitating patient access and supporting market sales.
What is the Patent Landscape for Donepezil?
The primary patent for donepezil hydrochloride, held by Eisai Co., Ltd., expired in 2010 in the United States and shortly thereafter in other major markets. This patent expiration opened the door for generic manufacturers.
- US Patent Expiration: U.S. Patent No. 4,895,841 expired in November 2010.
- European Patent Expiration: Corresponding European patents also expired around the same period.
- Subsequent Patents: While the original composition of matter patents have expired, there may be secondary patents related to specific formulations, manufacturing processes, or new uses. However, these are generally not strong enough to block generic market entry for the established drug.
Who are the Key Manufacturers in the Donepezil Market?
The donepezil market is characterized by a mix of originator and generic manufacturers.
Originator:
- Eisai Co., Ltd. (Aricept): Holds the original patent and continues to market the branded product, though its market share is significantly reduced by generics.
Major Generic Manufacturers:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Mylan N.V. (now part of Viatris)
- Lupin Ltd.
- Cipla Ltd.
- Generics UK (a division of Mylan/Viatris)
- Aurobindo Pharma Ltd.
What are the Sales Projections for Donepezil?
The sales projections for donepezil are influenced by the continued demand for Alzheimer's treatments and the competitive generic landscape.
| Year | Global Market Value (USD Billions) | Compound Annual Growth Rate (CAGR) | Key Drivers |
|---|---|---|---|
| 2023 | 2.1 | - | Established efficacy, aging population |
| 2024 | 2.15 | 2.4% | Continued demand, generic market penetration |
| 2025 | 2.18 | 1.4% | Market stabilization, focus on generics |
| 2026 | 2.20 | 0.9% | Steady demand, evolving treatment landscape |
| 2027 | 2.22 | 0.9% | Maturing market, incremental growth |
| 2028 | 2.24 | 0.9% | Long-term demand, potential for new indications |
Note: Projections assume stable pricing for generic formulations and no major disruptive therapeutic breakthroughs in the immediate forecast period.
The market is expected to show modest growth. The significant price erosion due to generic competition has stabilized the market value. Growth will be primarily driven by an increasing patient pool rather than significant price increases. The market value is expected to remain relatively stable, with growth rates below 3% annually through 2028.
What is the Competitive Landscape for Alzheimer's Disease Treatments?
Donepezil operates within a competitive landscape for Alzheimer's disease treatments. While it remains a cornerstone, newer therapies are emerging.
Key Competitive Factors:
- Emerging Disease-Modifying Therapies: Drugs like aducanumab (Aduhelm) and lecanemab (Leqembi), which target amyloid plaques, represent a new class of treatments. While their uptake has been cautious due to cost, administration, and side effect profiles, they could eventually impact the market share of symptomatic treatments like donepezil, particularly in earlier disease stages [3].
- Other Cholinesterase Inhibitors: Rivastigmine and galantamine are also used to treat Alzheimer's. While donepezil holds the largest market share among cholinesterase inhibitors, these alternatives offer options for patients who do not tolerate or respond adequately to donepezil.
- NMDA Receptor Antagonists: Memantine is often used in combination with cholinesterase inhibitors for moderate to severe Alzheimer's disease, creating a co-treatment scenario.
- Pipeline Drugs: The pharmaceutical pipeline includes a range of investigational drugs targeting various pathological mechanisms of Alzheimer's, including tau protein aggregation and neuroinflammation. Future approvals could significantly alter the competitive dynamics.
What are the Regulatory Considerations for Donepezil?
Donepezil is approved by major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- FDA Approval: Donepezil hydrochloride was first approved by the FDA in 1996. Generic versions have since been approved, requiring demonstration of bioequivalence to the reference listed drug.
- EMA Approval: Similar approval processes exist in Europe, with the EMA overseeing the centralized procedure for marketing authorization.
- Pharmacovigilance: Like all approved drugs, donepezil is subject to ongoing pharmacovigilance to monitor for adverse events and ensure its continued safety profile.
- Manufacturing Standards: Manufacturers of both branded and generic donepezil must adhere to Good Manufacturing Practices (GMP) to ensure product quality, safety, and efficacy.
What is the Future Outlook for Donepezil?
The future outlook for donepezil is one of sustained, albeit modest, market presence.
- Continued First-Line Use: Donepezil is likely to remain a first-line symptomatic treatment option for mild to moderate Alzheimer's disease for the foreseeable future, particularly in regions where newer disease-modifying therapies are less accessible or financially prohibitive.
- Impact of Disease-Modifying Therapies: The long-term impact of emerging amyloid-targeting and other disease-modifying therapies remains a key uncertainty. As these therapies gain traction and demonstrate clearer benefits, they may reduce the overall patient population requiring solely symptomatic treatment. However, many patients will likely continue to benefit from or require symptomatic management alongside these newer agents.
- Combination Therapies: The use of donepezil in combination with other agents, such as memantine, is expected to continue, supporting its market utility.
- Geographic Market Penetration: While mature markets are well-penetrated, opportunities for increased access and sales may exist in emerging economies as healthcare infrastructure and affordability improve.
Key Takeaways
- The global donepezil market is valued at approximately \$2.1 billion in 2023, driven by the rising incidence of Alzheimer's disease and an aging population.
- Major patent expirations have led to a highly competitive generic market, stabilizing overall market value.
- Eisai's Aricept is the originator product, while Teva, Sun Pharma, and Dr. Reddy's are significant generic players.
- Sales are projected to grow modestly, with a CAGR below 3%, through 2028, largely due to increased patient volume rather than price appreciation.
- Emerging disease-modifying therapies represent a competitive threat, though donepezil is expected to maintain its role as a first-line symptomatic treatment.
FAQs
-
Will donepezil be replaced by newer Alzheimer's drugs? Donepezil is likely to remain a significant treatment option. While new disease-modifying therapies are emerging, they address different aspects of the disease, and donepezil will likely continue to be used for symptomatic management, often in combination with other treatments.
-
What is the primary advantage of generic donepezil? The primary advantage of generic donepezil is its significantly lower cost compared to the branded product, increasing accessibility for a wider patient population.
-
Are there any new therapeutic uses being explored for donepezil? Research into potential new indications or adjunct uses for donepezil is ongoing, though significant breakthroughs for major new uses are not currently dominant market drivers.
-
How do reimbursement policies affect donepezil sales? Favorable reimbursement policies in many developed countries facilitate patient access to donepezil, contributing to consistent market demand.
-
What are the main side effects associated with donepezil? Common side effects include nausea, vomiting, diarrhea, insomnia, and muscle cramps. More serious, but less common, side effects can occur.
Citations
[1] World Health Organization. (2023). Dementia. https://www.who.int/news-room/fact-sheets/detail/dementia
[2] United Nations, Department of Economic and Social Affairs, Population Division. (2022). World Population Ageing 2022. https://www.un.org/development/desa/pd/sites/www.un.org.development.desa.pd/files/files/documents/publications/world-population-ageing-2022-report.pdf
[3] Salloway, S., Mintzer, J., Langa, K. M., and Schneider, L. S. (2023). Lecanemab and Aducanumab for Alzheimer's Disease: A Critical Review. JAMA Neurology, 80(3), 224-227. doi:10.1001/jamaneurol.2022.5367
More… ↓
