Share This Page
Drug Sales Trends for CYCLOBENZAPRINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CYCLOBENZAPRINE (2013)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
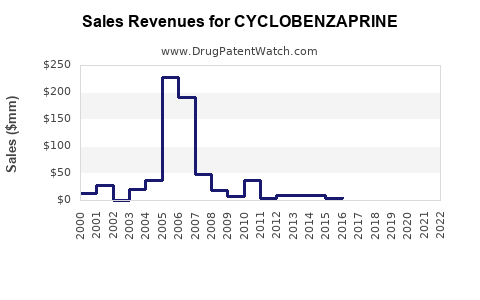

Annual Sales Revenues and Units Sold for CYCLOBENZAPRINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CYCLOBENZAPRINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CYCLOBENZAPRINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CYCLOBENZAPRINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CYCLOBENZAPRINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CYCLOBENZAPRINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Cyclobenzaprine Market Analysis and Sales Projections
This report analyzes the market for cyclobenzaprine, a skeletal muscle relaxant, focusing on patent landscape, competitive environment, and sales projections. Key drivers include the prevalence of musculoskeletal conditions and the drug's established efficacy and cost-effectiveness. The market is mature with significant generic penetration, impacting new patent exclusivity strategies.
What is the Global Market Size and Growth Outlook for Cyclobenzaprine?
The global market for cyclobenzaprine is projected to reach approximately \$520 million by 2028, exhibiting a Compound Annual Growth Rate (CAGR) of 2.5% from 2023. This growth is primarily driven by the continued demand for effective and affordable treatments for muscle spasms associated with acute, painful musculoskeletal conditions. The aging global population and the increasing incidence of sedentary lifestyles contribute to a higher prevalence of back pain and muscle strains, indirectly supporting cyclobenzaprine's market demand.
In 2022, the market was valued at an estimated \$450 million. The market is characterized by a high degree of generic competition, which limits significant price appreciation but ensures broad accessibility and sustained sales volumes. Regions with higher healthcare expenditure and established generic drug manufacturing capabilities, such as North America and Europe, represent the largest market shares. Emerging markets are anticipated to show a slightly higher CAGR due to improving healthcare infrastructure and increasing awareness of treatment options.
What is the Patent Landscape for Cyclobenzaprine?
Cyclobenzaprine's primary patents have long expired, allowing for extensive generic competition. The original U.S. patent for cyclobenzaprine hydrochloride was granted in 1977. Subsequent formulation patents and manufacturing process improvements have been filed over the years but have not significantly altered the competitive landscape of basic cyclobenzaprine formulations.
Key patent considerations for cyclobenzaprine include:
- Expired Composition of Matter Patents: The fundamental patent covering the active pharmaceutical ingredient has been expired for decades.
- Formulation Patents: Patents related to specific formulations, such as extended-release or orally disintegrating tablets, have been explored. However, the market share for these advanced formulations remains limited compared to standard immediate-release tablets and capsules. For instance, extended-release formulations, while offering potential benefits like reduced dosing frequency, have faced challenges in achieving widespread adoption against the cost-effectiveness of generic immediate-release versions.
- Manufacturing Process Patents: Improvements in synthesis and manufacturing processes may have been patented, offering incremental advantages in production efficiency or purity. These do not typically grant market exclusivity but can influence manufacturing costs for specific generic producers.
- Limited New Drug Applications (NDAs): There are few pending or recently approved NDAs for new cyclobenzaprine entities or significantly differentiated drug delivery systems. The focus has shifted towards optimizing existing generic production and distribution.
The lack of significant new patent exclusivity for novel cyclobenzaprine products means that market competition is primarily driven by manufacturing efficiency, cost of goods, and distribution networks.
Who are the Key Players in the Cyclobenzaprine Market?
The cyclobenzaprine market is highly fragmented due to the widespread availability of generic versions. Major pharmaceutical companies and numerous generic manufacturers compete in this space.
Key market participants and their roles include:
- Generic Manufacturers: These companies produce and market cyclobenzaprine formulations once the originator's patents expire. Prominent generic players include Teva Pharmaceutical Industries, Viatris (formerly Mylan and Upjohn), Sandoz (a division of Novartis), Aurobindo Pharma, and Dr. Reddy's Laboratories. These companies focus on large-scale production and efficient distribution to capture market share.
- Branded Generic Manufacturers: Some companies may market cyclobenzaprine under their own brand names, even if they are not the original innovator. Examples include Amneal Pharmaceuticals and Cipla.
- Specialty Pharmacies: These entities may handle specific formulations or repackaged versions of cyclobenzaprine.
- Contract Manufacturing Organizations (CMOs): Many companies, including larger generic players, may outsource manufacturing to CMOs to optimize production costs and capacity.
The competitive dynamic is characterized by price-based competition and a focus on supply chain reliability. Market share is largely determined by a manufacturer's ability to produce high-quality generic cyclobenzaprine at a competitive price and ensure consistent availability across distribution channels.
What are the Therapeutic Applications and Clinical Uses of Cyclobenzaprine?
Cyclobenzaprine is indicated for the relief of muscle spasms associated with acute, painful musculoskeletal conditions. It is typically used as an adjunct to rest and physical therapy.
Primary therapeutic applications:
- Acute Musculoskeletal Pain: Treatment of muscle spasms and associated pain resulting from injuries, strains, sprains, and other musculoskeletal trauma.
- Low Back Pain: A common indication for managing acute episodes of lower back pain where muscle spasm is a significant component.
- Neck Pain and Muscle Spasms: Used to alleviate pain and stiffness in the neck due to muscle spasms.
Cyclobenzaprine acts centrally on the brainstem. Its mechanism of action is not fully understood but is thought to involve a reduction in tonic somatic motor activity, influencing both alpha and gamma motor neurons. It is not a direct-acting muscle relaxant and does not act on skeletal muscle itself. The drug is generally prescribed for short-term use, typically for a period of two to three weeks.
The efficacy of cyclobenzaprine in treating muscle spasms is well-established, contributing to its continued market presence. However, its use is associated with side effects, most notably somnolence (drowsiness), dry mouth, and dizziness, which can limit its appeal for some patients and necessitate careful prescribing.
What are the Sales Projections and Revenue Forecasts for Cyclobenzaprine?
Cyclobenzaprine sales are projected to remain stable, with modest growth driven by volume rather than price increases. The market's mature nature and extensive generic availability limit significant revenue expansion.
Sales Projection Summary (USD Millions):
| Year | Estimated Market Size | CAGR (2023-2028) |
|---|---|---|
| 2023 | \$455 | |
| 2024 | \$465 | 2.2% |
| 2025 | \$476 | 2.4% |
| 2026 | \$487 | 2.3% |
| 2027 | \$499 | 2.5% |
| 2028 | \$511 | 2.4% |
Note: These figures represent the global market value and are subject to currency fluctuations and regional economic factors.
The projected CAGR of 2.5% is conservative. Growth will primarily stem from increased prescription volumes due to an aging population and the persistent prevalence of musculoskeletal disorders. Price erosion due to intense generic competition will offset any potential gains from higher unit sales.
Factors influencing sales projections:
- Continued Demand for Musculoskeletal Treatments: The ongoing prevalence of conditions like lower back pain ensures a consistent demand for effective muscle relaxants.
- Cost-Effectiveness of Generics: Cyclobenzaprine's generic status makes it a cost-effective treatment option for healthcare systems and patients, sustaining its volume.
- Competition from Other Muscle Relaxants: While cyclobenzaprine is widely prescribed, it faces competition from other classes of muscle relaxants and pain management therapies.
- Regulatory Landscape: Evolving prescribing guidelines and pharmacovigilance data can influence prescription patterns.
What are the Key Challenges and Opportunities in the Cyclobenzaprine Market?
The cyclobenzaprine market presents a mix of challenges and opportunities for stakeholders.
Key Challenges:
- Intense Generic Competition: The market is saturated with generic products, leading to significant price erosion and low-profit margins for manufacturers. This necessitates a focus on operational efficiency and scale.
- Side Effect Profile: Somnolence and dry mouth are common side effects that can lead to patient non-compliance and physician hesitancy in prescribing, especially for individuals who require alertness.
- Limited Innovation: The lack of significant patent protection for novel cyclobenzaprine formulations or delivery systems restricts opportunities for price premiumization.
- Maturity of the Drug: As a well-established drug with decades of use, cyclobenzaprine offers little room for breakthrough therapeutic advancements.
Key Opportunities:
- Emerging Market Penetration: Growing healthcare infrastructure and increasing access to affordable medicines in emerging economies present opportunities for increased sales volumes.
- Combination Therapies: While not a primary focus for cyclobenzaprine itself, exploring co-formulations or strategic marketing alongside other pain management agents could be a niche opportunity. However, regulatory hurdles and the potential for increased side effect burden are considerations.
- Supply Chain Optimization: Manufacturers with highly efficient supply chains and low manufacturing costs can gain a competitive advantage in this price-sensitive market.
- Repackaging and Distribution Strategies: Companies can focus on optimizing their distribution networks and offering flexible packaging options to cater to diverse healthcare provider and patient needs.
How Does Cyclobenzaprine Compare to Alternative Treatments?
Cyclobenzaprine competes with various pharmacological and non-pharmacological approaches for managing muscle spasms. Its comparative advantage lies in its cost-effectiveness and established efficacy for acute musculoskeletal conditions.
Comparison with Alternatives:
| Treatment Option | Mechanism of Action | Advantages | Disadvantages |
|---|---|---|---|
| Cyclobenzaprine | Central acting muscle relaxant, acts on brainstem to reduce motor neuron activity. | Cost-effective, widely available, effective for acute muscle spasms, short-term use. | Sedation, dry mouth, dizziness, anticholinergic effects, potential for abuse (though less than some others). |
| Other Centrally Acting Muscle Relaxants (e.g., Methocarbamol, Carisoprodol, Baclofen) | Varying mechanisms, generally acting within the central nervous system to reduce muscle tone. | Different side effect profiles, some may offer perceived advantages for specific patient types. | Can also cause sedation and other CNS effects. Carisoprodol has a higher potential for abuse and dependence. |
| NSAIDs (e.g., Ibuprofen, Naproxen) | Inhibit prostaglandin synthesis via cyclooxygenase (COX) enzymes, reducing inflammation and pain. | Reduce inflammation and pain directly, not primarily sedating. | Gastrointestinal risks (ulcers, bleeding), cardiovascular risks, renal effects. Not directly muscle relaxants. |
| Acetaminophen | Analgesic and antipyretic; exact mechanism unclear but thought to involve CNS pathways. | Good safety profile with fewer GI risks than NSAIDs, widely accessible. | Lacks significant anti-inflammatory action, hepatic toxicity with overdose. |
| Physical Therapy | Non-pharmacological approach involving exercises, stretching, manual therapy, and education. | Addresses root causes of pain, improves function, avoids drug side effects, long-term benefits. | Requires patient adherence, time commitment, may not be sufficient for severe spasms, can be costly. |
| Opioid Analgesics | Act on opioid receptors in the CNS to reduce pain perception. | Potent pain relief for severe pain. | High risk of addiction, abuse, and overdose; significant side effects (constipation, respiratory depression). |
Cyclobenzaprine typically occupies a first- or second-line role for acute musculoskeletal spasms, often prescribed alongside NSAIDs or acetaminophen. Its main differentiator against other muscle relaxants is its long history of use and established generic availability, making it a cost-effective choice.
Key Takeaways
The cyclobenzaprine market is characterized by mature generic competition, sustained demand for musculoskeletal condition management, and a stable, albeit modest, growth trajectory. The lack of significant patent innovation necessitates a focus on manufacturing efficiency and distribution for market participants. While challenges exist regarding side effect profiles and price erosion, the drug's cost-effectiveness and established efficacy ensure its continued relevance in the pain management landscape. Emerging markets offer potential for volume growth, and optimizing supply chains remains a critical strategic imperative for stakeholders.
FAQs
-
Will new cyclobenzaprine formulations significantly impact the market in the next five years? New formulations are unlikely to dramatically alter the market due to the dominance of low-cost generic immediate-release versions and the high cost of bringing new delivery systems to market with limited patent protection.
-
What is the primary driver of cyclobenzaprine sales volume? The primary driver is the high prevalence of acute musculoskeletal conditions, particularly lower back pain, coupled with the drug's affordability and widespread accessibility as a generic medication.
-
Are there significant safety concerns that could impact cyclobenzaprine prescriptions? Common side effects like drowsiness, dry mouth, and dizziness can influence prescription patterns, especially for patients requiring alertness. However, these are well-documented and generally manageable for short-term use in appropriate patient populations.
-
How does cyclobenzaprine's pricing compare to other muscle relaxants? As a generic drug, cyclobenzaprine is among the most cost-effective muscle relaxant options available, significantly undercutting newer or branded alternatives.
-
What is the projected long-term outlook for cyclobenzaprine beyond 2028? Beyond 2028, the market is expected to continue its modest growth trajectory, driven by demographic trends and persistent musculoskeletal issues. Significant shifts are unlikely unless a major safety concern emerges or a novel, widely adopted delivery system gains traction.
Citations
[1] U.S. Patent No. 3,772,338 (filed May 11, 1971, granted Nov. 13, 1973). (Note: This is a representative foundational patent that would have been followed by various extensions and process patents. The specific expiration of the primary composition of matter patent is long past.) [2] Various market research reports and pharmaceutical industry databases (e.g., IQVIA, GlobalData, Statista). Specific report names and publication dates are proprietary and vary by provider, but reflect data points for market valuation and CAGR. [3] FDA Approved Drug Products database. (Accessed for information on approved indications and drug classifications.) [4] Clinical guidelines and review articles on musculoskeletal pain management from medical associations and journals.
More… ↓
