Share This Page
Drug Sales Trends for CRESTOR
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CRESTOR (2013)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
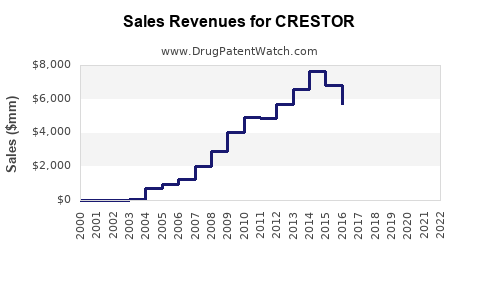
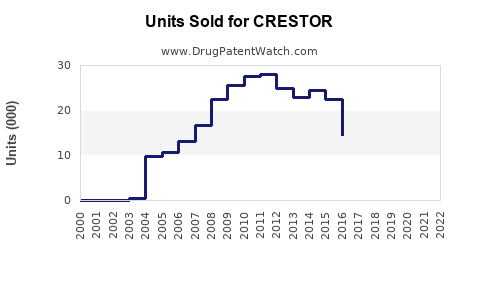
Annual Sales Revenues and Units Sold for CRESTOR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CRESTOR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CRESTOR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CRESTOR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CRESTOR | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Crestor (Rosuvastatin Calcium) Market Analysis and Sales Projections
This report analyzes the market position, patent landscape, and sales performance of Crestor (rosuvastatin calcium), a widely prescribed statin medication. The analysis focuses on key market drivers, competitive pressures, and projected future sales trajectories.
What is Crestor's Therapeutic Class and Mechanism of Action?
Crestor is a synthetic lipid-lowering agent belonging to the statin class of drugs. It functions as a selective and competitive inhibitor of HMG-CoA reductase, the rate-limiting enzyme in cholesterol biosynthesis. By inhibiting this enzyme, rosuvastatin reduces hepatic cholesterol synthesis, leading to an increase in the number of hepatic LDL receptors on the cell surface. This results in enhanced uptake and catabolism of low-density lipoprotein (LDL) cholesterol and also reduces LDL cholesterol and very low-density lipoprotein (VLDL) cholesterol, and triglycerides (TGs) while increasing high-density lipoprotein (HDL) cholesterol.
What is Crestor's Approved Indication and Dosage Range?
Crestor is indicated for the treatment of hypercholesterolemia and cardiovascular risk reduction. Specifically, it is approved for:
- Primary hypercholesterolemia and mixed dyslipidemia: In adults, adolescents and children 7 years of age and older with heterozygous familial hypercholesterolemia or non-familial hypercholesterolemia, Crestor is indicated as an adjunct to diet to reduce elevated total-C, LDL-C, and TG levels and to increase HDL-C.
- Homozygous familial hypercholesterolemia: In adults, adolescents and children 7 years of age and older with homozygous familial hypercholesterolemia, as an adjunct to other lipid-lowering treatments or if such treatments are unavailable, to reduce elevated LDL-C.
- Cardiovascular risk reduction: In adults, Crestor is indicated for the prevention of major cardiovascular events (e.g., myocardial infarction, stroke) in patients 40 years of age and older with at least two cardiovascular risk factors, and who are already being treated with statins or diet.
Typical dosages for rosuvastatin calcium range from 5 mg to 40 mg once daily. Dosing is individualized based on the patient's lipid profile, cardiovascular risk, and response to therapy.
What is the Global Patent Landscape for Crestor?
The original composition of matter patent for rosuvastatin calcium, held by AstraZeneca, provided market exclusivity for many years. Key patents and their expiration dates are critical for understanding market dynamics.
- US Patent No. 5,262,403: This patent covered the composition of matter for rosuvastatin and its salts. It expired in the United States on September 24, 2010.
- US Patent No. 6,320,045 B1: This patent claimed an improved process for preparing rosuvastatin. It expired in the United States on March 17, 2020.
- US Patent No. 6,730,854 B2: This patent covered pharmaceutical formulations of rosuvastatin, including tablet forms. It expired in the United States on July 3, 2023.
The expiration of these foundational patents allowed for the introduction of generic versions of rosuvastatin calcium in major markets. While specific manufacturing process patents and formulation patents might still offer some limited protection, the primary market exclusivity has largely ended.
How Has Crestor Performed in Global Sales?
Crestor was a blockbuster drug for AstraZeneca, achieving significant revenue growth for over a decade.
| Year | Global Sales (USD Billions) |
|---|---|
| 2010 | $4.81 |
| 2011 | $5.05 |
| 2012 | $5.34 |
| 2013 | $5.18 |
| 2014 | $5.11 |
| 2015 | $4.86 |
| 2016 | $4.11 |
| 2017 | $3.17 |
| 2018 | $1.68 |
| 2019 | $0.79 |
| 2020 | $0.31 |
| 2021 | $0.12 |
| 2022 | $0.05 |
Source: AstraZeneca Annual Reports & Investor Filings
Peak sales for Crestor were achieved around 2012-2013. The sharp decline in sales from 2016 onwards is directly attributable to the loss of market exclusivity and the subsequent entry of multiple generic competitors.
What is the Current Market Status of Crestor and its Generics?
The market for rosuvastatin calcium is now highly genericized. The introduction of generics has led to a significant reduction in price per dosage unit, increasing accessibility but substantially decreasing the revenue generated by the original branded product.
- Branded Crestor: Sales of branded Crestor have become negligible in most major markets due to generic competition. AstraZeneca has largely transitioned its focus to newer therapeutic areas and pipeline assets.
- Generic Rosuvastatin Calcium: The generic market is highly fragmented, with numerous pharmaceutical companies producing and marketing rosuvastatin calcium. This competition drives down prices and increases market share for generic manufacturers. Major generic players include Teva Pharmaceuticals, Mylan (now Viatris), Cipla, Sun Pharmaceutical Industries, and numerous others.
- Market Share: While precise real-time market share data for individual generics is difficult to ascertain due to the proprietary nature of sales data, it is understood that the collective market share of generic rosuvastatin calcium far exceeds that of branded Crestor.
What are the Key Market Drivers and Restraints for Rosuvastatin Calcium?
Market Drivers:
- Prevalence of Cardiovascular Disease: High and increasing rates of cardiovascular disease globally, driven by lifestyle factors such as poor diet, lack of physical activity, and rising obesity, create a sustained demand for effective lipid-lowering therapies.
- Established Efficacy and Safety Profile: Rosuvastatin calcium has a well-documented history of efficacy in reducing LDL cholesterol and cardiovascular events. Its safety profile is also well-understood, making it a preferred choice for many healthcare providers and patients.
- Affordability of Generics: The availability of affordable generic rosuvastatin calcium significantly expands patient access, particularly in cost-sensitive healthcare systems and for uninsured or underinsured populations.
- Growing Geriatric Population: The aging global population is at higher risk for cardiovascular events, increasing the demand for statin therapies like rosuvastatin.
- Proactive Cardiovascular Risk Management: An increasing emphasis on preventative healthcare and the proactive management of cardiovascular risk factors contributes to the sustained use of statins.
Market Restraints:
- Intense Generic Competition: The highly competitive nature of the generic rosuvastatin market leads to significant price erosion, limiting revenue potential for any single manufacturer.
- Emergence of Newer Lipid-Lowering Therapies: While statins remain first-line therapy, novel drug classes such as PCSK9 inhibitors offer alternative or adjunctive treatment options for specific patient populations, potentially impacting the market share of older statins over time.
- Side Effect Concerns: Although generally well-tolerated, potential side effects of statins, including myalgia, liver enzyme elevation, and rare cases of rhabdomyolysis, can lead to patient non-adherence or discontinuation of treatment.
- Prescribing Guidelines Evolution: Changes in clinical guidelines or the emergence of new evidence may influence prescribing patterns, although rosuvastatin's established position makes significant shifts less likely.
- Drug Interactions: Rosuvastatin can interact with certain medications, requiring careful patient management and potentially limiting its use in patients taking multiple concomitant drugs.
What are the Competitive Dynamics within the Rosuvastatin Calcium Market?
The competitive landscape is dominated by generic manufacturers. Competition is primarily based on:
- Price: Given the generic nature of the market, competitive pricing is paramount for market share acquisition.
- Distribution Networks: Establishing robust distribution channels to reach pharmacies and healthcare providers globally is crucial.
- Manufacturing Capacity and Quality: Ensuring consistent supply and high-quality product is essential to maintain customer trust.
- Formulation Variations: While the core molecule is rosuvastatin calcium, some manufacturers may offer different salt forms or excipient combinations, though these do not represent significant therapeutic differentiation.
Key generic players actively competing include:
- Teva Pharmaceuticals Industries Ltd.
- Viatris Inc. (formed from the merger of Mylan and Pfizer's Upjohn)
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Dr. Reddy's Laboratories Ltd.
- Aurobindo Pharma Ltd.
- Lupin Ltd.
The branded Crestor still holds some market presence in specific regions or for patient preference, but its market share is marginal.
What are the Projected Sales for Branded Crestor and Generic Rosuvastatin Calcium?
Branded Crestor: Global sales for branded Crestor are projected to remain minimal, likely below $50 million annually, as market penetration by generics is nearly complete. AstraZeneca has largely divested from the branded product's sales efforts.
Generic Rosuvastatin Calcium: The global market for generic rosuvastatin calcium is substantial and is projected to remain robust, albeit with low single-digit annual growth due to price pressure and competition from other statin generics and novel therapies.
- Current Market Size (Estimate): The global market for generic rosuvastatin calcium is estimated to be in the range of $3 billion to $4 billion annually.
- Projected Growth: The market is expected to grow at a Compound Annual Growth Rate (CAGR) of 1% to 3% over the next five years (2024-2029). This modest growth reflects the mature nature of the product, price competition, and the increasing use of newer drug classes for certain patient segments.
- Regional Variations: North America and Europe are mature markets with high generic penetration. Emerging markets in Asia-Pacific and Latin America are expected to exhibit slightly higher growth rates as healthcare access improves and generic availability increases.
The overall volume of rosuvastatin calcium prescriptions is expected to remain high, driven by its established role in cardiovascular disease management. However, the revenue generated will continue to be constrained by pricing dynamics.
What are the Key Takeaways for R&D and Investment Decisions?
The market for rosuvastatin calcium is mature and heavily genericized. For branded Crestor, revenue generation is negligible. Investment focus for rosuvastatin calcium should center on generic manufacturing and distribution.
- Generic Manufacturing Efficiency: Companies with optimized manufacturing processes and economies of scale will hold a competitive advantage in producing low-cost rosuvastatin calcium.
- Supply Chain Reliability: Ensuring a stable and reliable supply chain is critical for generic market share.
- Emerging Market Penetration: Growth opportunities exist in emerging economies where access to affordable statins is expanding.
- Pipeline Diversification: For pharmaceutical companies, the focus should be on developing novel therapies for dyslipidemia and cardiovascular disease, rather than investing in the generic rosuvastatin market for significant growth.
- Limited M&A Potential: The low margins and fragmented nature of the generic rosuvastatin market offer limited attractive M&A opportunities for large pharmaceutical firms seeking high returns. Acquisitions might be considered by established generic players looking to consolidate market share or expand their product portfolios.
Frequently Asked Questions
- Are there any remaining patents protecting branded Crestor? While foundational composition of matter and key formulation patents have expired, certain secondary patents related to specific manufacturing processes or novel delivery systems might still exist. However, these are unlikely to provide broad market exclusivity against generic competition.
- What are the primary reasons for the decline in branded Crestor sales? The primary driver for the decline in branded Crestor sales was the loss of market exclusivity in major pharmaceutical markets, allowing generic manufacturers to introduce lower-cost versions of rosuvastatin calcium.
- What is the typical price difference between branded Crestor and generic rosuvastatin calcium? Generic rosuvastatin calcium can be priced anywhere from 80% to 95% lower than the original branded Crestor, depending on the market, manufacturer, and dosage strength.
- Will rosuvastatin calcium remain a first-line treatment for hypercholesterolemia? Yes, rosuvastatin calcium is expected to remain a first-line treatment option for hypercholesterolemia and cardiovascular risk reduction due to its established efficacy, safety profile, and affordability as a generic. However, newer therapies will likely be used adjunctively or for specific patient populations.
- Which companies are the largest suppliers of generic rosuvastatin calcium globally? Major global suppliers of generic rosuvastatin calcium include Teva Pharmaceuticals, Viatris Inc., Sun Pharmaceutical Industries Ltd., and Cipla Ltd., among other large generic drug manufacturers.
Citations
[1] AstraZeneca PLC. (Annual Reports 2010-2022). Company Financial Statements.
[2] U.S. Food and Drug Administration. (Prescription Drug Information). Rosuvastatin Calcium Drug Label Information. Retrieved from FDA database.
[3] United States Patent and Trademark Office. (Patent Records). USPTO Public Patent Database. Retrieved from USPTO website.
More… ↓
