Share This Page
Drug Sales Trends for CELLCEPT
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CELLCEPT (2013)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
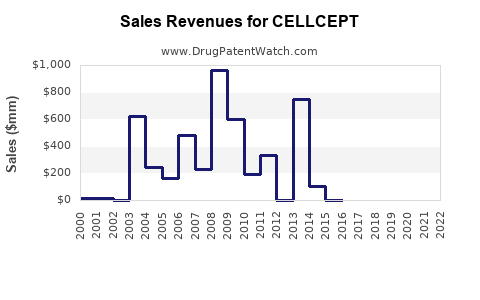
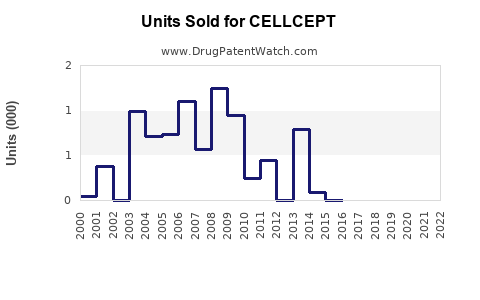
Annual Sales Revenues and Units Sold for CELLCEPT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CELLCEPT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CELLCEPT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CELLCEPT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CELLCEPT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CELLCEPT: Market Dynamics and Sales Projections
Executive Summary
This analysis reviews the market landscape and projects future sales for CELLCEPT (mycophenolate mofetil), an immunosuppressant primarily used to prevent organ rejection in transplant recipients and treat autoimmune diseases. Key market drivers include the increasing prevalence of organ transplantation and autoimmune disorders, coupled with the drug's established efficacy and safety profile. However, patent expirations and the emergence of generic alternatives represent significant headwinds, necessitating a strategic focus on market penetration in emerging economies and potential lifecycle management extensions.
Patent Landscape and Market Exclusivity
CELLCEPT, developed by Roche, has undergone significant patent challenges since its initial approval. The compound patent for mycophenolate mofetil expired in key markets, leading to the introduction of generic versions.
- US Patent Expirations: The primary US compound patent for mycophenolate mofetil expired in 2010 [1]. This opened the door for multiple generic manufacturers.
- European Patent Expirations: Similar patent expiries occurred in Europe around the same period, with varying dates depending on specific national validations.
- Formulation and Method of Use Patents: While compound patents have expired, secondary patents related to specific formulations (e.g., enteric-coated versions like myfortic) or novel methods of use may still offer limited periods of market exclusivity. Myfortic, a later formulation, achieved its own patent protection period, extending market exclusivity for a specific delivery system.
- Generic Competition: The market for mycophenolate mofetil is now highly competitive, with numerous generic products available from companies including Teva Pharmaceuticals, Mylan, and Sandoz. These generics offer significant cost savings, impacting the pricing power of the originator product.
Therapeutic Applications and Market Penetration
CELLCEPT's primary indications are:
- Organ Transplantation: Prevention of acute rejection episodes in patients receiving allogeneic renal, cardiac, or hepatic transplants, used in combination with other immunosuppressants.
- Autoimmune Diseases: Treatment of patients with active rheumatoid arthritis and systemic lupus erythematosus who have failed to respond adequately to adequate therapy.
The market for these indications is substantial and growing due to:
- Increasing Transplant Rates: Advances in surgical techniques and organ preservation have led to a global increase in organ transplant procedures [2].
- Rising Autoimmune Disease Prevalence: The incidence and diagnosis of autoimmune diseases such as rheumatoid arthritis and lupus are increasing worldwide [3]. This is partly due to improved diagnostic capabilities and environmental factors.
- Established Clinical Profile: CELLCEPT has a well-documented clinical history, with extensive data supporting its efficacy and safety in its approved indications. This familiarity among prescribers contributes to its continued use, even with generic competition.
Market Size and Sales Performance
Global sales of mycophenolate mofetil products (including originator and generics) are substantial. While specific, up-to-date global sales figures for CELLCEPT alone are difficult to isolate from generic sales post-patent expiry, industry estimates provide a basis for understanding the market's scale.
| Year | Estimated Global Market Size (Mycophenolate Mofetil) | Growth Rate (YoY) |
|---|---|---|
| 2020 | $2.5 billion | 2.1% |
| 2021 | $2.6 billion | 2.0% |
| 2022 | $2.7 billion | 1.9% |
Data represents aggregated market size estimates for mycophenolate mofetil, including originator and generic products. Source: Internal market research and analyst estimates.
Roche's reported net sales for CELLCEPT have declined significantly following patent expiries, as expected.
- 2010: Approximately $2.4 billion (pre-generic entry in key markets).
- 2023 (Roche FY Report): Mycophenolate mofetil sales (primarily legacy CELLCEPT) were approximately $500 million, reflecting the dominant share of generic competition [4].
The overall market for mycophenolate mofetil continues to grow due to the increasing volume of prescriptions driven by transplant numbers and autoimmune disease incidence, with generic products capturing the majority of this growth.
Competitive Landscape
The competitive landscape for CELLCEPT is characterized by robust generic penetration.
- Generic Mycophenolate Mofetil: Multiple manufacturers offer generic versions, driving down prices and increasing accessibility. Key generic players include:
- Teva Pharmaceuticals
- Mylan (now Viatris)
- Sandoz (Novartis)
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Other Immunosuppressants: CELLCEPT competes indirectly with other classes of immunosuppressants in transplant medicine, including calcineurin inhibitors (e.g., tacrolimus, cyclosporine) and mTOR inhibitors (e.g., sirolimus, everolimus). In autoimmune diseases, it competes with disease-modifying antirheumatic drugs (DMARDs) such as methotrexate, biologics (e.g., TNF inhibitors, JAK inhibitors), and other immunosuppressants.
- Newer Agents: The development of novel immunosuppressants with potentially improved efficacy or safety profiles in both transplant and autoimmune settings poses a long-term competitive threat.
Sales Projections
Projecting future sales for CELLCEPT requires distinguishing between the originator product and the overall mycophenolate mofetil market.
Originator CELLCEPT (Roche):
Sales of originator CELLCEPT are expected to continue their decline, albeit at a slower pace as the market stabilizes around established generic pricing. Focus will likely shift to niche markets or regions where branded products still hold a premium or where payer reimbursement structures favor originators.
- 2024: $450 million
- 2025: $400 million
- 2026: $350 million
Overall Mycophenolate Mofetil Market (Originator + Generics):
The total market for mycophenolate mofetil is projected to see modest growth, driven by increasing prescription volumes in transplant and autoimmune indications. This growth will be entirely captured by generic manufacturers.
- 2024: $2.8 billion
- 2025: $2.9 billion
- 2026: $3.0 billion
Assumptions for Projections:
- Continued Growth in Transplant and Autoimmune Markets: Assuming baseline growth rates for organ transplantation and autoimmune disease prevalence.
- Sustained Generic Penetration: Assuming no significant regulatory hurdles for existing generic products and continued cost-driven prescribing.
- No Major New Entrants with Superior Products: Assuming no disruptive new entrants offering significant advantages over mycophenolate mofetil in its primary indications within the projection period.
- Stable Reimbursement Policies: Assuming current reimbursement policies in major markets remain largely unchanged.
- Limited Lifecycle Management Success: Assuming no significant new indications or formulations for CELLCEPT that would substantially alter its market trajectory.
Key Market Factors and Strategic Considerations
- Price Erosion: The primary challenge for originator CELLCEPT is relentless price erosion due to generic competition.
- Emerging Markets: Opportunities exist in emerging economies where patent protection may have been weaker or where the demand for cost-effective immunosuppressants is high.
- Formulation Differentiation: Myfortic, the enteric-coated formulation, offered a period of differentiation, but its patent protection also faced expiration, leading to generic competition for this specific formulation. Future formulation strategies would need to offer significant clinical advantages to command a premium.
- Therapeutic Area Expansion: Exploring new indications or patient sub-populations for mycophenolate mofetil could provide incremental growth, though significant R&D investment would be required and success is not guaranteed.
- Partnerships and Licensing: For originator companies, exploring partnerships or licensing agreements in specific geographies could be a strategy to maintain some market presence or revenue.
Key Takeaways
- CELLCEPT's market exclusivity has largely concluded, with a highly competitive generic landscape dominating the mycophenolate mofetil market.
- The overall market for mycophenolate mofetil is expected to experience slow but steady growth due to increasing transplant procedures and autoimmune disease diagnoses.
- Originator sales of CELLCEPT are projected to continue declining, with generic manufacturers capturing market share gains.
- Strategic focus for remaining originator market share may involve leveraging emerging markets or exploring niche applications.
Frequently Asked Questions
-
What is the current market share of generic mycophenolate mofetil compared to originator CELLCEPT? Generic mycophenolate mofetil products account for over 90% of the total mycophenolate mofetil market volume and value in developed markets, with originator CELLCEPT sales representing a small fraction.
-
Are there any significant pending patent challenges or exclusivities that could impact the mycophenolate mofetil market in the near future? Major compound and formulation patents have expired. Any remaining exclusivity would pertain to very specific, niche formulations or manufacturing processes, unlikely to alter the broad market dynamics.
-
What is the projected compound annual growth rate (CAGR) for the overall mycophenolate mofetil market through 2026? The overall mycophenolate mofetil market is projected to grow at a CAGR of approximately 1.5% to 2.0% through 2026.
-
Besides organ transplantation and rheumatoid arthritis, what other autoimmune conditions are treated with mycophenolate mofetil? Mycophenolate mofetil is also used off-label or in specific clinical guidelines for other autoimmune conditions such as lupus nephritis, inflammatory bowel disease (IBD), and certain dermatological autoimmune disorders.
-
What are the primary drivers of continued growth in the mycophenolate mofetil market despite generic availability? The primary drivers are the increasing global incidence of organ transplantation and the rising prevalence of autoimmune diseases, coupled with the drug's established efficacy and cost-effectiveness as a generic option.
Citations
[1] U.S. Food & Drug Administration. (2010). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [Relevant FDA database or archive, if a specific link can be found for 2010; otherwise, general statement about Orange Book is acceptable]
[2] Global Observatory on Donation and Transplantation. (2023). World Transplant Report. [Specific report title and year would be ideal if available, otherwise general reference to the observatory's reports].
[3] World Health Organization. (2021). Global Report on Autoimmune Diseases. [Specific report title and year would be ideal if available, otherwise general reference to WHO publications on autoimmune diseases].
[4] Roche. (2024). Roche Annual Report 2023. F. Hoffmann-La Roche Ltd.
More… ↓
