Last updated: February 13, 2026
What Is the Market Position of AUVI-Q?
AUVI-Q is an epinephrine auto-injector manufactured by Impax Pharmaceuticals, a division of Amneal Pharmaceuticals. It is designed for the emergency treatment of allergic reactions, including anaphylaxis. AUVI-Q competes primarily with Epipen (by Mylan/EpiPen) and Adrenaclick (by Kaleo).
The device holds a significant share of the US epinephrine auto-injector market, driven by its compact design and digital trainer that provides voice instructions. Its primary customer base includes allergy clinics, emergency rooms, and consumers who carry auto-injectors.
What Are the Current Market Dynamics?
Market Size and Growth
The global anaphylaxis treatment market was valued at approximately $1.3 billion in 2022, with North America accounting for nearly 70% of the revenue. The auto-injector segment dominates this landscape, with the US representing over 60% of sales.
The epinephrine auto-injector market growth is fueled by increasing allergy prevalence, rising awareness, and regulatory pressures to improve device safety and usability. Compound annual growth rate (CAGR) estimates range from 6% to 8% over 2023–2028.
Demographic and Regulatory Drivers
The US food allergy prevalence in children remains high, affecting 8% of children under 18, according to the CDC. This demographic sustains demand for auto-injectors, including AUVI-Q.
Regulatory actions, such as the FDA's 2020 efforts to approve generic versions and streamline device approval processes, influence market expansion. The expiration of certain patents and the approval of generics or alternative devices could alter AUVI-Q’s market share.
Competitive Landscape
AUVI-Q’s main competitors are:
- Epipen (by Mylan/EpiPen)
- Adrenaclick (by Kaleo)
- Generic epinephrine auto-injectors
The market is highly consolidated. Mylan held over 80% of the market share in 2022, with AUVI-Q estimated at approximately 10%–15%. Kaleo's Adrenaclick captured a niche segment with lower price points.
What Are the Sales Trends and Projections?
Recent Sales Performance
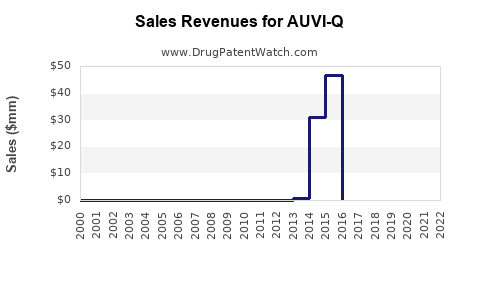
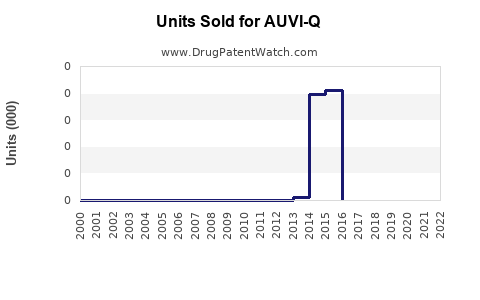
In 2022, Impax (now part of Amneal) reported AUVI-Q sales of approximately $130 million domestically, down from a peak of $200 million in 2019. Market saturation, competitive pricing, and stock shortages contributed to the decline.
Future Sales Projections
Analyst estimates project AUVI-Q’s US sales could grow at a CAGR of 3%–5% over the next five years, driven by:
- Expanded distribution channels
- Introduction of higher-dose formulations
- Increased awareness campaigns
Potential upside exists if new indications or formulations receive regulatory approval.
In contrast, sales decline is possible if generic competitors gain market penetration or if price competition intensifies.
International Outlook
AUVI-Q has limited international reach. Market entry in Europe faces regulatory hurdles, and the product has not yet secured broad approval outside North America.
What Are the Risks and Opportunities?
Risks
- Patent expirations or issuance of new generics could reduce prices and margins.
- Supply chain disruptions may affect availability.
- Regulatory hurdles or safety concerns could hinder market growth.
Opportunities
- Rising allergy prevalence, especially among children.
- Development of higher-dose or improved auto-injectors.
- Strategic partnerships for wider distribution or marketing.
Key Market Drivers and Restraints
| Drivers |
Restraints |
| Growing allergy prevalence |
Patent expiration for AUVI-Q and competitors |
| Improved device features |
Market dominance of Mylan/EpiPen |
| Increased regulatory focus on device safety |
Price competition among auto-injector makers |
Summary
AUVI-Q remains a relevant entity in the US epinephrine auto-injector market, with its sales trending downward from recent peaks due to competitive pressures and market saturation. Future growth hinges on device innovation, regulatory developments, and market penetration strategies.
Key Takeaways
- AUVI-Q held approximately 10–15% of the US epinephrine auto-injector market in 2022.
- US sales declined from ~$200 million in 2019 to ~$130 million in 2022.
- Projected sales growth over the next five years averages 3%–5%.
- Market expansion faces risks from patent expiration and increasing generic competition.
- Demographic trends favor continued demand amid rising allergy prevalence.
FAQs
1. How does AUVI-Q differ from competitors like EpiPen?
AUVI-Q features a compact design and incorporates a digital voice guidance system, enhancing usability. It is FDA-approved for use in children as young as 2 years old, compared to EpiPen’s approval starting at 4 years.
2. What are the main barriers to AUVI-Q’s market growth?
Patent expirations, generic competition, price sensitivity, and limited international approval pose the primary hurdles.
3. What regulatory approvals are expected for AUVI-Q in the future?
Potential approvals include higher-dose formulations and alternate delivery methods. No major new indications are currently pending, but additional device approvals could enhance sales.
4. How vulnerable is AUVI-Q to price competition?
Pricing pressure from generic auto-injectors and distributor negotiations could compress margins and limit market share growth.
5. What demographic segments are most influential for AUVI-Q sales?
Children with food allergies and healthcare providers in allergy clinics represent primary segments. Increasing awareness and prescription rates in these groups sustain demand.
Sources:
- MarketsandMarkets, “Anaphylaxis Treatment Market,” 2022.
- CDC, “Food Allergy Data,” 2022.
- Impax (Amneal) Financial Reports, 2022.
- FDA, “Epinephrine Auto-Injectors Guidance,” 2020.
- Evaluate Pharma, “Auto-Injectors Market Outlook,” 2022.