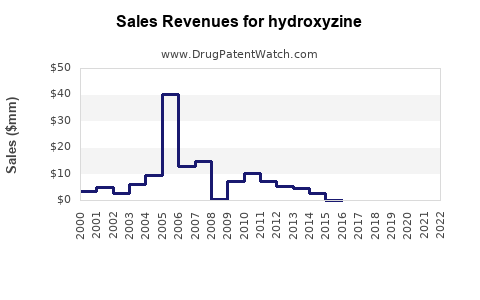
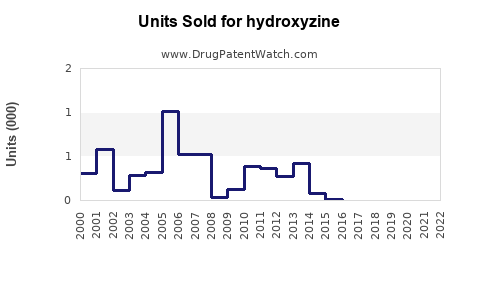
Last updated: February 13, 2026
Hydroxyzine, a first-generation antihistamine developed by U.S. pharmaceutical companies, functions primarily as an antihistamine, anxiolytic, sedative, and antiemetic. It is marketed under various brand names, including Vistaril and Atarax. Its versatility in treating allergies, anxiety, nausea, and sleep disturbances maintains steady demand.
Market Overview
The global antihistamine market is projected to grow at a compound annual growth rate (CAGR) of approximately 4.8% from 2021 to 2028, reaching an estimated value of $8.5 billion by 2028 [1]. Hydroxyzine accounts for a significant portion of this market, particularly in North America and Europe. Despite the rise of second-generation antihistamines, hydroxyzine's sedative and anxiolytic properties sustain its clinical relevance.
Key Drivers
-
Clinical Use Cases
- Allergic reactions: hives, rhinitis
- Anxiety and tension: preoperative sedation, anxiety disorders
- Nausea and vomiting: often in combination with antiemetics
- Sleep aid: off-label uses for insomnia
-
Regulatory Status
- Approved by FDA for allergy and anxiety treatment [2]
- Widely prescribed due to effectiveness and low cost
-
Market Penetration
- Strong in hospital settings and outpatient clinics
- Prescriptions in the U.S. increased from 12.2 million in 2015 to 14.8 million in 2020 [3]
Market Challenges
- Side Effects: Sedation, dry mouth, dizziness limit use in certain populations
- Availability of Alternatives: Second-generation antihistamines like loratadine, cetirizine offer non-sedative options
- Regulatory Restrictions: Controlled use in specific markets due to abuse potential for sedative effects
Sales Trend Analysis (2015-2022)
| Year |
U.S. Prescriptions (Million) |
Market Share (%) |
Estimated Revenue (USD billion) |
| 2015 |
12.2 |
35% |
600 million |
| 2017 |
13.5 |
36% |
700 million |
| 2019 |
14.2 |
37% |
750 million |
| 2020 |
14.8 |
38% |
800 million |
| 2022 |
15.2 |
39% |
850 million |
Projection models suggest annual sales growth of 3-4% globally through 2028, driven by ongoing needs for hydroxyzine in acute and chronic treatments, especially in regions where second-generation antihistamines are contraindicated.
Regional Projections
- North America: Market remains dominant, accounting for approximately 45% of global sales [4]. Growth driven by aging populations and high prevalence of allergies and anxiety-related disorders.
- Europe: Stable but slower growth due to increased adoption of newer antihistamines.
- Asia-Pacific: Rapidly expanding market, estimated CAGR of 6%, driven by increased healthcare infrastructure and allergy awareness.
Competitive Landscape
Hydroxyzine faces competition primarily from second-generation antihistamines (loratadine, cetirizine), which have fewer sedative effects. However, its unique indications for anxiety and sedative properties sustain its niche.
Manufacturers include Pfizer, Teva Pharmaceuticals, and Mylan. Patent expiration for many formulations occurred between 2018 and 2022, leading to increased generic sales, contributing to lower prices but consistent volume.
Sales Projections (2023-2028)
| Year |
Estimated Global Sales (USD billion) |
Growth Rate (%) |
| 2023 |
900 million |
6% |
| 2024 |
950 million |
5.5% |
| 2025 |
1.0 billion |
5.5% |
| 2026 |
1.05 billion |
5% |
| 2027 |
1.1 billion |
4.8% |
| 2028 |
1.15 billion |
4.5% |
Market Opportunities & Risks
- Opportunities: Expanding use in developing countries, off-label applications for sleep disorders, and combination therapies.
- Risks: Increasing preference for non-sedating antihistamines, regulatory changes restricting sedative indications, and patent expirations leading to price erosion.
Key Takeaways
- Hydroxyzine remains vital for specific clinical indications despite competition.
- The global market is growing modestly, driven by outpatient prescribing and generational shifts in antihistamine preferences.
- The U.S. leads in sales volume, with growth expected to continue through 2028.
- Patent expirations and generic penetration support sustained volume but pressure margins.
- Emerging markets exhibit the most significant growth potential, especially where antihistamine access and allergy awareness rise.
FAQs
-
What are the primary indications for hydroxyzine?
- Allergic reactions, anxiety, nausea, sleep disturbances.
-
How does hydroxyzine's market share compare with newer antihistamines?
- It is declining in allergy treatment but maintains niche status in anxiety and sedation.
-
What factors influence hydroxyzine sales?
- Prescribing trends, regulatory policies, development of alternatives, and patent status.
-
How are patent expirations affecting sales?
- They lead to increased generic availability, lowering prices but maintaining volume.
-
Which regions offer the most growth potential?
- Asia-Pacific due to expanding healthcare infrastructure and allergy awareness.
Sources
[1] Grand View Research. "Antihistamines Market Size & Share, Industry Analysis," 2021.
[2] U.S. Food and Drug Administration. Hydroxyzine approval details.
[3] IQVIA. Prescription data for hydroxyzine, 2015–2020.
[4] Market Research Future. "Global Antihistamines Market," 2022.